Ferric oxyhydroxide
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ammu Susheela, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Ferric oxyhydroxide is a phosphate binder that is FDA approved for the prevention of increased serum phosphorus levels in patients with chronic kidney disease on dialysis. Common adverse reactions include diarrhea.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Sucroferric oxyhydroxide is a phosphate binder indicated for the control of serum phosphorus levels in patients with chronic kidney disease on dialysis.
- Ferric oxyhydroxide tablets must be chewed and not swallowed whole. To aid with chewing and swallowing, the tablets may be crushed.
Starting Dose
- The recommended starting dose of ferric oxyhydroxide is 3 tablets (1,500 mg) per day, administered as 1 tablet (500 mg) 3 times daily with meals.
Titration and Maintenance
- Monitor serum phosphorus levels and titrate the dose of ferric oxyhydroxide in decrements or increments of 500 mg (1 tablet) per day as needed until an acceptable serum phosphorus level is reached, with regular monitoring afterwards. Titrate as often as weekly.
- Based on clinical studies, on average patients required 3 to 4 tablets (1,500 mg to 2,000 mg) a day to control serum phosphorus levels.
- The highest daily dose studied in a Phase 3 clinical trial in ESRD patients was 6 tablets (3,000 mg) per day.
Administration
- Ferric oxyhydroxide must be administered with meals. To maximize the dietary phosphate binding, distribute the total daily dose among meals. No additional fluid above the amount usually taken by the patient is required.
- If one or more doses of ferric oxyhydroxide are missed, the medication should be resumed with the next meal. Do not attempt to replace a missed dose.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ferric oxyhydroxide in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Ferric oxyhydroxide in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Ferric oxyhydroxide FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ferric oxyhydroxide in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Ferric oxyhydroxide in pediatric patients.
Contraindications
There is limited information regarding Ferric oxyhydroxide Contraindications in the drug label.
Warnings
Monitoring in Patients with Gastrointestinal Disorders or Iron Accumulation Disorders
- Patients with peritonitis during peritoneal dialysis, significant gastric or hepatic disorders, following major gastrointestinal (GI) surgery, or with a history of hemochromatosis or other diseases with iron accumulation have not been included in clinical studies with ferric oxyhydroxide. Monitor effect and iron homeostasis in such patients.
Adverse Reactions
Clinical Trials Experience
Clinical Trial Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- The safety data derived from ferric oxyhydroxide clinical trials reflect exposure to ferric oxyhydroxide in 2 active-controlled clinical studies involving a total of 778 patients on hemodialysis and 57 patients on peritoneal dialysis exposed for up to 55 weeks. Dosage regimens ranged from 250 mg to 3,000 mg per day.
- As expected with oral preparations containing iron, discolored (dark colored) feces was a commonly occurring adverse drug reaction.
- In a parallel design, dose-finding study of ferric oxyhydroxide with a treatment duration of 6 weeks in hemodialysis patients, adverse reactions for ferric oxyhydroxide (N=128) were similar to those reported for the active-control group (sevelamer hydrochloride) (N=26), with the exception of discolored feces (12%) which did not occur in the active-control group and diarrhea (6%).
- In a 55-week, open-label, active-controlled, parallel design, safety and efficacy study involving 968 hemodialysis patients and 86 peritoneal dialysis patients treated with either ferric oxyhydroxide (N=707 including 57 peritoneal dialysis patients) or the active-control (sevelamer carbonate) (N=348 including 29 peritoneal dialysis patients), adverse reactions occurring in more than 5% in the ferric oxyhydroxide group were diarrhea (24%), discolored feces (16%), and nausea (10%). The majority of diarrhea events in the ferric oxyhydroxide group were mild and transient, occurring soon after initiation of treatment, and resolving with continued treatment. Similar adverse reactions occurred at similar rates in hemodialysis and peritoneal dialysis patients. The most common adverse reactions (>1%) leading to withdrawal were diarrhea (4%), product taste abnormal (2%), and nausea (2%).
Postmarketing Experience
There is limited information regarding Ferric oxyhydroxide Postmarketing Experience in the drug label.
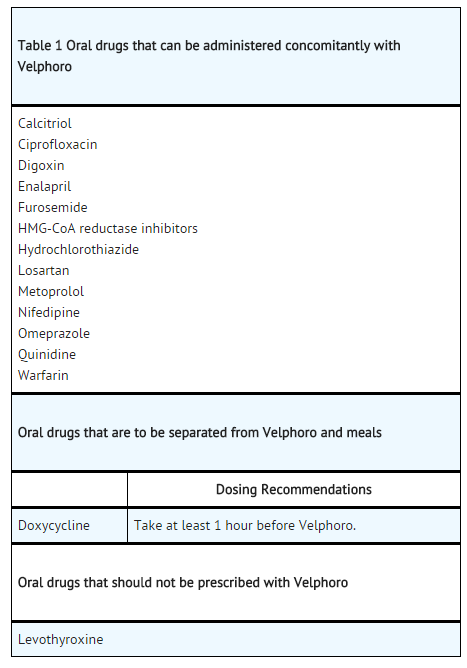
Drug Interactions
- There are no empirical data on avoiding drug interactions between ferric oxyhydroxide and most concomitant oral drugs. For oral medications where a reduction in the bioavailability of that medication would have a clinically significant effect on its safety or efficacy, consider separating the administration of the two drugs.
- The necessary separation depends upon the absorption characteristics of the medication concomitantly administered, such as the time to reach peak systemic levels and whether the drug is an immediate release or an extended release product. Where possible, consider monitoring for clinical response and/or blood levels of concomitant medications that have a narrow therapeutic range.
Use in Specific Populations
Pregnancy
- Pregnancy Category B: Reproduction studies have been performed in rats and rabbits at doses up to 16 and 4 times, respectively, the human maximum recommended clinical dose on a body weight basis, and have not revealed evidence of impaired fertility or harm to the fetus due to ferric oxyhydroxide. However, ferric oxyhydroxide at a dose up to 16 times the maximum clinical dose was associated with an increase in post-implantation loss in pregnant rats. Animal reproduction studies are not always predictive of human response.
- There are no adequate and well-controlled studies in pregnant women.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Ferric oxyhydroxide in women who are pregnant.
Labor and Delivery
- No ferric oxyhydroxide treatment-related effects on labor and delivery were seen in animal studies with doses up to 16 times the maximum recommended clinical dose on a body weight basis. * The effects of ferric oxyhydroxide on labor and delivery in humans are not known.
Nursing Mothers
- Since the absorption of iron from ferric oxyhydroxide is minimal, excretion of ferric oxyhydroxide in breast milk is unlikely.
Pediatric Use
- The safety and efficacy of ferric oxyhydroxide have not been established in pediatric patients.
Geriatic Use
- Of the total number of subjects in two active-controlled clinical studies of ferric oxyhydroxide (N=835), 29.7% (n=248) were 65 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects.
Gender
There is no FDA guidance on the use of Ferric oxyhydroxide with respect to specific gender populations.
Race
There is no FDA guidance on the use of Ferric oxyhydroxide with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Ferric oxyhydroxide in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Ferric oxyhydroxide in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Ferric oxyhydroxide in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Ferric oxyhydroxide in patients who are immunocompromised.
Administration and Monitoring
Administration
Monitoring
- Adjust by 1 tablet per day as needed until an acceptable serum phosphorus level is reached, with regular monitoring afterwards.
- Patients with peritonitis during peritoneal dialysis, significant gastric or hepatic disorders, following major gastrointestinal surgery, or with a history of hemochromatosis or other diseases with iron accumulation have not been included in clinical studies with ferric oxyhydroxide. Monitor effect and iron homeostasis in such patients
IV Compatibility
There is limited information regarding the compatibility of Ferric oxyhydroxide and IV administrations.
Overdosage
There are no reports of overdosage with ferric oxyhydroxide in patients. Since the absorption of iron from ferric oxyhydroxide is low, the risk of systemic iron toxicity is low.
- Hypophosphatemia should be treated by standard clinical practice.
- Ferric oxyhydroxide has been studied in doses up to 3,000 mg per day.
Pharmacology
| Template:Chembox header2 | Ferric oxyhydroxide | |
|---|---|
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | Lua error in Module:Wikidata at line 879: attempt to index field 'wikibase' (a nil value). Lua error in Module:Wikidata at line 879: attempt to index field 'wikibase' (a nil value). |
| MeSH | Goethite |
PubChem CID
|
|
| |
| |
| Properties | |
| Hazards | |
| Template:Chembox header2 | Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox disclaimer and references | |
Mechanism of Action
- In the aqueous environment of the GI tract, phosphate binding takes place by ligand exchange between hydroxyl groups and/or water in sucroferric oxyhydroxide and the phosphate in the diet. The bound phosphate is eliminated with feces.
- Both serum phosphorus levels and calcium-phosphorus product levels are reduced as a consequence of the reduced dietary phosphate absorption.
Structure
- Ferric oxyhydroxide chewable tablets are brown, circular, bi-planar, and are embossed with “PA 500” on 1 side. Each tablet of ferric oxyhydroxide contains 500 mg iron (in 2,500 mg sucroferric oxyhydroxide). The ferric oxyhydroxide drug substance is a mixture of polynuclear iron(III)-oxyhydroxide, sucrose, and starches. The active moiety, polynuclear iron(III)-oxyhydroxide, is practically insoluble and cannot be absorbed. The inactive ingredients are woodberry flavor, neohesperidin dihydrochalcone, magnesium stearate, and silica (colloidal, anhydrous).
Pharmacodynamics
- In vitro studies have demonstrated a robust phosphate binding capacity of ferric oxyhydroxide over the physiologically relevant pH range of the GI tract (1.2-7.5). The phosphate binding capacity of ferric oxyhydroxide peaked at pH 2.5, resulting in 96% of the available phosphate being adsorbed (phosphorus:iron concentration ratio 0.4:1).
Pharmacokinetics
- The active moiety of ferric oxyhydroxide, polynuclear iron(III)-oxyhydroxide (pn-FeOOH), is practically insoluble and therefore not absorbed and not metabolized. Its degradation product, mononuclear iron species, can however be released from the surface of pn-FeOOH and be absorbed.
- Because of the insolubility and degradation characteristics of ferric oxyhydroxide, no classical pharmacokinetic studies can be carried out.
- The sucrose and starch components of ferric oxyhydroxide can be digested to glucose and fructose, and maltose and glucose, respectively. These compounds can be absorbed in the blood. * One tablet is equivalent to 1.4 g of carbohydrates.
- The iron uptake from radiolabelled ferric oxyhydroxide drug substance, 2,000 mg in 1 day, was investigated in 16 chronic kidney disease patients (8 pre-dialysis and 8 hemodialysis patients) and 8 healthy volunteers with low iron stores (serum ferritin <100 mcg/L). In healthy subjects, the median uptake of radiolabelled iron in the blood was 0.43% on Day 21. In chronic kidney disease patients, the median uptake was much less, 0.04% on Day 21.
Drug Interaction Studies
In vitro
- In vitro interactions were studied in aqueous solutions which mimic the physico-chemical conditions of the gastro-intestinal tract with or without the presence of phosphate (400 mg). The study was conducted at pH 3.0, 5.5 and 8.0 with incubation at 37°C for 6 hours.
- Interaction with ferric oxyhydroxide was seen with the following drugs: alendronate, doxycycline, levothyroxine, and paricalcitol.
- The following drugs did not show interaction with ferric oxyhydroxide: ciprofloxacin, enalapril, hydrochlorothiazide, metformin, metoprolol, nifedipine, and quinidine.
In vivo
- Five in vivo drug interaction studies (N=40/study) were conducted with losartan, furosemide, digoxin, omeprazole and warfarin in healthy subjects receiving 1,000 mg ferric oxyhydroxide 3 times a day with meals. Ferric oxyhydroxide did not alter the systemic exposure as measured by the area under the curve (AUC) of the tested drugs when co-administered with ferric oxyhydroxide or given 2 hours later.
- Data from the clinical studies (Study-05A and Study-05B) show that ferric oxyhydroxide does not affect the lipid lowering effects of HMG-CoA reductase inhibitors or the PTH lowering effect of calcitriol.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Carcinogenicity studies were performed in mice and rats.
- In the 2-year carcinogenicity study in mice, animals were given ferric oxyhydroxide by diet at doses of 250, 500 or 1,000 mg/kg/day. Rare but not statistically significant neoplastic adenocarcinomas were seen in the colon of male mice at doses of 500 and 1,000 mg/kg/day. For a 60 kg person, the no-observed-adverse-effect level (NOAEL) of 250 mg/kg/day represents 5 times (on a body weight basis) the maximum recommended clinical dose of 3,000 mg/day. In addition, an increased incidence of epithelial hyperplasia was seen in the colon at all dosage levels (i.e., ≥5 times the maximum recommended clinical dose) and in the cecum at the highest dosage (equivalent to 20 times the maximum recommended clinical dose). The development of adenocarcinoma in the male mice was considered not a genotoxic effect, but the result of chronic local irritation from high amounts of intraluminal ferric oxyhydroxide in the GI tract.
- In the 2-year rat carcinogenicity study, animals were given ferric oxyhydroxide by diet at doses of 40, 150 or 500 mg/kg/day. No statistically significantly increased incidences of tumors were found, but there were increased incidences in epithelial hyperplasia with or without submucosal inflammation in duodenum, cecum and colon at the dose of 500 mg/kg/day (10 times the maximum recommended clinical dose).
- Fferric oxyhydroxide was not mutagenic, clastogenic or DNA damaging in vitro in the Ames bacterial reverse mutation test, or in the Chinese-hamster fibroblast chromosomal aberration test, or in vivo in the rat Comet assay or peripheral blood micronucleus test.
- In rats, mating performance and fertility were unaffected by ferric oxyhydroxide at oral doses up to 800 mg/kg/day (16 times the maximum recommended clinical dose).
Animal Toxicity and/or Pharmacology
- In pregnant rats given up to 800 mg/kg/day ferric oxyhydroxide by oral gavage from Days 6 to 17 post-mating, no embryo-fetal development toxicity was observed. This dose corresponds to 16 times the maximum recommended clinical dose.
- In pregnant rabbits given 50, 100 or 200 mg/kg/day ferric oxyhydroxide by oral gavage, from Days 6 to 19 post-mating, the number of fetuses with incomplete/unossified epiphyses and metacarpals/phalanges was increased at the highest dose (corresponding to 4 times the recommended maximum clinical dose). Litter parameters were not adversely affected.
- In pregnant rats given ferric oxyhydroxide at 100, 280, or 800 mg/kg/day by oral gavage from Day 6 post-mating to lactation Day 20, offspring body weight gain was lower at age 5-13 weeks and neuromuscular function was delayed at the dose of 800 mg/kg/day. This dose represented 16 times the maximum recommended clinical dose.
Clinical Studies
- The ability of ferric oxyhydroxide to lower serum phosphorus in ESRD patients on dialysis was demonstrated in 2 randomized clinical trials: one 6-week, open-label, active-controlled (sevelamer hydrochloride), dose-finding study; and one 55-week, open-label, active-controlled (sevelamer carbonate), parallel-group, safety and efficacy study.
- In clinical trials, control of serum phosphorus levels was demonstrated at doses starting from 1,000 mg (2 tablets) per day with treatment effect being observed as early as 1-2 weeks after starting ferric oxyhydroxide.
Fixed-dose Study
- In Study-03A, 154 ESRD patients on hemodialysis who were hyperphosphatemic (serum phosphorus >5.5 mg/dL but <7.75 mg/dL) following a 2-week phosphate binder washout period, were randomized to receive ferric oxyhydroxide at 250 mg/day, 1,000 mg/day, 1,500 mg/day, 2,000 mg/day, or 2,500 mg/day or active-control (sevelamer hydrochloride). Ferric oxyhydroxide treatment was divided across meals, depending on dose. No dose titration was allowed. Within each of the groups, the serum phosphorus level at the end of treatment was compared to baseline value. Ferric oxyhydroxide was shown to be efficacious (p≤0.016) for all doses except 250 mg/day. There were no patient-reported dose limiting treatment-emergent adverse events.
- Mean changes in iron parameters (ferritin, transferrin saturation (TSAT) and transferrin) and vitamins (A, D, E and K) were generally not clinically meaningful and showed no apparent trends across the treatment groups. Ferric oxyhydroxide had a similar GI adverse event profile to sevelamer hydrochloride, and no dose-dependent trend in GI events was observed.
Dose Titration Study
- In Study-05A, 1,054 patients on hemodialysis (N=968) or peritoneal dialysis (N=87) with serum phosphorus ≥6 mg/dL following a 2-4 week phosphate binder washout period, were randomized and treated with either ferric oxyhydroxide, at a starting dose of 1,000 mg/day (N=707), or active-control (sevelamer carbonate, N=348) for 24 weeks. At the end of Week 24, 93 patients on hemodialysis whose serum phosphorus levels were controlled (<5.5 mg/dL) with ferric oxyhydroxide in the first part of the study, were re-randomized to continue treatment with either their Week 24 maintenance dose (N=44 or a non-effective low dose control 250 mg/day, N=49) of ferric oxyhydroxide for a further 3 weeks. At Week 27, a superiority analysis of the ferric oxyhydroxide maintenance dose versus low dose was performed. The maximum dose of ferric oxyhydroxide was 3,000 mg/day (6 tablets/day) and the minimum dose was 1,000 mg/day (2 tablets/day). Ferric oxyhydroxide was administered with food and the daily dose was divided across the largest meals of the day.
- The ferric oxyhydroxide maintenance dose (1,000 to 3,000 mg/day) was statistically significantly superior in sustaining the phosphorus lowering effect in hemodialysis patients at Week 27 (p<0.001) compared with the non-effective low dose control.

This image is provided by the National Library of Medicine. 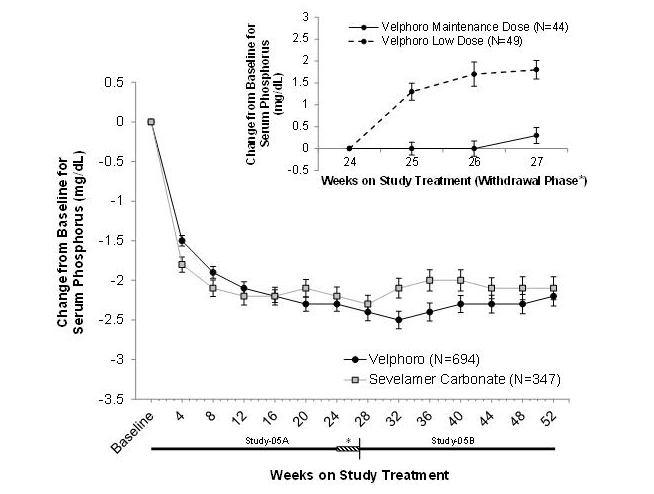
This image is provided by the National Library of Medicine. 
This image is provided by the National Library of Medicine.
How Supplied
- Ferric oxyhydroxide are chewable tablets supplied as brown, circular, bi-planar tablets, embossed with "PA 500" on 1 side. Each tablet of ferric oxyhydroxide contains 500 mg iron as sucroferric oxyhydroxide. ferric oxyhydroxide tablets are packaged as follows:
- NDC 49230-645-51 Bottle of 90 chewable tablets
Storage
- Store in the original package and keep the bottle tightly closed in order to protect from moisture.
- Store at 25°C (77°F) with excursions permitted to 15 to 30°C (59 to 86°F).
Images
Drug Images
{{#ask: Page Name::Ferric oxyhydroxide |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Ferric oxyhydroxide |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Inform patients that ferric oxyhydroxide tablets must be chewed and not swallowed whole. To aid with chewing and swallowing, the tablets may be crushed.
- ferric oxyhydroxide should be taken with meals.
- Instruct patients on concomitant medications that should be dosed apart from ferric oxyhydroxide.
- Inform patients that ferric oxyhydroxide can cause discolored (black) stool.
Precautions with Alcohol
- Alcohol-Ferric oxyhydroxide interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- VELPHORO®[2]
{{#subobject:
|Label Page=Ferric oxyhydroxide |Label Name=Valpa 04.jpg
}}
{{#subobject:
|Label Page=Ferric oxyhydroxide |Label Name=Valpa 05.jpg
}}
{{#subobject:
|Label Page=Ferric oxyhydroxide |Label Name=DailyMed - VELPHORO- ferric oxyhydroxide tablet, chewable .png
}}
Look-Alike Drug Names
There is limited information regarding Ferric oxyhydroxide Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- Pages with script errors
- Chemical articles with multiple compound IDs
- Multiple chemicals in an infobox that need indexing
- Chemical articles with multiple CAS registry numbers
- Articles without InChI source
- Articles without EBI source
- Articles without KEGG source
- Articles without UNII source
- Chemical articles with unknown parameter in Chembox
- ECHA InfoCard ID from Wikidata
- Oxides
- Hydroxides