Hepatitis B diagnostic criteria
|
Hepatitis B |
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Hepatitis B diagnostic criteria On the Web |
|
American Roentgen Ray Society Images of Hepatitis B diagnostic criteria |
|
Risk calculators and risk factors for Hepatitis B diagnostic criteria |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: João André Alves Silva, M.D. [2]
Overview
The diagnosis of hepatitis is made by biochemical assessment of liver function. A diagnosis is confirmed by demonstration in sera of specific antigens and/or antibodies. Three clinically useful antigen-antibody systems have been identified for hepatitis B, including hepatitis B surface antigen (HBsAg) and antibody to HBsAg (anti-HBs); antibody (anti-HBc IgM and anti-HBc IgG) to hepatitis B core antigen (HBcAg); and hepatitis B e antigen (HBeAg) and antibody to HBeAg (anti-HBe).[1]
Diagnostic Criteria
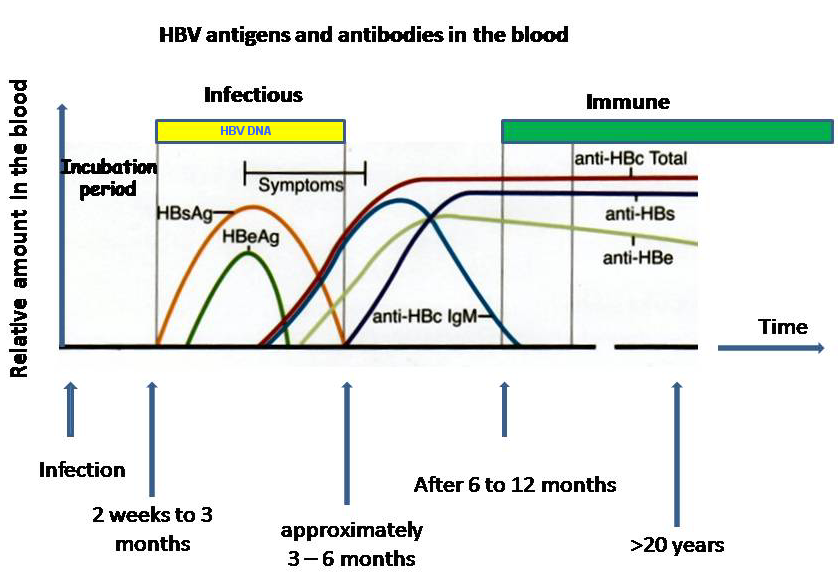
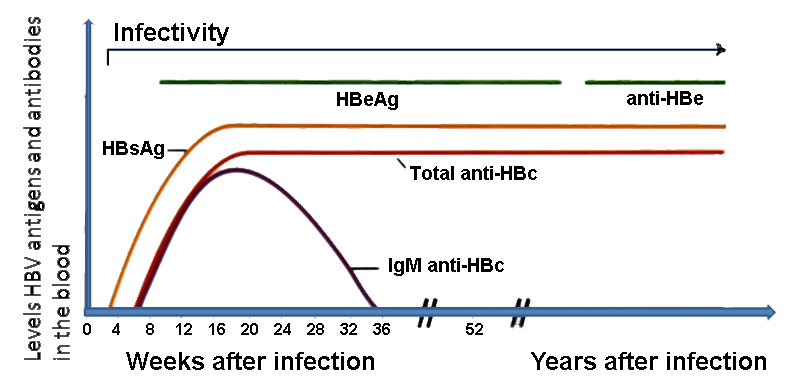
A diagnosis is confirmed by demonstration in sera of specific antigens and/or antibodies. Three clinically useful antigen-antibody systems have been identified for hepatitis B:
- Hepatitis B surface antigen (HBsAg) and antibody to HBsAg (anti-HBs)
- Antibody (anti-HBc IgM and anti-HBc IgG) to hepatitis B core antigen (HBcAg)
- Hepatitis B e antigen (HBeAg) and antibody to HBeAg (anti-HBe)
| Antigens | Description |
|---|---|
| HBsAg | Hepatitis B surface antigen is the earliest indicator of acute infection and is also indicative of chronic infection if its presence persists for more than 6 months. It is useful for the diagnosis of HBV infection and for screening of blood. Its specific antibody is anti-HBs. HBsAg indicates that the person is potentially infectious. |
| HBcAg | Hepatitis B core antigen is derived from the protein envelope that encloses the viral DNA, and it is not detectable in the bloodstream. When HBcAg peptides are expressed on the surface of hepatocytes, they induce an immune response that is crucial for killing infected cells. The HBcAg is a marker of the infectious viral material and it is the most accurate index of viral replication. Its specific antibody is anti-HBc. |
| HBeAg | Hepatitis B e antigen appearing during weeks 3 to 6 indicates an acute active infection at its most infectious period, and means that the patient is infectious. Persistence of this virological marker beyond 10 weeks shows progression to chronic infection and infectiousness. Continuous presence of anti-HBe indicates chronic or chronic active liver disease. HBeAg is not incorporated into virions, but is instead secreted into the serum. Mutant strains of HBV exist that replicate without producing HBeAg. HBeAg’s function is uncertain. Its specific antibody is anti-HBe. |
| HBxAg | Hepatitis B x antigen is detected in HBeAg positive blood in patients with both acute and chronic hepatitis. HBxAg is a transcriptional activator. It does not bind to DNA. Its specific antibody is anti-HBx. |
| HBV DNA | HBV DNA is detectable by PCR as soon as 1 week after initial infection, but the test is generally only performed for research purposes or to detect mutants that escape detection by current methods. |
| HBV DNA Polymerase | Tests for the presence of HBV DNA polymerase, detectable within 1 week of initial infection, are only performed for research purposes. |
| Antibodies | Description |
|---|---|
| Anti-HBs | This is the specific antibody to hepatitis B surface antigen. Its appearance 1 to 4 months after onset of symptoms indicates clinical recovery and subsequent immunity to HBV. Anti-HBs can neutralize HBV and provide protection against HBV infection. |
| Anti-HBc | This is the specific antibody to hepatitis B core antigen. Antibodies to HBc are of class IgM and IgG. They do not neutralize the virus. The presence of IgM identifies an early acute infection. In the absence of HBsAg and anti-HBs, it shows recent infection. IgG with no IgM may be present in chronic and resolved infections. Anti-HBc testing identifies all previously infected persons, including HBV carriers, but does not differentiate between carriers and non-carriers. |
| Anti-HBe | This is the specific antibody to hepatitis B e antigen. During the acute stage of infection the seroconversion from e antigen to e antibody is prognostic for resolution of infection. Its presence in the patient’s blood along with anti-HBc and in the absence of HBsAg and anti-HBs indicates low contagiousness and convalescence. |
| Anti-HBx | This is the specific antibody to hepatitis B x antigen. It appears when other virological markers are becoming undetectable. |
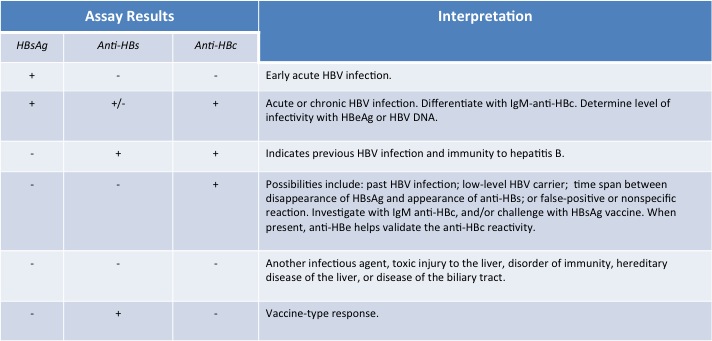
The tests, or assays, for detection of hepatitis B virus infection involve serum or blood tests that detect either viral antigens (proteins produced by the virus) or antibodies produced by the host. Interpretation of these assays is complex.[2]
References
- ↑ World Health Organization, Guidelines for the Prevention, Care, and Treatment of persons with chronic Hepatitis B Infection. (March 2015). http://apps.who.int/iris/bitstream/10665/154590/1/9789241549059_eng.pdf Accessed on October 4th, 2016
- ↑ Bonino F, Chiaberge E, Maran E, Piantino P (1987). "Serological markers of HBV infectivity". Ann. Ist. Super. Sanita. 24 (2): 217–23. PMID 3331068.
- ↑ "WHO".