Deslorelin
 | |
| Clinical data | |
|---|---|
| Trade names | Ovuplant |
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | implant / Inhalation |
| ATCvet code | |
| Pharmacokinetic data | |
| Excretion | renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| UNII | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
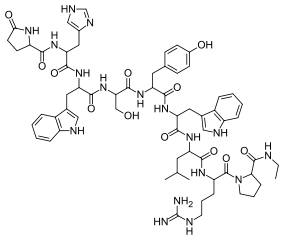
| Formula | C64H83N17O12 |
| Molar mass | 1282.45 g/mol |
| 3D model (JSmol) | |
| |
| | |
|
WikiDoc Resources for Deslorelin |
|
Articles |
|---|
|
Most recent articles on Deslorelin |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Deslorelin at Clinical Trials.gov Clinical Trials on Deslorelin at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Deslorelin
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Deslorelin Discussion groups on Deslorelin Patient Handouts on Deslorelin Directions to Hospitals Treating Deslorelin Risk calculators and risk factors for Deslorelin
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Deslorelin |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Deslorelin acetate is an injectable gonadotropin releasing hormone super-agonist (GnRH agonist) also known as an LHRH agonist. It stops the production of sex hormones (testosterone and oestrogen).
Deslorelin Acetate is marketed by Peptech with the brand name Ovuplant.[1] It is currently approved for use in veterinary medicine and is used to induce ovulation in mares as part of the artificial insemination process. It is also used to stabilize high-risk pregnancies, mainly of livestock. Unlike other GnRH agonists, which are mainly used to inhibit luteinizing hormone and follicle-stimulating hormone by their ultimate downregulation of the pituitary gland, Deslorelin is primarily used for the initial flare effect upon the pituitary, and its associated surge of LH secretion.
Pharmacokinetics
Deslorelin is a synthetic analogue of a naturally occurring luteinising-hormone releasing hormone (LHRH). Bioavailability is almost complete.
Clinical trials
Deslorelin was successfully trialed in the US and was approved for veterinary use under certain circumstances. In Europe, it was approved for use in equine assisted reproduction.[2]
It is also being trialed in humans to study its efficacy in treatment of breast cancer in women,[3] and in treating precocious puberty and congenital adrenal hyperplasia in male and female children.[4]
As of August 2011 this drug was not approved for general use in the US, other than within approved clinical trials. Orphan drug status has been designated in the US, though approval had not been issued as of 2011.[5]
References
- ↑ Peptech official Ovuplant site
- ↑ Deslorelin Acetate - European Agency for the Evaluation of Medicinal Products
- ↑ Deslorelin Combined With Low-Dose Add-Back Estradiol and Testosterone in Preventing Breast Cancer in Premenopausal Women Who Are at High Risk for This Disease - National Cancer Institute
- ↑ Three Drug Combination Therapy Versus Conventional Treatment of Children With Congenital Adrenal Hyperplasia - National Institutes of Health Clinical Center
- ↑ "Results for Orphan Drug Product Designations Search". FDA. Retrieved 3 September 2011.
- Pages with script errors
- Template:drugs.com link with non-standard subpage
- Articles with changed CASNo identifier
- Articles with changed EBI identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- GnRH agonists
- Peptides
- Drug