Drug eluting stent
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
In cardiology, a drug-eluting stent is a stent (a scaffold) placed into narrowed, diseased coronary arteries that slowly releases a drug to block cell proliferation. This prevents scar-tissue–like growth that, together with clots (thrombus), could otherwise block the stented artery.
Heart attacks, or myocardial infarctions, are major causes of death and disability; they result when a portion of heart muscle dies from inadequate blood flow. This typically occurs at sites where coronary arteries are already narrowed and damaged. Fat and cholesterol deposition as well as inflammation in the arterial wall cause injury; excessive tissue growth and additional cholesterol deposition occur. Clots can then occur when these plaques rupture releasing their noxious contents in to the passing blood flow. These noxious chemicals from within the plaques stimulate blood clotting elements to initiate thrombus or clot, formation. These narrowings are prone to being suddenly blocked, or a piece may break off and block a smaller branch downstream (embolism). If blood flow can be restored early enough, permanent damage can be prevented, and pre-emptive manipulation can prevent heart attacks from occurring in the first place.
Coronary artery stents, typically a metal framework, can be placed inside the artery to help keep it open. However, as the stent is a foreign object (not native to the body), it incites an immune response. This may cause scar tissue (cell proliferation) to rapidly grow over the stent. In addition, there is a strong tendency for clots to form at the site where the stent damages the arterial wall. Since platelets are involved in the clotting process, patients must take antiplatelet therapy afterwards, usually clopidogrel for six months and aspirin indefinitely.[1] However, the antiplatelet therapy may be insufficient to fully prevent clots; these and the cell proliferation may cause the standard (“bare-metal”) stents to become blocked. Drug-eluting stents were designed to lessen this problem; by releasing an antiproliferative drug (drugs typically used against cancer or as immunosuppressants), they can help avoid this in-stent restenosis (re-narrowing).
Drug-eluting stents have been shown to be superior to traditional stents ("bare-metal stents") in reducing short-term complications. Their long term effectiveness compared to traditional stents or coronary bypass grafting is under scrutiny by the FDA.[2]
Structure
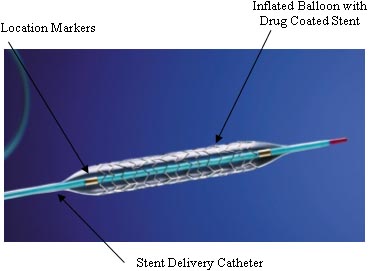
Drug-eluting stents consist of three parts. The stent itself is an expandable framework, usually metal. Added to this is a drug to prevent the artery from being re-occluded, or blocked. These typically have been drugs already in use as anti-cancer drugs or drugs that suppress the immune system, although new drugs are being developed specifically for drug-eluting stents. Finally, there must be a carrier which slowly releases the drug over months. The carrier is typically a polymer, although phosphorylcholine or ceramics are also being researched.[3] Different carriers release the loaded drug at different rates.
Placement
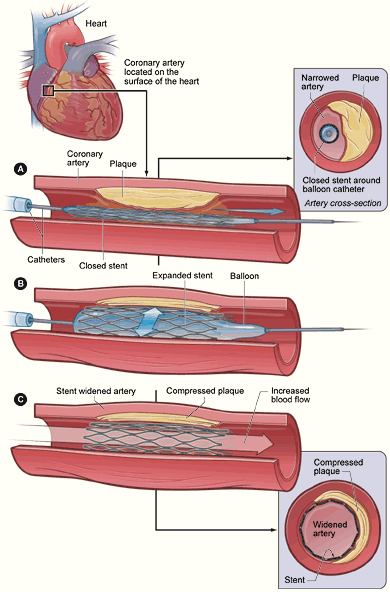
Treating a blocked ("stenosed") coronary artery with a drug eluting stent follows the same steps as other angioplasty procedures with a few important differences. The interventional cardiologist uses angiography to assess the location and estimate the size of the blockage ("lesion") by injecting a contrast medium through the guide catheter and viewing the flow of blood through the downstream coronary arteries. Intravascular ultrasound (IVUS) may be used to assess the lesion's thickness and hardness ("calcification"). The cardiologist uses this information to decide whether to treat the lesion with a stent, and if so, what kind and size. Drug eluting stents are most often sold as a unit, with the stent in its collapsed form attached onto the outside of a balloon catheter. Outside the US, physicians may perform "direct stenting" where the stent is threaded through the lesion and expanded. Common practice in the US is to predilate the blockage before delivering the stent. Predilation is accomplished by threading the lesion with an ordinary balloon catheter and expanding it to the vessel's original diameter. The physician withdraws this catheter and threads the stent on its balloon catheter through the lesion. The physician expands the balloon which deforms the metal stent to its expanded size. The cardiologist may "customize" the fit of the stent to match the blood vessel's shape, using IVUS to guide the work.[4] It is critically important that the framework of the stent be in direct contact with the walls of the vessel to minimize potential complications such as blood clot formation. Very long lesions may require more than one stent -- this result of this treatment is sometimes referred to as a "full metal jacket".[5]
The procedure itself is performed in a catheterization clinic ("cath lab"). Barring complications, patients undergoing catheterizations are kept at least overnight for observation.[6]
Current devices
In the US market, FDA has approved two drug-eluting stents as of December 2007: Cypher from Cordis Corporation (a Johnson & Johnson Company, approved April 2003)[7] and Taxus from Boston Scientific (approved March 2004).[8] Both products use previously approved bare metal stents (BxVelocity and Express2, respectively) bearing polymeric coatings. These coatings elute drugs with antiproliferative properties that FDA had approved earlier for other indications. Both products are available in a range of diameters and lengths that the interventionalist selects to treat the target lesion.
Cypher releases sirolimus (rapamycin), a powerful immunosuppressive and antiproliferative drug that was first approved by FDA to prevent organ transplant rejection; Cordis licenses this drug from Wyeth and its polymeric coating from Surmodics. Produced by the bacterium Streptomyces hygroscopicus, rapamycin binds to the immunophilin FKBP-12. The resulting complex inhibits the mammalian target of rapamycin (mTOR), which has several effects, including preventing the cell from duplicating its genetic material and thereby blocking the cell cycle at the G1→S transition.[9] .Rapamycin inhibits both rat and human vascular smooth muscle proliferation and migration in vitro. A study investigated (1) whether rapamycin administration could reduce neointimal thickening in a porcine model of restenosis post-PTCA and (2) the mechanism by which rapamycin inhibits VSMCs in vivo. It was found that rapamycin administration significantly reduced the arterial proliferative response after PTCA in the pig by increasing the level of the CDKI p27(kip1) and inhibition of the pRb phosphorylation within the vessel wall. Therefore, pharmacological interventions that elevate CDKI in the vessel wall and target cyclin-dependent kinase activity may have a therapeutic role in the treatment of restenosis after angioplasty in humans[10].
Taxus delivers paclitaxel, an antiproliferative drug first approved to treat various forms of cancer; Boston Scientific licenses the drug and coating from Angiotech Pharmaceuticals. Paclitaxel binds to and stabilizes microtubules. Without the dynamic framework provided by these components of the cytoskeleton, the cell cannot undergo mitosis and so is arrested at the M stage.[11]
FDA is the final stages of reviewing applications for two other drug eluting stents: Abbott Laboratories' XIENCE V[12] and Medtronic's Endeavor.[13] Final approval for both is expected in 2008.
All four of these drug eluting stents (and many others) are available in markets outside the US, where the regulatory barrier to entry is generally lower.[14]
History
The first procedural method to treat blocked coronary arteries was a type of open-heart surgery called coronary artery bypass graft (CABG) surgery, which uses a section of vein or artery from elsewhere in the body to bypass the diseased vessel. In 1977, Andreas Grüntzig introduced percutaneous transluminal coronary angioplasty (PTCA), also called balloon angioplasty, in which a catheter was introduced through a peripheral artery and a balloon expanded to compress and crack the obstructive plaque.[15]
As equipment and techniques improved, the use of PTCA rapidly increased, and by the mid-1980s, PTCA and CABG were being performed at equivalent rates.[16] PTCA could only be used on limited scenarios, and the vessels had a high rate (30–40% in six months) of restenosis; additionally, 3% required emergency bypass surgery.[16] Dotter and Judkins had suggested using prosthetic devices inside arteries to maintain blood flow (in arteries of the leg) in 1964,[17] and in 1986, Puel and Sigwart implanted the first stent in humans.[3] Several trials in the 1990s showed the superiority of stent placement to simple balloon angioplasty, and stent placement became increasingly prevalent, reaching 84% of percutaneous interventions (those done via needle-puncture rather than incision) by 1999.[3]
Initial difficulties included blood clotting and occluding the stent in the hours or days after placement.[16] Coating the stent with biologically inert substances like platinum or gold did not help.[3] Eventually, using high balloon pressures to tightly fix the stent against the vessel and administering aspirin and (usually) clopidogrel as anticoagulants were established; these changes eliminated most of the difficulty with in-stent thrombosis.[3][16]
Difficulties still remained, however, with the formation of scar tissue inside the stent (in-stent neointimal hyperplasia) and clotting problems not addressed by the antiplatelet drug regimen. The stent itself was a logical choice for delivering medication. The slow release of drugs from the stent spares the patient the inconvenience of taking yet another medication, and prevents the danger of the patient forgetting to take or losing interest in taking the medicine. But more importantly, a stent that releases a drug can deliver high concentrations directly to the target region, analogous to placing a medicated cream on a skin problem or taking an inhaler to help the lungs or airways. Taking the medication orally or intravenously would require much higher doses to ensure a sufficient concentration at the target; this could cause unacceptable side effects or patient injury.
The first successful trials were of sirolimus-eluting stents. A successful trial in 2002 led to approval of the Cypher stent in Europe, followed by FDA approval in the U.S. in 2003.[3] Soon thereafter, a series of trials of paclitaxel-eluting stents led to FDA approval of the Taxus stent in 2004.[8]
Drug eluting stents versus bare metal stents
DES significantly reduces restenosis and target lesion revascularization (TLR) but there has been no difference in incidence of death or myocardial infarction.[18]
| Death % | Myocardial infarction % | TLR % | MACE % | Angiographic restenosis % | Edge restenosis % | Stent thrombosis % | Late incompetent stent apposition % | |
| DES | 0.9 | 2.7 | 4.2 | 7.8 | 8.9 | 3.0 | 0.7 | 8.5 |
| BMS | 0.9 | 2.9 | 13.2 | 16.4 | 29.3 | 1.9 | 0.5 | 5.1 |
Uses
There has been considerable research showing the benefits of coronary stents. Data specifically on drug-eluting stents are less abundant, though where studied, they have usually been shown to be superior to bare-metal stents, and in some cases, may be used for lesions for which surgery was previously the only option. Drug-eluting stents are used both for restoring blood flow immediately after a heart attack and also electively for improving blood flow in a compromised vessel. Only certain types of blockages are amenable to stent placement, though drug-eluting stents may be successful in lesions for which bare-metal stents were insufficient. Drug-eluting stents are used to reopen grafts from prior CABG surgery that have themselves become blocked, and also can be used in cases of in-stent restenosis in prior stents.[3]
Current research
Research focuses on establishing the roles for drug-eluting stents and for developing new types of stents. Different materials for all three components—the scaffolding (framework), the carrier, and the drug—are being actively investigated.
Alternative frameworks
In place of the stainless steel currently used in stents, various biodegradable frameworks are under early phases of investigation. Since metal, as a foreign substance, provokes inflammation, scarring, and thrombosis (clotting), it is hoped that biodegradable or bioabsorbable stents may prevent some of these effects. A magnesium alloy–based stent has been tested in animals, though there is currently no carrier for drug elution.[19] A promising biodegradable framework is made from poly-L-lactide, a polymer of a derivative of L-lactic acid. One of these stents, the Igaki-Tamai stent, has been studied in pigs; tranilast[20] and paclitaxel[21] have been used as eluted drugs.
Alternative drugs
There are also several other anti-proliferative drugs under investigation in human clinical trials. In general, these are analogues of sirolimus. Like sirolimus, these block the action of mTOR. Abbott has developed zotarolimus; unlike sirolimus and paclitaxel, this sirolimus analogue designed for use in stents with phosphorylcholine as a carrier. Their ZoMaxx stent is a zotarolimus-eluting, stainless steel and tantalum–based stent; a modified phosphorylcholine slowly releases the zotarolimus.[22] Zotarolimus has been licensed to Medtronic which is researching the effectiveness in a drug-eluting stent of their own. Their Endeavor stent, which is a cobalt alloy,[3] also uses phosphorylcholine to carry the zotarolimus was approved for use in Europe in 2005 is now close to U.S. FDA approval[23]
Clinical trials are currently examining two stents carrying everolimus,[3] an immunosuppressant that like sirolimus is used to prevent organ rejection.[9] Guidant, which has the exclusive license to use everolimus in drug-eluting stents, is the manufacturer of both stents. This Guidant business has subsequently been sold to Abbott Labs.[24] The Champion stent uses a bioabsorbable polylactic acid carrier on a stainless steel stent.[25][26] In contrast, its Xience stent uses a durable (non-bioabsorbable) polymer on a cobalt stent.[27]
Roles of Coronary Bypass Surgery versus Percutaneous Coronary Intervention
The rapid developments in both surgical and percutaneous techniques have been such that the choice of the optimum revascularisation strategy is changing, often without an established evidence base; this is particularly true in complex conditions including patients with three-vessel and left main stem anatomy. The widespread use of drug eluting stents has resulted in a significant reduction in patients referred for CABG. The SYNTAX Trial [2]aims to explore the interface between treatment with coronary bypass grafting and PCI in patients with three-vessel and left main stem disease, comparing coronary bypass grafting using contemporary techniques and PCI using drug eluting TAXUS stents.
Complications
In the last several years, drug-eluting stent use has become increasingly popular, both in place of surgery and for lesions not severe enough for surgery. Placing stents is not without risk, however, and the recent development of the drug-eluting stents means that long-term data, especially in comparison to traditional bare-metal stents, are not available.
Risks due to cardiac catheterization
As with all cardiac catheterization, there are several risks. Patients may exhibit severe allergic response to the contrast agents used to visualize the coronary arteries, and occasionally, the peripheral entry artery fails to properly heal after the catheter is removed, causing a collection of blood called a hematoma.
Coronary artery perforation
Rarely, a coronary artery can be perforated while the catheter is advanced or during stent placement.[16]
Stent thrombosis
Finally, stent occlusion can occur. Thrombosis may occur during the procedure, in the following days, or much later. Stents cause damage to the vessel wall, and, as foreign objects, they provoke inflammation and clot formation. And tissue proliferation in the stent can cause the vessel to narrow again.
Patients with stents (but not those undergoing isolated balloon angioplasty) must remain on an antiplatelet drug like clopidogrel for at least three to six months; discontinuing it, even for a short time, can cause a clot to form;[16] aspirin must be taken for life.[3] The role of dual antiplatelet therapy (aspirin plus a second agent) beyond six months is not clear.[28]
Drug-eluting stents have been shown to have significantly lower rates of in-stent proliferation compared to bare-metal stents. However, some studies suggest that the proliferation may be merely delayed; when the drug has been completely eluted, proliferation may occur.[3] Both sirolimus and paclitaxel-eluting stents are associated with a small but statistically higher risk of thrombosis after the first year, compared to bare metal stents. Although this risk is still small, fatality results in one-third of patients who develop late thrombosis.[29] This risk is offset by drug-eluting stents' markedly reduced risk of restenosis and its complications including myocardial infarction. A meta-analysis concluded "the risks of mortality associated with drug-eluting and bare-metal stents are similar." The advantage of drug-eluting stents is in reduction of restenosis.
Whether sirolimus or paclitaxel-eluting stents are measurably different in their outcomes is a topic of great interest, including to the marketing departments of the manufacturers themselves. Analyses favoring one or the other stent have been advanced. The differences, if any, between the two devices are small. [30]
Allergic reaction
Rarely, a type of allergic reaction to the drug may occur; episodes of fatality have been reported.[31]
Controversy
As enthusiasm for the new devices abates, there is some concern about overzealous use of stents in general. Two studies found that about half of patients received stents for unapproved reasons.[32][33] Both studies found worse outcomes for these patients.
References
- ↑ Michel, Thomas (2006) [1941]. "Treatment of Myocardial Ischemia". In Laurence L. Brunton, John S. Lazo, & Keith L. Parker. Goodman & Gilman's The Pharmacological Basis of Therapeutics (11th ed. ed.). New York: McGraw-Hill. p. 842.
- ↑ FDA Statement on Coronary Drug-Eluting Stents (September 14, 2006)
- ↑ 3.00 3.01 3.02 3.03 3.04 3.05 3.06 3.07 3.08 3.09 3.10 Serruys PW, Kutryk MJ, Ong AT. "Coronary-Artery Stents". N Engl J Med. 354 (5): 483–95. PMID 16452560.
- ↑ http://www.ptca.org/imaging/ivus.html
- ↑ http://www.medscape.com/viewarticle/517637
- ↑ http://www.angioplasty.org/nv/angio101.html
- ↑ "New Device Approval — Cypher Sirolimus-eluting Coronary Stent". Food and Drug Administration. Retrieved 2006-07-22.
- ↑ 8.0 8.1 "New Device Approval — P030025 — TAXUS™ Express2™ Paclitaxel-Eluting Coronary Stent System". Food and Drug Administration. 2004-09-09. Retrieved 2006-07-22.
- ↑ 9.0 9.1 Krensky, Alan M. (2006) [1941]. "Immunosuppressants, Tolerogens, and Immunostimulants". In Laurence L. Brunton, John S. Lazo, & Keith L. Parker. Goodman & Gilman's The Pharmacological Basis of Therapeutics (11th ed. ed.). New York: McGraw-Hill. p. 1413. Unknown parameter
|coauthors=ignored (help) - ↑ Gallo R, Padurean A, Jayaraman T, Marx S, Roque M, Adelman S; et al. (1999). "Inhibition of intimal thickening after balloon angioplasty in porcine coronary arteries by targeting regulators of the cell cycle". Circulation. 99 (16): 2164–70. PMID 10217658.
- ↑ Chabner, Bruce A. (2006) [1941]. "Antineoplastic Agents". In Laurence L. Brunton, John S. Lazo, & Keith L. Parker. Goodman & Gilman's The Pharmacological Basis of Therapeutics (11th ed. ed.). New York: McGraw-Hill. pp. 1352–1353. Unknown parameter
|coauthors=ignored (help) - ↑ http://online.wsj.com/article/SB119637142021108412.html
- ↑ http://online.wsj.com/article/SB119205142965455086.html
- ↑ http://www.ptca.org/des.html
- ↑ Grüntzig, AR (1979-07-12). "Nonoperative dilatation of coronary-artery stenosis: percutaneous transluminal coronary angioplasty". New England Journal of Medicine. 301 (2): 61–68. PMID 449946. Retrieved 2006-07-22. Unknown parameter
|coauthors=ignored (help); Check date values in:|date=(help) (abstract) - ↑ 16.0 16.1 16.2 16.3 16.4 16.5 Baim, Donald S. (2005) [1958]. "Percutaneous Coronary Revascularization". In Dennis L. Kasper, Anthony S. Fauci, Dan L. Longo, Eugene Braunwald, Stephen L. Hauser, & J. Larry Jameson. Harrison's Principles of Internal Medicine (16th ed. ed.). New York: McGraw-Hill. pp. 1459–1462.
- ↑ Dotter, Charles T. (1964). "Transluminal Treatment of Arteriosclerotic Obstruction". Circulation. 30: 654–670. PMID 14226164. Retrieved 2006-07-22. Unknown parameter
|coauthors=ignored (help) (abstract) - ↑ 18.0 18.1 Babapulle MN, Joseph L, Bélisle P, Brophy JM, Eisenberg MJ (2004). "A hierarchical Bayesian meta-analysis of randomised clinical trials of drug-eluting stents". Lancet. 364 (9434): 583–91. doi:10.1016/S0140-6736(04)16850-5. PMID 15313358.
- ↑ Heublein, B. (2003). "Biocorrosion of magnesium alloys: a new principle in cardiovascular implant technology?". Heart. 89: 651–656. PMID 12748224. Retrieved 2006-07-23. Unknown parameter
|coauthors=ignored (help) - ↑ Tsuji, T. (2003). "Biodegradable stents as a platform to drug loading". International Journal of Cardiovascular Interventions. 5 (1): 13–6. PMID 12623560. Unknown parameter
|coauthors=ignored (help);|access-date=requires|url=(help) - ↑ Vogt, Felix (2004). "Long-term assessment of a novel biodegradable paclitaxel-eluting coronary polylactide stent". European Heart Journal. 25: 1330–1340. PMID 15288161. Retrieved 2006-07-22. Unknown parameter
|coauthors=ignored (help) - ↑ "Vascular Devices". Abbott. Retrieved 2006-07-23.
- ↑ "FDA advisers OK Medtronic stent". Star Tribune. Retrieved 2007-10-10.
- ↑ "Abbott Completes Acquisition of Guidant Vascular Business". Retrieved 2007-01-12.
- ↑ Grube, Eberhard (2004). "Six- and Twelve-Month Results From First Human Experience Using Everolimus-Eluting Stents With Bioabsorbable Polymer". Circulation. 109: 2168–2171. doi:10.1161/01.CIR.0000128850.84227.FD. PMID 15123533. Unknown parameter
|coauthors=ignored (help) - ↑ "Guidant News Release — April 5, 2004". Guidant. 2004-04-05. Retrieved 2007-07-23.
- ↑ "Guidant News Release — June 22, 2005". Guidant. 2005-06-22. Retrieved 2006-07-23.
- ↑ Gargiulo G, Windecker S, da Costa BR, Feres F, Hong MK, Gilard M; et al. (2016). "Short term versus long term dual antiplatelet therapy after implantation of drug eluting stent in patients with or without diabetes: systematic review and meta-analysis of individual participant data from randomised trials". BMJ. 355: i5483. doi:10.1136/bmj.i5483. PMC 5094199. PMID 27811064.
- ↑ Bavry AA, Kumbhani DJ, Helton TJ, Borek PP, Mood GR, Bhatt DL (2006). "Late thrombosis of drug-eluting stents: a meta-analysis of randomized clinical trials". Am. J. Med. 119 (12): 1056–61. doi:10.1016/j.amjmed.2006.01.023. PMID 17145250.
- ↑ Stettler C, Wandel S, Allemann S; et al. (2007). "Outcomes associated with drug-eluting and bare-metal stents: a collaborative network meta-analysis". Lancet. 370 (9591): 937–48. doi:10.1016/S0140-6736(07)61444-5. PMID 17869634.
- ↑ Virmani, Renu (2004). "Localized Hypersensitivity and Late Coronary Thrombosis Secondary to a Sirolimus-Eluting Stent". Circulation. 109: 701–706. doi:10.1161/01.CIR.0000116202.41966.D4. PMID 14744976. Unknown parameter
|coauthors=ignored (help) - ↑ Win HK, Caldera AE, Maresh K; et al. (2007). "Clinical outcomes and stent thrombosis following off-label use of drug-eluting stents". JAMA. 297 (18): 2001–9. doi:10.1001/jama.297.18.2001. PMID 17488965.
- ↑ Beohar N, Davidson CJ, Kip KE; et al. (2007). "Outcomes and complications associated with off-label and untested use of drug-eluting stents". JAMA. 297 (18): 1992–2000. doi:10.1001/jama.297.18.1992. PMID 17488964.
Further reading
- Fischetti, Mark (2006). "Vascular Stents: Expanding Use". Scientific American: 94. Unknown parameter
|month=ignored (help) (layperson overview, subscription required) - Serruys, Patrick W. (2006-02-02). "Coronary-Artery Stents". New England Journal of Medicine. 354 (5): 483–495. Unknown parameter
|coauthors=ignored (help) (journal review article, subscription required)
External links
| Wikimedia Commons has media related to Stent. |
- CS1 maint: Multiple names: editors list
- CS1 maint: Extra text
- CS1 maint: Uses authors parameter
- Pages with citations using unsupported parameters
- CS1 maint: Explicit use of et al.
- CS1 maint: Multiple names: authors list
- CS1 errors: dates
- Pages using citations with accessdate and no URL
- Pages with script errors
- Commons category link is locally defined
- Implants
- Cardiology
- Radiology