Maraviroc clinical pharmacology
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ahmed Zaghw, M.D. [2]
Clinical Pharmacology
Mechanism of Action
Maraviroc is an antiviral drug.
Pharmacodynamics
Exposure Response Relationship in Treatment-Experienced Subjects
The relationship between maraviroc, modeled plasma trough concentration (Cmin) (1–9 samples per patient taken on up to 7 visits), and virologic response was evaluated in 973 treatment-experienced HIV-1-infected subjects with varied optimized background antiretroviral regimens in studies A4001027 and A4001028. The Cmin, baseline viral load, baseline CD4+ cell count and overall sensitivity score (OSS) were found to be important predictors of virologic success (defined as viral load < 400 copies/mL at 24 weeks). Table 7 illustrates the proportion of subjects with virologic success (%) within each Cmin quartile for 150 mg twice daily and 300 mg twice daily groups.
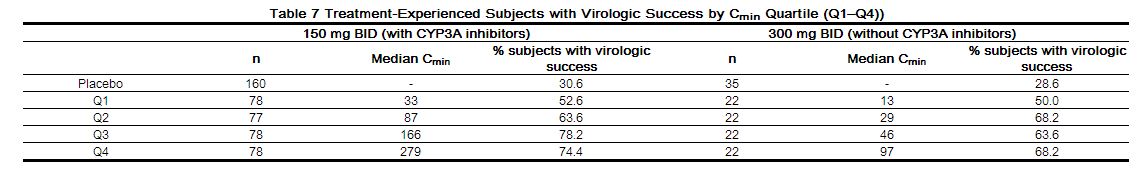 |
Exposure Response Relationship in Treatment-Naive Subjects
The relationship between maraviroc, modeled plasma trough concentration (Cmin) (1–12 samples per patient taken on up to 8 visits), and virologic response was evaluated in 294 treatment-naive HIV-1-infected subjects receiving maraviroc 300 mg twice daily in combination with zidovudine/lamivudine in study A4001026. Table 8 illustrates the proportion (%) of subjects with virologic success < 50 copies/mL at 48 weeks within each Cmin quartile for the 300 mg twice daily dose.
 |
Effects on Electrocardiogram
A placebo-controlled, randomized, crossover study to evaluate the effect on the QT interval of healthy male and female volunteers was conducted with three single oral doses of maraviroc and moxifloxacin. The placebo-adjusted mean maximum (upper 1-sided 95% CI) increases in QTc from baseline after 100, 300 and 900 mg of maraviroc were –2 (0), -1 (1), and 1 (3) msec, respectively, and 13 (15) msec for moxifloxacin 400 mg. No subject in any group had an increase in QTc of ≥60 msec from baseline. No subject experienced an interval exceeding the potentially clinically relevant threshold of 500 msec.
Pharmacokinetics
 |
Absorption
Peak maraviroc plasma concentrations are attained 0.5–4h following single oral doses of 1–1200 mg administered to uninfected volunteers. The pharmacokinetics of oral maraviroc are not dose-proportional over the dose range.
The absolute bioavailability of a 100 mg dose is 23% and is predicted to be 33% at 300 mg. Maraviroc is a substrate for the efflux transporter P-glycoprotein.
Effect of Food on Oral Absorption
Coadministration of a 300mg tablet with a high fat breakfast reduced maraviroc Cmax and AUC by 33% in healthy volunteers. There were no food restrictions in the studies that demonstrated the efficacy and safety of maraviroc [see Clinical Studies (14)]. Therefore, maraviroc can be taken with or without food at the recommended dose [see Dosage and Administration (2)].
Distribution
Maraviroc is bound (approximately 76%) to human plasma proteins, and shows moderate affinity for albumin and alpha-1 acid glycoprotein. The volume of distribution of maraviroc is approximately 194L.
Metabolism
Studies in humans and in vitro studies using human liver microsomes and expressed enzymes have demonstrated that maraviroc is principally metabolized by the cytochrome P450 system to metabolites that are essentially inactive against HIV-1. In vitro studies indicate that CYP3A is the major enzyme responsible for maraviroc metabolism. In vitro studies also indicate that polymorphic enzymes CYP2C9, CYP2D6 and CYP2C19 do not contribute significantly to the metabolism of maraviroc.
Maraviroc is the major circulating component (~42% drug-related radioactivity) following a single oral dose of 300 mg[14C]-maraviroc. The most significant circulating metabolite in humans is a secondary amine (~22% radioactivity) formed by N-dealkylation. This polar metabolite has no significant pharmacological activity. Other metabolites are products of mono-oxidation and are only minor components of plasma drug-related radioactivity.
Excretion=
The terminal half-life of maraviroc following oral dosing to steady state in healthy subjects was 14–18 hours. A mass balance/excretion study was conducted using a single 300mg dose of 14C-labeled maraviroc. Approximately 20% of the radiolabel was recovered in the urine and 76% was recovered in the feces over 168 hours. Maraviroc was the major component present in urine (mean of 8% dose) and feces (mean of 25% dose). The remainder was excreted as metabolites.
Hepatic Impairment
Maraviroc is primarily metabolized and eliminated by the liver. A study compared the pharmacokinetics of a single 300 mg dose of SELZENTRY in subjects with mild (Child-Pugh Class A, n=8), and moderate (Child-Pugh Class B, n=8) hepatic impairment to pharmacokinetics in healthy subjects (n=8). The mean Cmax and AUC were 11% and 25% higher, respectively, for subjects with mild hepatic impairment, and 32% and 46% higher, respectively, for subjects with moderate hepatic impairment compared to subjects with normal hepatic function. These changes do not warrant a dose adjustment. Maraviroc concentrations are higher when SELZENTRY 150 mg is administered with a potent CYP3A inhibitor compared to following administration of 300 mg without a CYP3A inhibitor, so patients with moderate hepatic impairment who receive SELZENTRY 150 mg with a potent CYP3A inhibitor should be monitored closely for maraviroc-associated adverse events. The pharmacokinetics of maraviroc have not been studied in subjects with severe hepatic impairment.
Renal Impairment
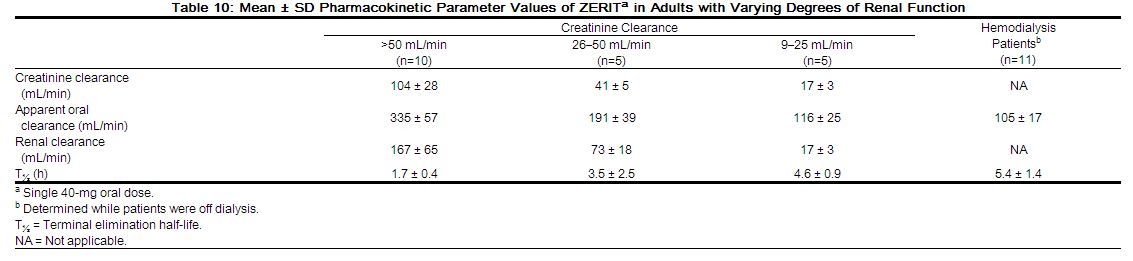
A study compared the pharmacokinetics of a single 300 mg dose of SELZENTRY in subjects with severe renal impairment (CLcr < 30 mL/min, n=6) and end-stage renal disease (ESRD) (n=6) to healthy volunteers (n=6). Geometric mean ratios for maraviroc Cmax and AUCinf were 2.4-fold and 3.2-fold higher respectively for subjects with severe renal impairment, and 1.7-fold and 2.0-fold higher respectively for subjects with ESRD as compared to subjects with normal renal function in this study. Hemodialysis had a minimal effect on maraviroc clearance and exposure in subjects with ESRD. Exposures observed in subjects with severe renal impairment and ESRD were within the range observed in previous SELZENTRY 300 mg single-dose studies in healthy volunteers with normal renal function. However, maraviroc exposures in the subjects with normal renal function in this study were 50% lower than that observed in previous studies. Based on the results of this study, no dose adjustment is recommended for patients with renal impairment receiving SELZENTRY without a potent CYP3A inhibitor or inducer. However, if patients with severe renal impairment or ESRD experience any symptoms of postural hypotension while taking SELZENTRY 300 mg twice daily, their dose should be reduced to 150 mg twice daily [see Dosage and Administration (2.2); Warnings and Precautions (5.2)].
In addition, the study compared the pharmacokinetics of multiple dose SELZENTRY in combination with saquinavir/ritonavir 1000/100 mg twice daily (a potent CYP3A inhibitor combination) for 7 days in subjects with mild renal impairment (CLcr >50 and ≤80 mL/min, n=6) and moderate renal impairment (CLcr ≥30 and ≤50 mL/min, n=6) to healthy volunteers with normal renal function (n=6). Subjects received 150 mg of SELZENTRY at different dose frequencies (healthy volunteers – every 12 hours; mild renal impairment – every 24 hours; moderate renal impairment – every 48 hours). Compared to healthy volunteers (dosed every 12 hours), geometric mean ratios for maraviroc AUCtau, Cmaxand Cmin were 50% higher, 20% higher and 43% lower, respectively for subjects with mild renal impairment (dosed every 24 hours). Geometric mean ratios for maraviroc AUCtau, Cmax and Cmin were 16% higher, 29% lower and 85% lower, respectively for subjects with moderate renal impairment (dosed every 48 hours) compared to healthy volunteers (dosed every 12 hours). Based on the data from this study, no adjustment in dose is recommended for patients with mild or moderate renal impairment [see Dosage and Administration (2.2)].
Effect of Concomitant Drugs on the Pharmacokinetics of Maraviroc
Maraviroc is a substrate of CYP3A and Pgp and hence its pharmacokinetics are likely to be modulated by inhibitors and inducers of these enzymes/transporters. The CYP3A/Pgp inhibitors ketoconazole, lopinavir/ritonavir, ritonavir, darunavir/ritonavir, saquinavir/ritonavir and atazanavir ± ritonavir all increased the Cmax and AUC of maraviroc [see Table 10]. The CYP3A inducers rifampin, etravirine and efavirenz decreased the Cmax and AUC of maraviroc [see Table 10].
Tipranavir/ritonavir (net CYP3A inhibitor/Pgp inducer) did not affect the steady state pharmacokinetics of maraviroc (see Table 10). Co-trimoxazole and tenofovir did not affect the pharmacokinetics of maraviroc.
 |
Effect of Maraviroc on the Pharmacokinetics of Concomitant Drugs
Maraviroc is unlikely to inhibit the metabolism of coadministered drugs metabolized by the following cytochrome P enzymes (CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, and CYP3A) because maraviroc did not inhibit activity of those enzymes at clinically relevant concentrations in vitro. Maraviroc does not induce CYP1A2 in vitro.
In vitro results indicate that maraviroc could inhibit P-glycoprotein in the gut and may thus affect bioavailability of certain drugs.
Drug interaction studies were performed with maraviroc and other drugs likely to be coadministered or commonly used as probes for pharmacokinetic interactions [see Table 6]. Maraviroc had no effect on the pharmacokinetics of zidovudine or lamivudine. Maraviroc decreased the Cmin and AUC of raltegravir by 27% and 37%, respectively, which is not clinically significant. Maraviroc had no clinically relevant effect on the pharmacokinetics of midazolam, the oral contraceptives ethinylestradiol and levonorgestrel, no effect on the urinary 6β-hydroxycortisol/cortisol ratio, suggesting no induction of CYP3A in vivo. Maraviroc had no effect on the debrisoquine metabolic ratio (MR) at 300 mg twice daily or less in vivo and did not cause inhibition of CYP2D6 in vitro until concentrations >100µM. However, there was 234% increase in debrisoquine MR on treatment compared to baseline at 800 mg once daily, suggesting potential inhibition of CYP2D6 at higher dose.[1]
References
Adapted from the FDA Package Insert.