Colestipol
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Sophia Saad, Associate Editor - WikiDoc [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Colestipol is a bile acid sequestrant that is FDA approved for the treatment of primary hypercholesterolemia. Common adverse reactions include constipation and vomiting.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Primary Hypercholesterolemia
- Tablet dosing information
- Initial dosage: 2 g PO qd or bid
- Dosage increases of 2 g PO qd or bid should occur at 1- or 2-month intervals.
- Recommended dosage: 2 to 16 g/day given once or in divided doses
- Appropriate use of lipid profiles as per NCEP guidelines including LDL-C and triglycerides, is advised so that optimal but not excessive doses are used to obtain the desired therapeutic effect on LDL-C level. If the desired therapeutic effect is not obtained at 2 to 16 grams/day with good compliance and acceptable side effects, combined therapy or alternate treatment should be considered.
- Colestipol tablets must be taken one at a time and be promptly swallowed whole, using plenty of water or other appropriate liquid. Do not cut, crush, or chew the tablets. Patients should take other drugs at least one hour before or four hours after colestipol tablets to minimize possible interference with their absorption.
- During Administration of Colestipol Tablets
- The patient should be carefully monitored clinically, including serum cholesterol and triglyceride levels. Periodic determinations of serum cholesterol levels as outlined in the NCEP guidelines should be done to confirm a favorable initial and long-term response.
- Failure of total or LDL-C to fall within the desired range should lead one to first examine dietary and drug compliance. If these are deemed acceptable, combined therapy or alternate treatment should be considered.
- Significant rise in triglyceride level should be considered as indication for dose reduction, drug discontinuation, or combined or alternate therapy.
- Suspension dosing information
- One dose (1 packet or 1 level scoopful) of colestipol HCl for oral suspension contains 5 gram of colestipol hydrochloride.
- Initial dosage: one dose PO qd or bid with an increment of one dose/day at one- or two-month intervals.
- Recommended daily adult dose: one to six doses given once or in divided doses. '
- Appropriate use of lipid profiles as per NCEP guidelines including LDL-cholesterol and triglycerides is advised so that optimal, but not excessive doses are used to obtain the desired therapeutic effect on LDL-cholesterol level. If the desired therapeutic effect is not obtained at one to six doses/day with good compliance and acceptable side effects, combined therapy or alternate treatment should be considered.
- To avoid accidental inhalation or esophageal distress, colestipol HCl for oral suspension should not be taken in its dry form. Colestipol HCl for oral suspension should always be mixed with water or other fluids before ingesting. Patients should take other drugs at least one hour before or four hours after colestipol HCl for oral suspension to minimize possible interference with their absorption.
- The scoop accompanying this product is not interchangeable with other scoops.
- Before Administration of Colestipol Tablets
- Define the type of hyperlipoproteinemia, as described in NCEP guidelines.
- Institute a trial of diet and weight reduction.
- Establish baseline serum total and LDL-C and triglyceride levels.
- Before Colestipol HCl for Oral Suspension Administration
- Define the type of hyperlipoproteinemia, as described in NCEP guidelines.
- Institute a trial of diet and weight reduction.
- Establish baseline serum total and LDL-cholesterol and triglyceride levels.
- During Colestipol HCl for Oral Suspension Administration
- The patient should be carefully monitored clinically, including serum cholesterol and triglyceride levels. Periodic determinations of serum cholesterol levels as outlined in the NCEP guidelines should be done to confirm a favorable initial and longer term response.
- Failure of total or LDL-cholesterol to fall within the desired range should lead one to first examine dietary and drug compliance. If these are deemed acceptable, combined therapy or alternate treatment should be considered.
- Significant rise in triglyceride level should be considered as indication for dose reduction, drug discontinuation, or combined or alternate therapy.
- Mixing and Administration Guide
- Colestipol HCl for oral suspension should always be mixed in a liquid such as water or the beverage of your choice. It may also be taken in soups or with cereals or pulpy fruits. Colestipol HCl for oral suspension should never be taken in its dry form.
- With Beverages
- Add the prescribed amount of colestipol HCl for oral suspension to a glassful (three ounces or more) of water or the beverage of your choice. A heavy or pulpy juice may minimize complaints relative to consistency.
- Stir the mixture until the medication is completely mixed. (Colestipol HCl for oral suspension will not dissolve in the liquid.) Colestipol HCl for oral suspension may also be mixed with carbonated beverages, slowly stirred in a large glass. However, this mixture may be associated with GI complaints.
- Rinse the glass with a small amount of additional beverage to make sure all the medication is taken.
- With cereals, soups, and fruits
- Colestipol HCl for oral suspension may be taken mixed with milk in hot or regular breakfast cereals, or even mixed in soups that have a high fluid content. It may also be added to fruits that are pulpy such as crushed pineapple, pears, peaches, or fruit cocktail.
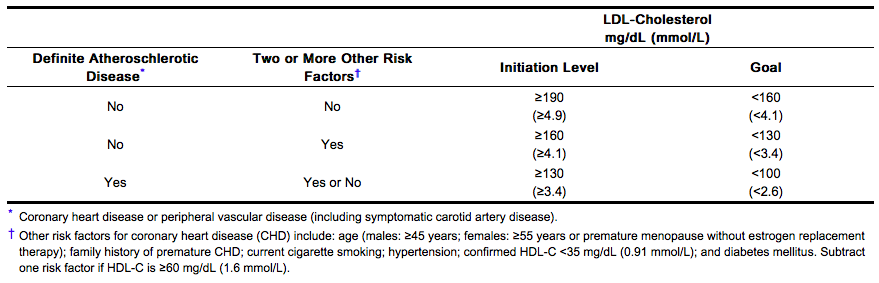
The NCEP guideline
- According to the NCEP guidelines, the goal of treatment is to lower LDL-C, and LDL-C is to be used to initiate and assess treatment response. Only if LDL-C levels are not available, should the Total-C be used to monitor therapy. The NCEP treatment guidelines are shown below.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information about Off-Label Guideline-Supported Use of Colestipol sandbox in adult patients.
Non–Guideline-Supported Use
Generalized Atherosclerosis
- Dosing information
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- Safety and effectiveness in the pediatric population have not been established.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
- There is limited information about Off-Label Guideline-Supported Use of Colestipol sandbox in pediatric patients.
Non–Guideline-Supported Use
Familial hypercholesterolemia
- Dosing information
Contraindications
- Colestipol tablets are contraindicated in those individuals who have shown hypersensitivity to any of its components.
- Colestipol HCl for oral suspension is contraindicated in those individuals who have shown hypersensitivity to any of its components.
Warnings
- To avoid accidental inhalation or esophageal distress, colestipol hcl for oral suspension should not be taken in its dry form. Always mix colestipol hcl for oral suspension with water or other fluids before ingesting.
Precautions
- Prior to initiating therapy with colestipol tablets, secondary causes of hypercholesterolemia (e.g., poorly controlled diabetes mellitus, hypothyroidism, nephrotic syndrome, dysproteinemias, obstructive liver disease, other drug therapy, alcoholism), should be excluded, and a lipid profile performed to assess total cholesterol, HDL-C, and triglycerides (TG). For individuals with TG less than 400 mg/dL (<4.5 mmol/L), LDL-C can be estimated using the following equation:
- LDL-C = Total cholesterol - [ (Triglycerides / 5)+HDL-C]
- For TG levels >400 mg/dL, this equation is less accurate and LDL-C concentrations should be determined by ultracentrifugation. In hypertriglyceridemic patients, LDL-C may be low or normal despite elevated Total-C. In such cases colestipol tablets may not be indicated.
- Because it sequesters bile acids, colestipol hydrochloride may interfere with normal fat absorption and, thus, may reduce absorption of folic acid and fat soluble vitamins such as A, D, and K.
- Chronic use of colestipol hydrochloride may be associated with an increased bleeding tendency due to hypoprothrombinemia from vitamin K deficiency. This will usually respond promptly to parenteral vitamin K1 and recurrences can be prevented by oral administration of vitamin K1.
- Serum cholesterol and triglyceride levels should be determined periodically based on NCEP guidelines to confirm a favorable initial and adequate long-term response.
- Colestipol tablets may produce or severely worsen pre-existing constipation. The dosage should be increased gradually in patients to minimize the risk of developing fecal impaction. In patients with pre-existing constipation, the starting dose should be 2 grams once or twice a day. Increased fluid and fiber intake should be encouraged to alleviate constipation and a stool softener may occasionally be indicated. If the initial dose is well tolerated, the dose may be increased by a further 2 to 4 grams/day (at monthly intervals) with periodic monitoring of serum lipoproteins. If constipation worsens or the desired therapeutic response is not achieved at 2 to 16 grams/day, combination therapy or alternate therapy should be considered. Particular effort should be made to avoid constipation in patients with symptomatic coronary artery disease. constipation associated with colestipol tablets may aggravate hemorrhoids.
- While there have been no reports of hypothyroidism induced in individuals with normal thyroid function, the theoretical possibility exists, particularly in patients with limited thyroid reserve.
- Since colestipol hydrochloride is a chloride form of an anion exchange resin, there is a possibility that prolonged use may lead to the development of hyperchloremic acidosis.
Adverse Reactions
Clinical Trials Experience
Gastrointestinal
- The most common adverse reactions are confined to the gastrointestinal tract. To achieve minimal GI disturbance with an optimal LDL-C lowering effect, a gradual increase of dosage starting with one dose/day is recommended. Constipation is the major single complaint and at times is severe. Most instances of constipation are mild, transient, and controlled with standard treatment. Increased fluid intake and inclusion of additional dietary fiber should be the first step; a stool softener may be added if needed. Some patients require decreased dosage or discontinuation of therapy. Hemorrhoids may be aggravated.
- Other, less frequent gastrointestinal complaints consist of abdominal discomfort (abdominal pain and cramping), intestinal gas, (bloating and flatulence), indigestion and heartburn, diarrhea and loose stools, and nausea and vomiting. Bleeding hemorrhoids and blood in the stool have been infrequently reported. Peptic ulceration, cholecystitis, and cholelithiasis have been rarely reported in patients receiving colestipol hydrochloride granules, and are not necessarily drug related.
- Difficulty swallowing and transient esophageal obstruction have been rarely reported in patients taking colestipol tablets.
- Transient and modest elevations of aspartate aminotransferase (AST, SGOT), alanine aminotransferase (ALT, SGPT) and alkaline phosphatase were observed on one or more occasions in various patients treated with colestipol hydrochloride.
- The following non-gastrointestinal adverse reactions have been reported with generally equal frequency in patients receiving colestipol tablets, colestipol hydrochloride granules or placebo in clinical studies:
Cardiovascular
- Chest pain, angina, and tachycardia have been infrequently reported.
Hypersensitivity
- Rash has been infrequently reported. Urticaria and dermatitis have been rarely noted in patients receiving colestipol hydrochloride granules.
Musculoskeletal
- Musculoskeletal pain, aches and pains in the extremities, joint pains, arthritis, and backache have been reported.
Neurologic
- Headache, migraine headache and sinus headache have been reported. Other infrequently reported complaints include dizziness, light-headedness, and insomnia.
Miscellaneous
- Anorexia, fatigue, weakness, shortness of breath, and swelling of the hands or feet, have been infrequently reported.
Postmarketing Experience
- FDA Package Insert for Abcixmab contains no information regarding Adverse Reactions.
Drug Interactions
- Since colestipol hydrochloride is an anion exchange resin, it may have a strong affinity for anions other than the bile acids. In vitro studies have indicated that colestipol hydrochloride binds a number of drugs. Therefore, colestipol tablets may delay or reduce the absorption of concomitant oral medication. The interval between the administration of colestipol tablets and any other medication should be as long as possible. Patients should take other drugs at least one hour before or four hours after colestipol tablets to avoid impeding their absorption.
- Repeated doses of colestipol hydrochloride given prior to a single dose of propranolol in human trials have been reported to decrease propranolol absorption. However, in a follow-up study in normal subjects, single dose administration of colestipol hydrochloride and propranolol and twice-a-day administration for 5 days of both agents did not effect the extent of propranolol absorption, but had a small yet statistically significant effect on its rate of absorption; the time to reach maximum concentration was delayed 30 minutes. Effects on the absorption of other beta-blockers have not been determined. Therefore, patients on propranolol should be observed when colestipol tablets are either added or deleted from a therapeutic regimen.
- Studies in humans show that the absorption of chlorothiazide as reflected in urinary excretion is markedly decreased even when administered one hour before colestipol hydrochloride. The absorption of tetracycline, furosemide, penicillin G, hydrochlorothiazide, and gemfibrozil was significantly decreased when given simultaneously with colestipol hydrochloride; these drugs were not tested to determine the effect of administration one hour before colestipol hydrochloride.
- No depressant effect on blood levels in humans was noted when colestipol hydrochloride was administered with any of the following drugs: aspirin, clindamycin, clofibrate, methyldopa, nicotinic acid (niacin), tolbutamide, phenytoin or warfarin. Particular caution should be observed with digitalis preparations since there are conflicting results for the effect of colestipol hydrochloride on the availability of digoxin and digitoxin. The potential for binding of these drugs if given concomitantly is present. Discontinuing colestipol hydrochloride could pose a hazard to health if a potentially toxic drug that is significantly bound to the resin has been titrated to a maintenance level while the patient was taking colestipol hydrochloride.
- Bile acid binding resins may also interfere with the absorption of oral phosphate supplements and hydrocortisone.
Use in Specific Populations
Pregnancy
- Since colestipol hydrochloride is essentially not absorbed systemically (less than 0.17% of the dose), it is not expected to cause fetal harm when administered during pregnancy in recommended dosages. There are no adequate and well controlled studies in pregnant women, and the known interference with absorption of fat soluble vitamins may be detrimental even in the presence of supplementation. The use of colestipol HCl for oral suspension in pregnancy or by women of childbearing potential requires that the potential benefits of drug therapy be weighed against possible hazards to the mother or child.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Colestipol in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Colestipol during labor and delivery.
Nursing Mothers
- Caution should be exercised when colestipol tablets are administered to a nursing mother. The possible lack of proper vitamin absorption described in the "pregnancy" section may have an effect on nursing infants.
Pediatric Use
- Safety and effectiveness in the pediatric population have not been established.
Geriatic Use
There is no FDA guidance on the use of Colestipol in geriatric settings.
Gender
There is no FDA guidance on the use of Colestipol with respect to specific gender populations.
Race
There is no FDA guidance on the use of Colestipol with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Colestipol in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Colestipol in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Colestipol in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Colestipol in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
Monitoring
- FDA Package Insert for Colestipol contains no information regarding drug monitoring.
IV Compatibility
- FDA Package Insert for Colestipol contains no information regarding IV Compatibility.
Overdosage
- Overdosage of colestipol tablets has not been reported. Should overdosage occur, however, the chief potential harm would be obstruction of the gastrointestinal tract. The location of such potential obstruction, the degree of obstruction and the presence or absence of normal gut motility would determine treatment.
- Overdosage of colestipol HCl for oral suspension has not been reported. Should overdosage occur, however, the chief potential harm would be obstruction of the gastrointestinal tract. The location of such potential obstruction, the degree of obstruction and the presence or absence of normal gut motility would determine treatment.
Pharmacology
| |
Colestipol
| |
| Systematic (IUPAC) name | |
| Copolymer of bis(2-aminoethyl)amine and 2-(chloromethyl)oxirane | |
| Identifiers | |
| CAS number | 37296-80-3 (HCl) |
| ATC code | C10 |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | (C4H10N3)m(C3H6O)n |
| Mol. mass | ? |
| Pharmacokinetic data | |
| Bioavailability | None |
| Metabolism | ? |
| Half life | ? |
| Excretion | Faeces, in complex with bile acids |
| Therapeutic considerations | |
| Pregnancy cat. |
B(US) |
| Legal status |
[[Prescription drug|Template:Unicode-only]](US) |
| Routes | Oral (suspension or tablets) |
Mechanism of Action
- Bile acid sequestrants are polymeric compounds which serve as ion exchange resins. Bile acid sequestrants exchange anions such as chloride ions for bile acids. By doing so, they bind bile acids and sequester them from enterohepatic circulation.
- Since bile acid sequestrants are large polymeric structures, they are not well-absorbed from the gut into the bloodstream. Thus, bile acid sequestrants, along with any bile acids bound to the drug, are excreted via the feces after passage through the gastrointestinal tract.[9]
Structure
- The active ingredient in colestipol tablets is colestipol hydrochloride, which is a lipid lowering agent for oral use. Colestipol is an insoluble, high molecular weight basic anion-exchange copolymer of diethylenetriamine and 1-chloro-2, 3-epoxypropane, with approximately 1 out of 5 amine nitrogens protonated (chloride form). It is a light yellow water-insoluble resin which is hygroscopic and swells when suspended in water or aqueous fluids.
Pharmacodynamics
- FDA Package Insert for colestipol contains no information regarding pharmacodynamics.
Pharmacokinetics
- FDA Package Insert for colestipol contains no information regarding pharmacokinetics.
Nonclinical Toxicology
- In studies conducted in rats in which cholestyramine resin (a bile acid sequestering agent similar to colestipol hydrochloride) was used as a tool to investigate the role of various intestinal factors, such as fat, bile salts and microbial flora, in the development of intestinal tumors induced by potent carcinogens, the incidence of such tumors was observed to be greater in cholestyramine resin treated rats than in control rats.
- The relevance of this laboratory observation from studies in rats with cholestyramine resin to the clinical use of colestipol hydrochloride is not known. In the LRC-CPPT study referred to above, the total incidence of fatal and non-fatal neoplasms was similar in both treatment groups. When the many different categories of tumors are examined, various alimentary system cancers were somewhat more prevalent in the cholestyramine group. The small numbers and the multiple categories prevent conclusions from being drawn. Further follow-up of the LRC-CPPT participants by the sponsors of that study is planned for cause-specific mortality and cancer morbidity.
- When colestipol hydrochloride was administered in the diet to rats for 18 months, there was no evidence of any drug related intestinal tumor formation. In the Ames assay, colestipol hydrochloride was not mutagenic.
Clinical Studies
- FDA Package Insert for Colestipol contains no information regarding Clinical Studies.
How Supplied
Tablet
- Colestipol hydrochloride tablets, 1 gram of colestipol hydrochloride, are off-white to pale yellow, film-coated, oval tablets, debossed with "G" on one side and plain on the other.
- Bottles of 120 NDC 0115-5211-16
- Bottles of 500 NDC 0115-5211-02
Suspension
- Colestipol HCl for oral suspension USP is available as follows:
- Carton of 30 foil packets NDC 0115-5212-18
- Carton of 90 foil packets NDC 0115-5212-29
- Bottle of 500 grams with scoop NDC 0115-5213-02
Storage
- Store at 20°C to 25°C (68°F to 77°F)
Images
Drug Images
{{#ask: Page Name::Colestipol |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Colestipol |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Colestipol tablets may be larger than pills you have taken before. If you have had swallowing problems or choking with food, liquids or other tablets or capsules in the past, you should discuss this with your doctor before taking colestipol tablets.
- It is important that you take colestipol tablets correctly:
- Always take one tablet at a time and swallow promptly.
- Swallow each tablet whole. Do not cut, crush, or chew the tablets.
- Colestipol tablets must be taken with water or another liquid that you prefer. Swallowing the tablets will be easier if you drink plenty of liquid as you swallow each tablet.
- Difficulty swallowing and temporary obstruction of the esophagus (the tube between your mouth and stomach) have been rarely reported in patients taking colestipol tablets. If a tablet does get stuck after you swallow it, you may notice pressure or discomfort. If this happens to you, you should contact your doctor. Do not take colestipol tablets again without your doctor's advice.
- If you are taking other medications, you should take them at least one hour before or four hours after taking colestipol tablets.
Precautions with Alcohol
- Alcohol-Colestipol interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- COLESTID
Look-Alike Drug Names
There is limited information about the Look-Alike Drug Names.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Brown G, Albers JJ, Fisher LD, Schaefer SM, Lin JT, Kaplan C; et al. (1990). "Regression of coronary artery disease as a result of intensive lipid-lowering therapy in men with high levels of apolipoprotein B." N Engl J Med. 323 (19): 1289–98. doi:10.1056/NEJM199011083231901. PMID 2215615.
- ↑ Blankenhorn DH, Nessim SA, Johnson RL, Sanmarco ME, Azen SP, Cashin-Hemphill L (1987). "Beneficial effects of combined colestipol-niacin therapy on coronary atherosclerosis and coronary venous bypass grafts". JAMA. 257 (23): 3233–40. PMID 3295315.
- ↑ Nash DT, Gensini G, Esente P (1982). "Effect of lipid-lowering therapy on the progression of coronary atherosclerosis assessed by scheduled repetitive coronary arteriography". Int J Cardiol. 2 (1): 43–55. PMID 7129689.
- ↑ Tonstad S, Sivertsen M, Aksnes L, Ose L (1996). "Low dose colestipol in adolescents with familial hypercholesterolaemia". Arch Dis Child. 74 (2): 157–60. PMC 1511517. PMID 8660081.
- ↑ Groot PH, Dijkhuis-Stoffelsma R, Grose WF, Ambagtsheer JJ, Fernandes J (1983). "The effects of colestipol hydrochloride on serum lipoprotein lipid and apolipoprotein B and A-I concentrations in children heterozygous for familial hypercholesterolemia". Acta Paediatr Scand. 72 (1): 81–5. PMID 6407278.
- ↑ Schlierf G, Mrozik K, Heuck CC, Middelhoff G, Oster P, Riesen W; et al. (1982). ""Low dose" colestipol in children, adolescents and young adults with familial hypercholesterolemia". Atherosclerosis. 41 (1): 133–8. PMID 7073790.
- ↑ Mordasini R, Twelsiek F, Oster P, Schellenberg B, Raetzer H, Heuck CC; et al. (1978). "Abnormal low density lipoproteins in children with familial hypercholesterolemia--effect of polyanion exchange resins". Klin Wochenschr. 56 (16): 805–8. PMID 211316.
- ↑ McCrindle BW, Helden E, Cullen-Dean G, Conner WT (2002). "A randomized crossover trial of combination pharmacologic therapy in children with familial hyperlipidemia". Pediatr Res. 51 (6): 715–21. doi:10.1203/00006450-200206000-00009. PMID 12032266.
- ↑ FDA Heart Health Online - Bile Acid Sequestrants
{{#subobject:
|Page Name=Colestipol |Pill Name=No_image.jpg |Drug Name=Colestipol |Pill Ingred=cellulose, microcrystalline, copovidone, silicon dioxide, polyethylene glycol 6000, hypromellose 2910 (6 mpa.s), cellacefate, dibutyl sebacate|+sep=; |Pill Imprint=G |Pill Dosage=1 g |Pill Color=Yellow|+sep=; |Pill Shape=Oval |Pill Size (mm)=21.00 |Pill Scoring=1 |Pill Image= |Drug Author=Global Pharmaceuticals, Division of Impax Laboratories Inc. |NDC=0115-5211
}}
{{#subobject:
|Label Page=Colestipol |Label Name=Colestipol_label_01.jpg
}}
{{#subobject:
|Label Page=Colestipol |Label Name=Colestipol_label_02.jpg
}}
{{#subobject:
|Label Page=Colestipol |Label Name=Colestipol_label_03.jpg
}}
{{#subobject:
|Label Page=Colestipol |Label Name=Colestipol_panel_01.png
}}
{{#subobject:
|Label Page=Colestipol |Label Name=Colestipol_panel_02.png
}}
{{#subobject:
|Label Page=Colestipol |Label Name=Colestipol_panel_03.png
}}
{{#subobject:
|Label Page=Colestipol |Label Name=Colestipol_panel_04.png
}}
{{#subobject:
|Label Page=Colestipol |Label Name=Colestipol_panel_05.png
}}