Iclaprim
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral, intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | Good (oral)[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
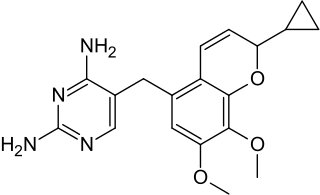
| Formula | C19H22N4O3 |
| Molar mass | 354.403 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
|
WikiDoc Resources for Iclaprim |
|
Articles |
|---|
|
Most recent articles on Iclaprim |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Iclaprim at Clinical Trials.gov Clinical Trials on Iclaprim at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Iclaprim
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Directions to Hospitals Treating Iclaprim Risk calculators and risk factors for Iclaprim
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Iclaprim |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Iclaprim (INN), codenamed AR-100 and RO-48-2622, is a diaminopyrimidine dihydrofolate reductase (DHFR)-inhibiting extended-spectrum antibiotic active against gram positive organisms being developed for the treatment of complicated skin and soft tissue infections caused by antibiotic-resistant bacteria.[2] It is structurally related to trimethoprim. In Phase III clinical trials, intravenously-administered iclaprim was found to be as effective as and better tolerated than linezolid in people with skin and soft tissue infections, many caused by methicillin-resistant Staphylococcus aureus (MRSA).[3][4] In vitro, iclaprim is highly active against MRSA, vancomycin-resistant Staphylococcus aureus (VRSA), strains of Streptococcus pneumoniae resistant to several common antibiotics, and some Gram-negative bacteria.[1]
A new drug application for iclaprim was filed with the U.S. Food and Drug Administration in March 2008,[3] and a marketing authorisation application (MAA) was accepted by the European Medicines Agency on August 21, 2008. Phase II clinical trials are being conducted to assess whether iclaprim can be taken by mouth as well as intravenously and whether it is effective for the treatment of hospital-acquired pneumonia.[4][5]
Iclaprim was granted fast track status by the FDA,[6] but approval of the drug was eventually declined in 2009[7] and no further development has taken place since then.
References
- ↑ 1.0 1.1 Kohlhoff SA, Sharma R (September 2007). "Iclaprim". Expert Opin Investig Drugs. 16 (9): 1441–8. doi:10.1517/13543784.16.9.1441. PMID 17714029.
- ↑ Kohlhoff, SA; Sharma, R (September 2007). "Iclaprim". Expert Opinion on Investigational Drugs. 16 (9): 1441–8. doi:10.1517/13543784.16.9.1441. PMID 17714029.
- ↑ 3.0 3.1 "Arpida Submits New Drug Application for Intravenous Iclaprim for Treatment of Skin Infections" (Press release). Reuters. March 19, 2008. Retrieved 2008-08-24.
- ↑ 4.0 4.1 "Arpida's iclaprim MAA Accepted for Review by EMEA" (Press release). PR Newswire. August 21, 2008. Retrieved 2008-08-24.
- ↑ Peppard, WJ; Schuenke, CD (February 2008). "Iclaprim, a diaminopyrimidine dihydrofolate reductase inhibitor for the potential treatment of antibiotic-resistant staphylococcal infections". Current Opinion on Investigational Drugs. 9 (2): 210–25. PMID 18246524.
- ↑ Morgan A, Cofer C, Stevens DL (March 2009). "Iclaprim: a novel dihydrofolate reductase inhibitor for skin and soft tissue infections". Future Microbiol. 4 (2): 131–44. doi:10.2217/17460913.4.2.131. PMID 19257839.
- ↑ Arpida receives FDA complete response letter for Iclaprim
Further reading
- Schneider P, Hawser S, Islam K (December 2003). "Iclaprim, a novel diaminopyrimidine with potent activity on trimethoprim sensitive and resistant bacteria" (PDF). Bioorg Med Chem Lett. 13 (23): 4217–21. doi:10.1016/j.bmcl.2003.07.023. PMID 14623005.
- Pages with script errors
- CS1 maint: Multiple names: authors list
- Drugs with non-standard legal status
- Articles with changed ChemSpider identifier
- Articles with changed EBI identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Articles with changed InChI identifier
- Chemical articles with unknown parameter in Infobox drug
- Infobox drug articles with non-default infobox title
- Chemical pages without DrugBank identifier
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Pyrimidines
- Phenol ethers
- Aromatic amines
- Drug