Decay chain
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
In nuclear science, the decay chain refers to the radioactive decay of different discrete radioactive decay products as a chained series of transformations. Most radioactive elements do not decay directly to a stable state, but rather undergo a series of decays until eventually a stable isotope is reached.
Decay stages are referred to by their relationship to previous or subsequent stages. A parent isotope is one that undergoes decay to form a daughter isotope. The daughter isotope may be stable or it may decay to form a daughter isotope of its own. The daughter of a daughter isotope is sometimes called a granddaughter isotope.
The time it takes for a single parent atom to decay to an atom of its daughter isotope can vary widely, not only for different parent-daughter chains, but also for identical pairings of parent and daughter isotopes. While the decay of a single atom occurs spontaneously, the decay of an initial population of identical atoms over time, t, follows a decaying exponential distribution, e-λt, where λ is called a decay constant. Because of this exponential nature, one of the properties of an isotope is its half-life, the time by which half of an initial number of identical parent radioisotopes have decayed to their daughters. Half-lives have been determined in laboratories for thousands of radioisotopes (or, radionuclides). These can range from nearly instantaneous to as much as 1019 years or more.
The intermediate stages often emit more radioactivity than the original radioisotope: when equilibrium is achieved, a granddaughter isotope is present in proportion to its half-life; but since its activity is inversely proportional to its half-life, any nucleid in the decay chain finally contributes as much as the head of the chain. For example, natural uranium is not significantly radioactive, but samples of pitchblende, a uranium ore, are 13 times more radioactive because of the radium and other daughter isotopes they contain. Not only are unstable radium isotopes significant radioactive emitters, but they also generate gaseous radon as the next stage in the decay chain. Thus, radon is a naturally occurring radioactive gas, which is the leading cause of lung cancer in non-smokers[2].
Types
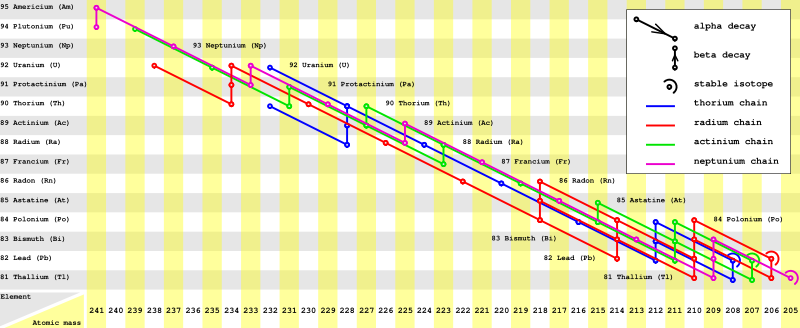
The four most common modes of radioactive decay are: alpha decay, beta minus decay, beta plus decay (considered as both positron emission and electron capture) and isomeric transition. Of these decay processes, alpha decay changes the atomic mass number of the nucleus, and always decreases it by four. Because of this, almost any decay will result in a nucleus whose atomic mass number has the same residue mod 4, dividing all nuclides into four classes. The members of any possible decay chain must be drawn entirely from one of these classes.
Three main decay chains (or families) are observed in nature, commonly called the thorium series, the radium series (not uranium series), and the actinium series, representing three of these four classes, and ending in three different, stable isotopes of lead. The mass number of every isotope in these chains can be represented as A=4n, A=4n+2 and A=4n+3, respectively. The long lived starting isotopes 232Th, 238U and 235U of these three have existed since the formation of the earth; the precursor 244Pu has also been found in minute amounts on earth[1]. The fourth chain, the neptunium series with A=4n+1, due to quite short half life time of its starting isotope 237Np, is already extinct, except for the final rate-limiting step. The ending isotope of this chain is 205Tl. Some older sources give the final isotope as 209Bi, but it was recently discovered that 209Bi is radioactive with half-life of 1.9×1019 years.
There are also many shorter chains, for example carbon-14. On the earth, most of the starting isotopes of these chains are generated by cosmic radiation.
Actinide alpha decay chains
In the tables below, the minor branches of decay (with the branching ratio of less than 0.0001%) are omitted. The energy release includes the total kinetic energy of all the emitted particles (electrons, alpha particles, gamma quanta, neutrinos, Auger electrons and X-rays) and the recoil nucleus, assuming that the original nucleus was at rest.
In the tables below, the historic names of the naturally occurring nuclides are also given. These names were used at the time when the decay chains were first discovered and investigated. From these names one can infer the particular chain to which the nuclide belongs. Also, the names indicate similarities: for example, Tn, Rn and An are all inert gases.
Beta decay chains
Since heavy nuclei have a greater proportion of neutrons, fission product nuclei almost always start out with a neutron/proton ratio greater than what is stable for their mass range; therefore they undergo multiple beta decays in succession, each converting a neutron to a proton. The first decays tend to have higher decay energy and shorter half-life; the last decays may have low decay energy and/or long half-life.
For example, uranium-235 has 92 protons and 143 neutrons. Fission takes one more neutron, then produces two or three more neutrons; assume that 92 protons and 142 neutrons are available for the two fission product nuclei. Suppose they have mass 99 with 39 protons and 60 neutrons (yttrium-99), and mass 135 with 53 protons and 82 neutrons (iodine-135); then the decay chains are:
| Nuclide | Halflife |
|---|---|
| 99Y | 1.470(7) s |
| 99Zr | 2.1(1) s |
| 99Nb | 15.0(2) s |
| 99Mo | 2.7489(6) d |
| 99Tc | 2.111(12)E+5 a |
| 99Ru | Stable |
| Nuclide | Halflife |
|---|---|
| 135I | 6.57(2) h |
| 135Xe | 9.14(2) h |
| 135Cs | 2.3(3)E+6 a |
| 135Ba | Stable |
Literature
C.M. Lederer, J.M. Hollander, I. Perlman, Table of Isotopes, 6th ed., Wiley & Sons, New York 1968
- ↑ D.C . Hoffman, F. O. Lawrence, J. L. Mewheter, F. M. Rourke: Detection of Plutonium-244 in Nature. In: Nature, Nr. 34, 1971, pp. 132–134
External links
| Wikimedia Commons has media related to Decay chain. |
ast:Desintegración
ca:Cadena de desintegració
cs:Rozpadová řada
de:Zerfallsreihe
nl:Vervalreeks
sv:Sönderfallskedja