Bohr effect
|
WikiDoc Resources for Bohr effect |
|
Articles |
|---|
|
Most recent articles on Bohr effect Most cited articles on Bohr effect |
|
Media |
|
Powerpoint slides on Bohr effect |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Bohr effect at Clinical Trials.gov Clinical Trials on Bohr effect at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Bohr effect
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Bohr effect Discussion groups on Bohr effect Patient Handouts on Bohr effect Directions to Hospitals Treating Bohr effect Risk calculators and risk factors for Bohr effect
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Bohr effect |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
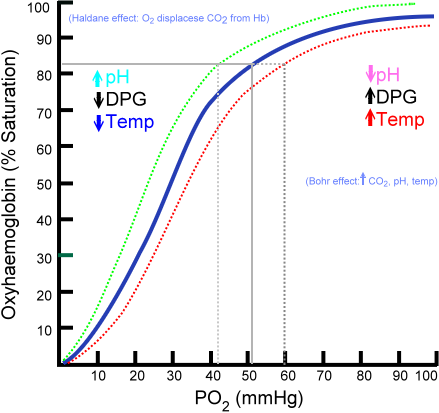
The Bohr effect is a property of hemoglobin first described by the Danish physiologist Christian Bohr in 1904, and often erroneously attributed to his son, physicist Niels Bohr, which states that in the presence of carbon dioxide, the oxygen affinity for dissociation of respiratory pigments, such as hemoglobin decreases; because of the Bohr effect, an increase in blood carbon dioxide level or a decrease in pH causes hemoglobin to bind to oxygen with less affinity.
This effect facilitates oxygen transport as hemoglobin binds to oxygen in the lungs, but then releases it in the tissues, particularly those tissues in most need of oxygen. When a tissue's metabolic rate increases, its carbon dioxide production increases. The carbon dioxide is quickly converted into bicarbonate molecules and acidic protons by the enzyme carbonic anhydrase:
- CO2+ H2O <math>\rightleftharpoons</math> H+ + HCO3−
This causes the pH of the tissue to decrease, and so increases the dissociation of oxygen from hemoglobin, allowing the tissue to obtain enough oxygen to meet its demands.
The dissociation curve shifts to the right when carbon dioxide or hydrogen ion concentration is increased. This facilitates increased oxygen dumping. This makes sense because increased CO2 concentration and lactic acid build-up occur when the muscles need more oxygen. Changing hemoglobin's oxygen affinity is the body's way of adapting quickly to this problem.
In the Hiroshima variant hemoglobinopathy the Bohr effect is diminished so the hemoglobin has a higher affinity for oxygen and tissue may suffer minor oxygen starvation during high work.
See also
External links
de:Bohr-Effekt hr:Bohrov efekt it:Effetto Bohr he:אפקט בוהר nl:Bohr-effect