Clopidogrel
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Sophia Saad, Associate Editor - WikiDoc [2]; Sree Teja Yelamanchili, MBBS [3]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING: DIMINISHED EFFECTIVENESS IN POOR METABOLIZERS
See full prescribing information for complete Boxed Warning.
* Effectiveness of clopidogrel depends on activation to an active metabolite by the cytochrome P450 (CYP) system, principally CYP2C19.
|
Overview
Clopidogrel is a P2Y12 platelet inhibitor, platelet aggregation inhibitor that is FDA approved for the treatment of acute coronary syndrome (ACS), recent MI, recent stroke, or established peripheral arterial disease. There is a Black Box Warning for this drug as shown here. Common adverse reactions include non-major bleeding.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Acute Coronary Syndrome
- For patients with non-ST-elevation ACS
- Initial loading dosage: 300 mg PO
- Maitaining dosage: 75 mg PO qd
- In combination with: Aspirin 75-300 mg PO qd
- For patients with STEMI
- Recommended dosage: 75 mg PO qd (With or without the loading dosage)
- In combination with: Aspirin 75-300 mg PO qd
Recent MI, Recent Stroke, or Established Peripheral Arterial Disease
- Dosing information
- 75 mg PO qd
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information about the guideline-supported use.
Non–Guideline-Supported Use
Prophylaxis of Thrombosis in Patient with Atrial Fibrillation
- Dosing Information
Prophylaxis of Thrombosis in Patient with Congestive Heart Failure
- Dosing information
- 75 mg/day[2]
Stasis Ulcer
- Dosing information
- Recommended dosage: 75 mg/day for 2-4 weeks[3]
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Clopidogrel FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information about the guideline-supported use.
Non–Guideline-Supported Use
Prophylaxis of Arterial thrombosis
- Dosing Information
- Recommended dosage: 0.2 mg/kg/day[4]
Contraindications
- Active Bleeding
- Clopidogrel is contraindicated in patients with active pathological bleeding such as peptic ulcer or intracranial hemorrhage.
- Clopidogrel tablets are contraindicated in patients with hypersensitivity (e.g., anaphylaxis) to clopidogrel or any component of the product.
Warnings
|
WARNING: DIMINISHED EFFECTIVENESS IN POOR METABOLIZERS
See full prescribing information for complete Boxed Warning.
* Effectiveness of clopidogrel depends on activation to an active metabolite by the cytochrome P450 (CYP) system, principally CYP2C19.
|
- Diminished Antiplatelet Activity Due to Impaired CYP2C19 Function
- Clopidogrel is a prodrug. Inhibition of platelet aggregation by clopidogrel is achieved through an active metabolite. The metabolism of clopidogrel to its active metabolite can be impaired by genetic variations in CYP2C19 and by concomitant medications that interfere with CYP2C19.
- Proton Pump Inhibitors
- Avoid concomitant use of clopidogrel with omeprazole or esomeprazole because both significantly reduce the antiplatelet activity of clopidogrel.
- General Risk of Bleeding
- Thienopyridines, including clopidogrel, increase the risk of bleeding. If a patient is to undergo surgery and an antiplatelet effect is not desired, discontinue clopidogrel five days prior to surgery. In patients who stopped therapy more than five days prior to CABG the rates of major bleeding were similar (event rate 4.4% clopidogrel + aspirin; 5.3% placebo + aspirin). In patients who remained on therapy within five days of CABG, the major bleeding rate was 9.6% for clopidogrel + aspirin, and 6.3% for placebo + aspirin.
- Thienopyridines inhibit platelet aggregation for the lifetime of the platelet (7 to 10 days), so withholding a dose will not be useful in managing a bleeding event or the risk of bleeding associated with an invasive procedure. Because the half-life of clopidogrel's active metabolite is short, it may be possible to restore hemostasis by administering exogenous platelets; however, platelet transfusions within 4 hours of the loading dose or 2 hours of the maintenance dose may be less effective.
- Discontinuation of Clopidogrel
- Avoid lapses in therapy, and if clopidogrel must be temporarily discontinued, restart as soon as possible. Premature discontinuation of clopidogrel may increase the risk of cardiovascular events.
- Patients With Recent Transient Ischemic Attack (TIA) or Stroke
- Thrombotic Thrombocytopenic Purpura (TTP)
- TTP, sometimes fatal, has been reported following use of clopidogrel, sometimes after a short exposure (< 2 weeks). TTP is a serious condition that requires urgent treatment including plasmapheresis (plasma exchange). It is characterized by thrombocytopenia, microangiopathic hemolytic anemia (schistocytes [fragmented RBCs] seen on peripheral smear), neurological findings, renal dysfunction, and fever.
- Cross-Reactivity Among Thienopyridines
- Hypersensitivity including rash, angioedema or hematologic reaction have been reported in patients receiving clopidogrel, including patients with a history of hypersensitivity or hematologic reaction to other thienopyridines.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions and durations of follow up, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- Clopidogrel bisulfate has been evaluated for safety in more than 54,000 patients, including over 21,000 patients treated for 1 year or more.
- The clinically important adverse reactions observed in trials comparing clopidogrel plus aspirin to placebo plus aspirin and trials comparing clopidogrel bisulfate alone to aspirin alone are discussed below.
Bleeding
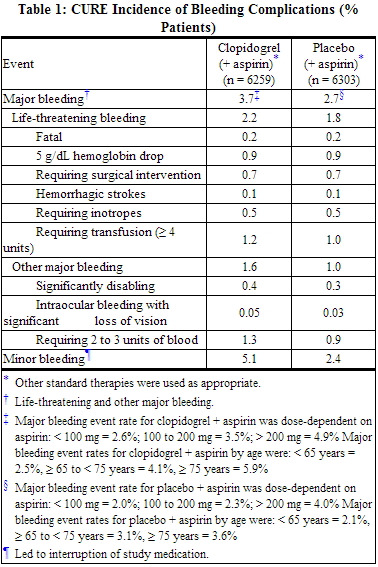
- CURE
- In CURE, clopidogrel bisulfate use with aspirin was associated with an increase in major bleeding (primarily gastrointestinal bleeding and at puncture sites) compared to placebo with aspirin. The incidence of intracranial hemorrhage (0.1%) and fatal bleeding (0.2%) were the same in both groups. Other bleeding events that were reported more frequently in the clopidogrel group were epistaxis, hematuria, and bruise.
The overall incidence of bleeding is described in Table 1.
- Ninety-two percent (92%) of the patients in the CURE study received heparin or low molecular weight heparin (LMWH), and the rate of bleeding in these patients was similar to the overall results.
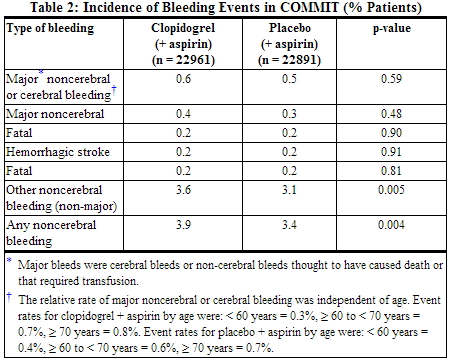
- COMMIT
- In COMMIT, similar rates of major bleeding were observed in the clopidogrel bisulfate and placebo groups, both of which also received aspirin.
- Ninety-two percent (92%) of the patients in the CURE study received heparin or low molecular weight heparin (LMWH), and the rate of bleeding in these patients was similar to the overall results.
COMMIT
- In COMMIT, similar rates of major bleeding were observed in the clopidogrel bisulfate and placebo groups, both of which also received aspirin.
- CAPRIE (Clopidogrel bisulfate vs. Aspirin)
- In CAPRIE, gastrointestinal hemorrhage occurred at a rate of 2.0% in those taking clopidogrel bisulfate vs. 2.7% in those taking aspirin; bleeding requiring hospitalization occurred in 0.7% and 1.1%, respectively. The incidence of intracranial hemorrhage was 0.4% for clopidogrel bisulfate compared to 0.5% for aspirin.
- Other bleeding events that were reported more frequently in the clopidogrel bisulfate group were epistaxis and hematoma.
- Other Adverse Events
- In CURE and CHARISMA, which compared clopidogrel bisulfate plus aspirin to aspirin alone, there was no difference in the rate of adverse events (other than bleeding) between clopidogrel bisulfate and placebo.
- In CAPRIE, which compared clopidogrel bisulfate to aspirin, pruritus was more frequently reported in those taking clopidogrel bisulfate. No other difference in the rate of adverse events (other than bleeding) was reported.
Postmarketing Experience
The following adverse reactions have been identified during post-approval use of clopidogrel. Because these reactions are reported voluntarily from a population of an unknown size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. Blood and lymphatic system disorders: Agranulocytosis, aplastic anemia/pancytopenia, thrombotic thrombocytopenic purpura (TTP), acquired hemophilia A
- Eye disorders: Eye (conjunctival, ocular, retinal) bleeding
- Gastrointestinal disorders: Gastrointestinal and retroperitoneal hemorrhage with fatal outcome, colitis (including ulcerative or lymphocytic colitis), pancreatitis, stomatitis, gastric/duodenal ulcer, diarrhea
- General disorders and administration site condition: Fever, hemorrhage of operative wound
- Hepato-biliary disorders: Acute liver failure, hepatitis (non-infectious), abnormal liver function test
- Immune system disorders: Hypersensitivity reactions, anaphylactoid reactions, serum sickness
- Musculoskeletal, connective tissue and bone disorders: Musculoskeletal bleeding, myalgia, arthralgia, arthritis
- Nervous system disorders: Taste disorders, fatal intracranial bleeding, headache
- Psychiatric disorders: Confusion, hallucinations
- Respiratory, thoracic and mediastinal disorders: Bronchospasm, interstitial pneumonitis, respiratory tract bleeding, eosinophilic pneumonia
- Renal and urinary disorders: Increased creatinine levels
- Skin and subcutaneous tissue disorders: Maculopapular, erythematous or exfoliative rash, urticaria, bullous dermatitis, eczema, toxic epidermal necrolysis, Stevens-Johnson syndrome, angioedema, drug-induced hypersensitivity syndrome, drug rash with eosinophilia and systemic symptoms (DRESS), erythema multiforme, skin bleeding, lichen planus, generalized pruritus
- Vascular disorders: Vasculitis, hypotension
Drug Interactions
CYP2C19 Inhibitors
- Clopidogrel is metabolized to its active metabolite in part by CYP2C19. Concomitant use of certain drugs that inhibit the activity of this enzyme results in reduced plasma concentrations of the active metabolite of clopidogrel and a reduction in platelet inhibition.
Proton Pump Inhibitors (PPI)
- Avoid concomitant use of clopidogrel with omeprazole or esomeprazole. In clinical studies, omeprazole was shown to reduce the antiplatelet activity of clopidogrel when given concomitantly or 12 hours apart. A higher dose regimen of clopidogrel concomitantly administered with omeprazole increases antiplatelet response; an appropriate dose regimen has not been established. A similar reduction in antiplatelet activity was observed with esomeprazole when given concomitantly with clopidogrel. Consider using another acid-reducing agent with minimal or no CYP2C19 inhibitory effect on the formation of clopidogrel active metabolite. Dexlansoprazole, lansoprazole and pantoprazole had less effect on the antiplatelet activity of clopidogrel than did omeprazole or esomeprazole.
Non-Steroidal Anti-Inflammatory Drugs (NSAIDs)
- Coadministration of clopidogrel and NSAIDs increases the risk of gastrointestinal bleeding.
Warfarin (CYP2C9 Substrates)
- Although the administration of clopidogrel 75 mg per day did not modify the pharmacokinetics of S-warfarin (a CYP2C9 substrate) or INR in patients receiving long-term warfarin therapy, coadministration of clopidogrel with warfarin increases the risk of bleeding because of independent effects on hemostasis.
- However, at high concentrations in vitro, clopidogrel inhibits CYP2C9.
SSRIs and SNRIs
- Since selective serotonin reuptake inhibitors (SSRIs) and serotonin norepinephrine reuptake inhibitors (SNRIs) affect platelet activation, the concomitant administration of SSRIs and SNRIs with clopidogrel may increase the risk of bleeding.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): B
Reproduction studies performed in rats and rabbits at doses up to 500 and 300 mg/kg/day, respectively (65 and 78 times the recommended daily human dose, respectively, on a mg/m2 basis), revealed no evidence of impaired fertility or fetotoxicity due to clopidogrel. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of a human response, clopidogrel should be used during pregnancy only if clearly needed.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Clopidogrel in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Clopidogrel during labor and delivery.
Nursing Mothers
Studies in rats have shown that clopidogrel and/or its metabolites are excreted in the milk. It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from clopidogrel, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
Safety and effectiveness in pediatric populations have not been established. Additional information describing a clinical study in which efficacy was not demonstrated in neonates and infants is approved in the package insert for Bristol-Myers Squibb’s clopidogrel tablets. However, due to Bristol-Myers Squibb’s marketing exclusivity rights, this drug product is not labeled with that pediatric information.
Geriatic Use
Of the total number of subjects in the CAPRIE and CURE controlled clinical studies, approximately 50% of patients treated with clopidogrel were 65 years of age and older, and 15% were 75 years and older. In COMMIT, approximately 58% of the patients treated with clopidogrel were 60 years and older, 26% of whom were 70 years and older. The observed risk of bleeding events with clopidogrel plus aspirin versus placebo plus aspirin by age category is provided in Table 1 and Table 2 for the CURE and COMMIT trials, respectively. No dosage adjustment is necessary in elderly patients.
Gender
There is no FDA guidance on the use of Clopidogrel with respect to specific gender populations.
Race
There is no FDA guidance on the use of Clopidogrel with respect to specific racial populations.
Renal Impairment
Experience is limited in patients with severe and moderate renal impairment.
Hepatic Impairment
No dosage adjustment is necessary in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Clopidogrel in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Clopidogrel in patients who are immunocompromised.
Administration and Monitoring
Administration
Acute Coronary Syndrome
Clopidogrel tablets USP can be administered with or without food. For patients with non-ST-elevation ACS (UA/NSTEMI), initiate clopidogrel tablets USP with a single 300 mg oral loading dose and then continue at 75 mg once daily. Initiate aspirin (75 to 325 mg once daily) and continue in combination with clopidogrel tablets USP. For patients with STEMI, the recommended dose of clopidogrel tablets USP is 75 mg once daily orally, administered in combination with aspirin (75 to 325 mg once daily), with or without thrombolytics. Clopidogrel tablets USP may be initiated with or without a loading dose.
Recent MI, Recent Stroke, or Established Peripheral Arterial Disease
The recommended daily dose of clopidogrel tablets USP is 75 mg once daily orally, with or without food.
CYP2C19 Poor Metabolizers
CYP2C19 poor metabolizer status is associated with diminished antiplatelet response to clopidogrel. Although a higher dose regimen in poor metabolizers increases antiplatelet response, an appropriate dose regimen for this patient population has not been established.
Use With Proton Pump Inhibitors (PPI)
Avoid using omeprazole or esomeprazole with clopidogrel tablets USP. Omeprazole and esomeprazole significantly reduce the antiplatelet activity of clopidogrel tablets USP. When concomitant administration of a PPI is required, consider using another acid-reducing agent with minimal or no CYP2C19 inhibitory effect on the formation of clopidogrel active metabolite.
Monitoring
There is limited information regarding Clopidogrel Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Clopidogrel and IV administrations.
Overdosage
Platelet inhibition by clopidogrel is irreversible and will last for the life of the platelet. Overdose following clopidogrel administration may result in bleeding complications. A single oral dose of clopidogrel at 1500 or 2000 mg/kg was lethal to mice and to rats and at 3000 mg/kg to baboons. Symptoms of acute toxicity were vomiting, prostration, difficult breathing, and gastrointestinal hemorrhage in animals. Based on biological plausibility, platelet transfusion may restore clotting ability.
Pharmacology
Mechanism of Action
Clopidogrel is an inhibitor of platelet activation and aggregation through the irreversible binding of its active metabolite to the P2Y12 class of ADP receptors on platelets.
Structure
Clopidogrel bisulfate, USP is a thienopyridine class inhibitor of P2Y12 ADP platelet receptors. Chemically it is methyl (+)-(S)-α-(2-chlorophenyl)-6,7-dihydrothieno[3,2-c]pyridine-5(4H)-acetate sulfate (1:1). The structural formula is as follows:
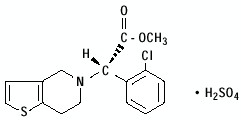
C16H16ClNO2S•H2SO4 M.W. 419.9 Clopidogrel bisulfate, USP is a white to off-white powder. It is practically insoluble in water at neutral pH but freely soluble at pH 1. It also dissolves freely in methanol, dissolves sparingly in methylene chloride, and is practically insoluble in ethyl ether. It has a specific optical rotation of about +56°. Clopidogrel bisulfate, USP for oral administration is provided as light-pink to pink, debossed, film-coated, capsule shaped tablets containing 97.875 mg of clopidogrel bisulfate, USP which is the molar equivalent of 75 mg of clopidogrel base. Each tablet contains the following inactive ingredients: crospovidone, hydrogenated vegetable oil, hydroxypropyl cellulose, hypromellose, indigo carmine aluminum lake FD&C blue #2, iron oxide red, iron oxide yellow, lactose, lactose monohydrate, microcrystalline cellulose, polyethylene glycol, sodium lauryl sulfate, and titanium dioxide.
Pharmacodynamics
Clopidogrel must be metabolized by CYP450 enzymes to produce the active metabolite that inhibits platelet aggregation. The active metabolite of clopidogrel selectively inhibits the binding of adenosine diphosphate (ADP) to its platelet P2Y12 receptor and the subsequent ADP-mediated activation of the glycoprotein GPIIb/IIIa complex, thereby inhibiting platelet aggregation. This action is irreversible. Consequently, platelets exposed to clopidogrel’s active metabolite are affected for the remainder of their lifespan (about 7 to 10 days). Platelet aggregation induced by agonists other than ADP is also inhibited by blocking the amplification of platelet activation by released ADP. Dose-dependent inhibition of platelet aggregation can be seen 2 hours after single oral doses of clopidogrel. Repeated doses of 75 mg clopidogrel per day inhibit ADP-induced platelet aggregation on the first day, and inhibition reaches steady state between Day 3 and Day 7. At steady state, the average inhibition level observed with a dose of 75 mg clopidogrel per day was between 40% and 60%. Platelet aggregation and bleeding time gradually return to baseline values after treatment is discontinued, generally in about 5 days.
Geriatric Patients
Elderly (≥ 75 years) and young healthy subjects had similar effects on platelet aggregation.
Renally-Impaired Patients
After repeated doses of 75 mg clopidogrel per day, patients with severe renal impairment (creatinine clearance from 5 to 15 mL/min) and moderate renal impairment (creatinine clearance from 30 to 60 mL/min) showed low (25%) inhibition of ADP-induced platelet aggregation.
Hepatically-Impaired Patients
After repeated doses of 75 mg clopidogrel per day for 10 days in patients with severe hepatic impairment, inhibition of ADP-induced platelet aggregation was similar to that observed in healthy subjects.
Gender
In a small study comparing men and women, less inhibition of ADP-induced platelet aggregation was observed in women.
Pharmacokinetics
Clopidogrel is a prodrug and is metabolized to a pharmacologically active metabolite and inactive metabolites.
Absorption
- After single and repeated oral doses of 75 mg per day, clopidogrel is rapidly absorbed. Absorption is at least 50%, based on urinary excretion of clopidogrel metabolites.
Effect of Food
- Clopidogrel can be administered with or without food. In a study in healthy male subjects when clopidogrel 75 mg per day was given with a standard breakfast, mean inhibition of ADP-induced platelet aggregation was reduced by less than 9%. The active metabolite AUC0-24 was unchanged in the presence of food, while there was a 57% decrease in active metabolite Cmax. Similar results were observed when a clopidogrel 300 mg loading dose was administered with a high-fat breakfast.
Metabolism
- Clopidogrel is extensively metabolized by two main metabolic pathways: one mediated by esterases and leading to hydrolysis into an inactive carboxylic acid derivative (85% of circulating metabolites) and one mediated by multiple cytochrome P450 enzymes. Cytochromes first oxidize clopidogrel to a 2-oxo-clopidogrel intermediate metabolite. Subsequent metabolism of the 2-oxo-clopidogrel intermediate metabolite results in formation of the active metabolite, a thiol derivative of clopidogrel. This metabolic pathway is mediated by CYP2C19, CYP3A, CYP2B6 and CYP1A2. The active thiol metabolite binds rapidly and irreversibly to platelet receptors, thus inhibiting platelet aggregation for the lifespan of the platelet.
- The Cmax of the active metabolite is twice as high following a single 300 mg clopidogrel loading dose as it is after four days of 75 mg maintenance dose. Cmax occurs approximately 30 to 60 minutes after dosing. In the 75 to 300 mg dose range, the pharmacokinetics of the active metabolite deviates from dose proportionality: increasing the dose by a factor of four results in 2.0 and 2.7 fold increases in Cmax and AUC, respectively.
Elimination
- Following an oral dose of 14C-labeled clopidogrel in humans, approximately 50% of total radioactivity was excreted in urine and approximately 46% in feces over the 5 days post-dosing. After a single, oral dose of 75 mg, clopidogrel has a half-life of approximately 6 hours. The half-life of the active metabolite is about 30 minutes.
Drug Interactions
- Clopidogrel is metabolized to its active metabolite in part by CYP2C19. Concomitant use of certain inhibitors of this enzyme results in reduced plasma concentrations of the active metabolite of clopidogrel and a reduction in platelet inhibition.
Proton Pump Inhibitors (PPI)
- The effect of proton pump inhibitors (PPI) on the systemic exposure to the clopidogrel active metabolite following multiple doses of clopidogrel 75 mg evaluated in dedicated drug interaction studies is presented in Figure 1.
Nonclinical Toxicology
There was no evidence of tumorigenicity when clopidogrel was administered for 78 weeks to mice and 104 weeks to rats at dosages up to 77 mg/kg per day, which afforded plasma exposures > 25 times that in humans at the recommended daily dose of 75 mg. Clopidogrel was not genotoxic in four in vitro tests (Ames test, DNA-repair test in rat hepatocytes, gene mutation assay in Chinese hamster fibroblasts, and metaphase chromosome analysis of human lymphocytes) and in one in vivo test (micronucleus test by oral route in mice). Clopidogrel was found to have no effect on fertility of male and female rats at oral doses up to 400 mg/kg per day (52 times the recommended human dose on a mg/m2 basis).
Clinical Studies
Acute Coronary Syndrome
CURE
The CURE study included 12,562 patients with ACS without ST-elevation (UA or NSTEMI) and presenting within 24 hours of onset of the most recent episode of chest pain or symptoms consistent with ischemia. Patients were required to have either ECG changes compatible with new ischemia (without ST-elevation) or elevated cardiac enzymes or troponin I or T to at least twice the upper limit of normal. The patient population was largely Caucasian (82%) and included 38% women, and 52% patients ≥ 65 years of age. Patients were randomized to receive clopidogrel (300 mg loading dose followed by 75 mg once daily) or placebo, and were treated for up to one year. Patients also received aspirin (75 to 325 mg once daily) and other standard therapies such as heparin. The use of GPIIb/IIIa inhibitors was not permitted for three days prior to randomization. The number of patients experiencing the primary outcome (CV death, MI, or stroke) was 582 (9.3%) in the clopidogrel-treated group and 719 (11.4%) in the placebo-treated group, a 20% relative risk reduction (95% CI of 10% to 28%; p < 0.001) for the clopidogrel-treated group.

Most of the benefit of clopidogrel occurred in the first two months, but the difference from placebo was maintained throughout the course of the trial (up to 12 months).

In CURE, the use of clopidogrel was associated with a lower incidence of CV death, MI or stroke in patient populations with different characteristics, as shown in Figure 3. The benefits associated with clopidogrel were independent of the use of other acute and long-term cardiovascular therapies, including heparin/LMWH, intravenous glycoprotein IIb/IIIa (GPIIb/IIIa) inhibitors, lipid-lowering drugs, beta-blockers, and ACE-inhibitors. The efficacy of clopidogrel was observed independently of the dose of aspirin (75 to 325 mg once daily). The use of oral anticoagulants, non-study antiplatelet drugs, and chronic NSAIDs was not allowed in CURE.
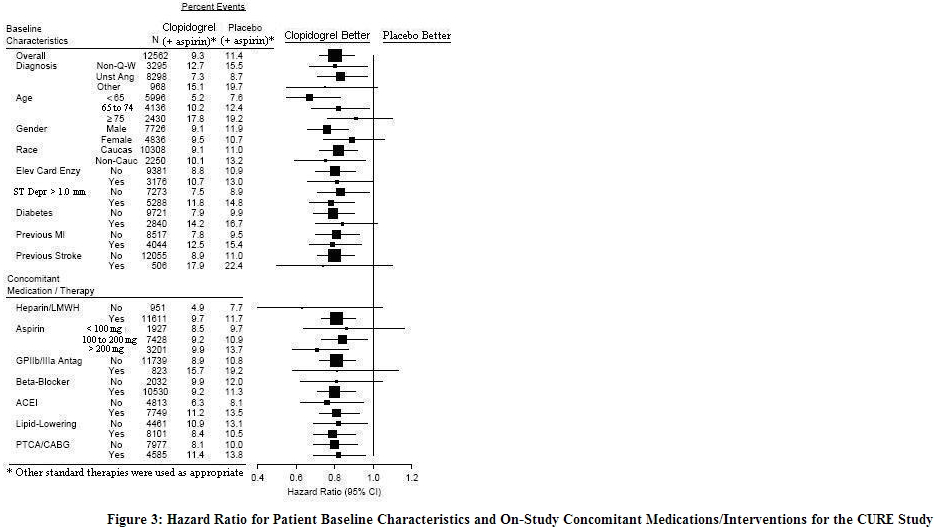
The use of clopidogrel in CURE was associated with a decrease in the use of thrombolytic therapy (71 patients [1.1%] in the clopidogrel group, 126 patients [2.0%] in the placebo group; relative risk reduction of 43%), and GPIIb/IIIa inhibitors (369 patients [5.9%] in the clopidogrel group, 454 patients [7.2%] in the placebo group, relative risk reduction of 18%). The use of clopidogrel in CURE did not affect the number of patients treated with CABG or PCI (with or without stenting), (2253 patients [36.0%] in the clopidogrel group, 2324 patients [36.9%] in the placebo group; relative risk reduction of 4.0%).
COMMIT
In patients with STEMI, the safety and efficacy of clopidogrel were evaluated in the randomized, placebo-controlled, double-blind study, COMMIT. COMMIT included 45,852 patients presenting within 24 hours of the onset of the symptoms of myocardial infarction with supporting ECG abnormalities (i.e., ST-elevation, ST-depression or left bundle-branch block). Patients were randomized to receive clopidogrel (75 mg once daily) or placebo, in combination with aspirin (162 mg per day), for 28 days or until hospital discharge, whichever came first. The primary endpoints were death from any cause and the first occurrence of re-infarction, stroke or death. The patient population included 28% women, 58% age ≥ 60 years (26% age ≥ 70 years), 55% patients who received thrombolytics, 68% who received ACE-inhibitors, and only 3% who underwent PCI. As shown in Table 5 and Figures 4 and 5 below, clopidogrel significantly reduced the relative risk of death from any cause by 7% (p = 0.029), and the relative risk of the combination of re-infarction, stroke or death by 9% (p = 0.002).
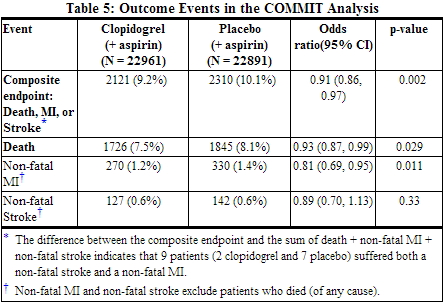
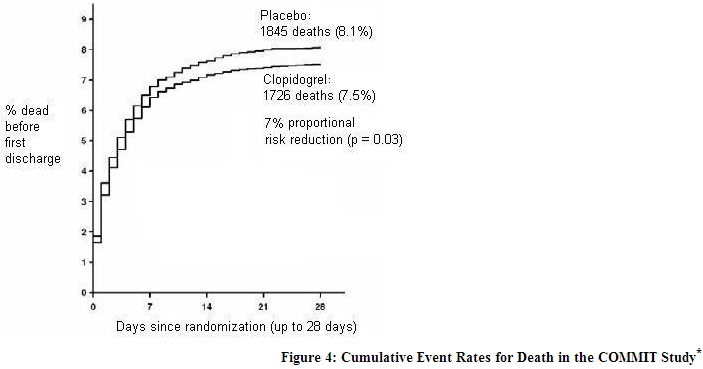
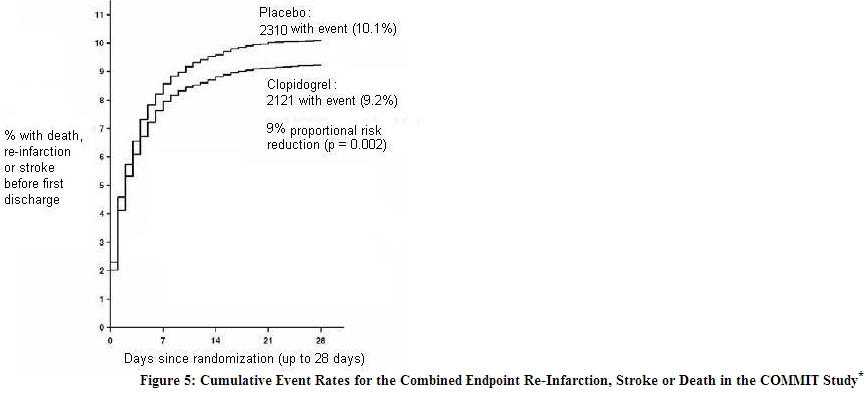
- All treated patients received aspirin.
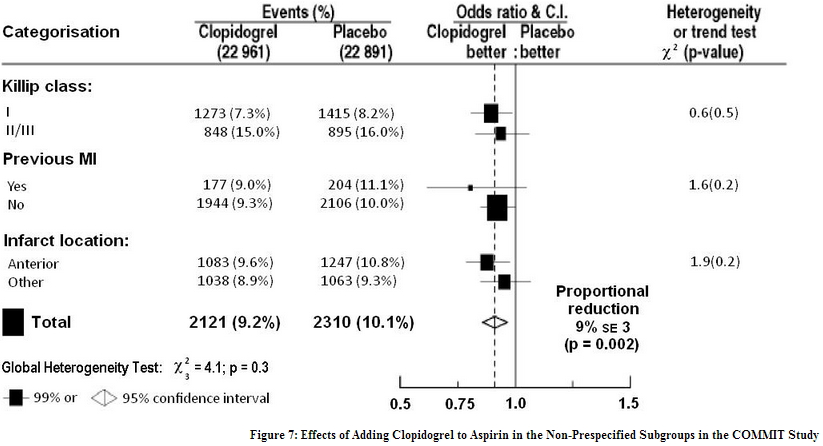
The effect of clopidogrel did not differ significantly in various pre-specified subgroups as shown in Figure 6. The effect was also similar in non-prespecified subgroups including those based on infarct location, Killip class or prior MI history. Such subgroup analyses should be interpreted cautiously.
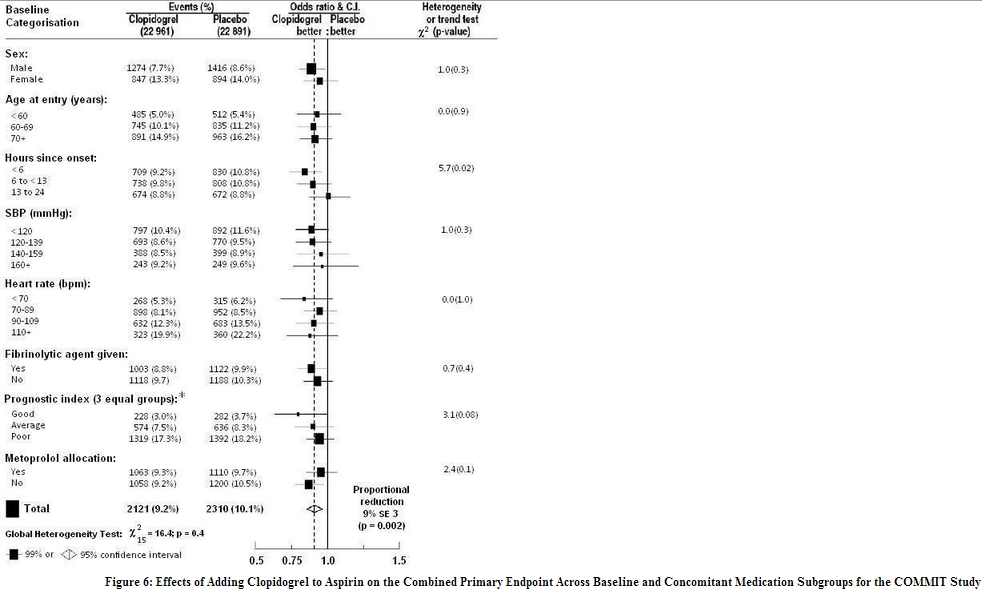
- Three similar-sized prognostic index groups were based on absolute risk of primary composite outcome for each patient calculated from baseline prognostic variables (excluding allocated treatments) with a Cox regression model.
Recent Myocardial Infarction, Recent Stroke, or Established Peripheral Arterial Disease
CAPRIE
The CAPRIE trial was a 19,185 patient, 304 center, international, randomized, double-blind, parallel-group study comparing clopidogrel (75 mg daily) to aspirin (325 mg daily). The patients randomized had: 1) recent histories of myocardial infarction (within 35 days); 2) recent histories of ischemic stroke (within 6 months) with at least a week of residual neurological signs; or 3) established peripheral arterial disease. Patients received randomized treatment for an average of 1.6 years (maximum of 3 years). The trial's primary outcome was the time to first occurrence of new ischemic stroke (fatal or not), new myocardial infarction (fatal or not), or other vascular death. Deaths not easily attributable to nonvascular causes were all classified as vascular.
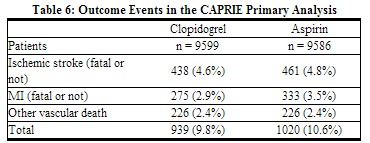
As shown in Table 6, clopidogrel was associated with a lower incidence of outcome events, primarily MI. The overall relative risk reduction (9.8% vs. 10.6%) was 8.7%, p = 0.045. Similar results were obtained when all-cause mortality and all-cause strokes were counted instead of vascular mortality and ischemic strokes (risk reduction 6.9%). In patients who survived an on-study stroke or myocardial infarction, the incidence of subsequent events was lower in the clopidogrel group. The curves showing the overall event rate are shown in Figure 8. The event curves separated early and continued to diverge over the 3 year follow-up period.
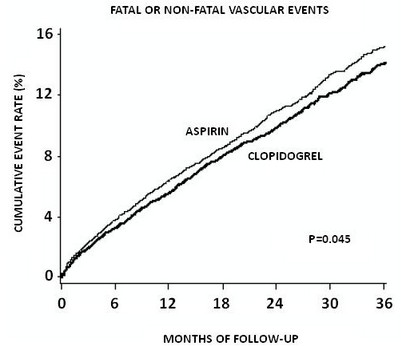
The statistical significance favoring clopidogrel over aspirin was marginal (p = 0.045). However, because aspirin is itself effective in reducing cardiovascular events in patients with recent myocardial infarction or stroke, the effect of clopidogrel is substantial. The CAPRIE trial included a population that was randomized on the basis of 3 entry criteria. The efficacy of clopidogrel relative to aspirin was heterogeneous across these randomized subgroups (p = 0.043). It is not clear whether this difference is real or a chance occurrence. Although the CAPRIE trial was not designed to evaluate the relative benefit of clopidogrel over aspirin in the individual patient subgroups, the benefit appeared to be strongest in patients who were enrolled because of peripheral vascular disease (especially those who also had a history of myocardial infarction) and weaker in stroke patients. In patients who were enrolled in the trial on the sole basis of a recent myocardial infarction, clopidogrel was not numerically superior to aspirin.
Lack of Established Benefit of Clopidogrel Plus Aspirin in Patients With Multiple Risk Factors or Established Vascular Disease
CHARISMA
The CHARISMA trial was a 15,603 subject, randomized, double-blind, parallel group study comparing clopidogrel (75 mg daily) to placebo for prevention of ischemic events in patients with vascular disease or multiple risk factors for atherosclerosis. All subjects were treated with aspirin 75 to 162 mg daily. The mean duration of treatment was 23 months. The study failed to demonstrate a reduction in the occurrence of the primary endpoint, a composite of CV death, MI, or stroke. A total of 534 (6.9%) patients in the clopidogrel group versus 573 (7.4%) patients in the placebo group experienced a primary outcome event (p = 0.22). Bleeding of all severities was more common in the subjects randomized to clopidogrel.
How Supplied
Clopidogrel tablets USP are available as follows: 75 mg – light-pink to pink, film-coated, capsule shaped tablets debossed with “TV” on one side and “7314” on the other side, in bottles of 30, 90, and 500.
Storage
Store at 20° to 25°C (68° to 77°F) [See USP Controlled Room Temperature]. Dispense in a tight, light-resistant container as defined in the USP, with a child-resistant closure (as required).
Images
Drug Images
{{#ask: Page Name::Clopidogrel |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Clopidogrel |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
For patient information about clopidogrel, click here.
Precautions with Alcohol
Alcohol-Clopidogrel interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Clopidogrel Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Clopidogrel Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ ACTIVE Investigators. Connolly SJ, Pogue J, Hart RG, Hohnloser SH, Pfeffer M et al. (2009) Effect of clopidogrel added to aspirin in patients with atrial fibrillation. N Engl J Med 360 (20):2066-78. DOI:10.1056/NEJMoa0901301 PMID: 19336502
- ↑ Massie BM, Collins JF, Ammon SE, Armstrong PW, Cleland JG, Ezekowitz M et al. (2009) Randomized trial of warfarin, aspirin, and clopidogrel in patients with chronic heart failure: the Warfarin and Antiplatelet Therapy in Chronic Heart Failure (WATCH) trial. Circulation 119 (12):1616-24. DOI:10.1161/CIRCULATIONAHA.108.801753 PMID: 19289640
- ↑ Bick RL, Scott RG (2001) Stasis ulcers refractory to therapy--accelerated healing by treatment with clopidogrel +/- dalteparin: a preliminary report. Clin Appl Thromb Hemost 7 (1):21-4. PMID: 11190899
- ↑ Li JS, Yow E, Berezny KY, Bokesch PM, Takahashi M, Graham TP et al. (2008) Dosing of clopidogrel for platelet inhibition in infants and young children: primary results of the Platelet Inhibition in Children On Clopidogrel (PICOLO) trial. Circulation 117 (4):553-9. DOI:10.1161/CIRCULATIONAHA.107.715821 PMID: 18195173
{{#subobject:
|Page Name=Clopidogrel |Pill Name=Clopidogrel 75 mg NDC 0093-7314.JPG |Drug Name=Clopidogrel 75 MG (as clopidogrel bisulfate 97.875 MG) Oral Tablet |Pill Ingred=crospovidone, hydrogenated cottonseed oil, hydroxypropyl cellulose (type h), hypromellose 2910 (15 mpa.s), indigotindisulfonate sodium, aluminum oxide, fd&c blue no. 2, ferric oxide red, ferric oxide yellow, lactose, lactose monohydrate, cellulose, microcrystalline, polyethylene glycol 4000, sodium lauryl sulfate, titanium dioxide|+sep=; |Pill Imprint=TV;7314 |Pill Dosage=75 mg |Pill Color=Pink|+sep=; |Pill Shape=Oval |Pill Size (mm)=13.00 |Pill Scoring=1 |Pill Image= |Drug Author=Teva Pharmaceuticals USA Inc |NDC=0093-7314
}}
{{#subobject:
|Label Page=Clopidogrel |Label Name=Clopidogrel_label_01.jpg
}}
{{#subobject:
|Label Page=Clopidogrel |Label Name=Clopidogrel_panel_01.jpg
}}