Alendronate detailed information
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | oral tablets |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 0.6% |
| Metabolism | excreted unchanged |
| Elimination half-life | 126 months |
| Excretion | renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
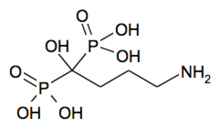
| Formula | C4H18NNaO10P2 |
| Molar mass | 325.124 |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Alendronate (Fosamax, Merck) is a bisphosphonate drug used for osteoporosis and several other bone diseases. It is marketed alone as well as in combination with vitamin D (2,800 U and 5600 U, under the name Fosamax+D).
Pharmacokinetics
As with all potent bisphosphonates, the systemic bioavailability after oral dosing is low, averaging only 0.6 - 0.7 % in women and in men under fasting conditions. Intake together with meals and beverages other than water further reduces the bioavailability. The absorbed drug rapidly partitions, with approximately 50% binding to the exposed bone surface; the remainder is excreted unchanged by the kidneys. Unlike most drugs, the strong negative charge on the two phosphate moieties limits oral bioavailability, and in turn, the exposure to tissues other than bone is very low. After absorption in the bone, alendronate has an estimated terminal half-life of 10 years.}
Pharmacology
Alendronate inhibits osteoclast-mediated bone-resorption. Like all bisphosphonates it is chemically related to inorganic pyrophosphate, the endogenous regulator of bone turnover. Whereas pyrophosphate and the first bisphosphonate, etidronate, are capable of inhibiting both osteoclastic bone resorption as well as the mineralization of the bone newly formed by osteoblasts, alendronate and the other potent N-containing bisphosphonates such as risedronate and ibandronate and zoledronate specifically inhibit bone resorption without any effect on mineralization at pharmacalogically achievable doses. Its inhibition of bone-resorption is dose-dependent and approximately 1,000 times stronger than the equimolar effect of etidronate. Under therapy normal bone tissue develops, and alendronate is deposited in the bone-matrix in pharmacologically inactive form. For optimal action enough calcium and vitamin D are needed in the body in order to promote normal bone development. Hypocalcemia should therefore be corrected before starting therapy.
Clinical Data
Treatment of post-menopausal women with Fosamax has demonstrated normalization of the rate of bone turnover, significant increase in BMD (bone mineral density) of the spine, hip, wrist and total body, and significant reductions in the risk of vertebral (spine) fractures, wrist fractures, hip fractures, and all non-vertebral fractures. In the women with the highest risk of fracture (by virtue of pre-existing vertebral fractures) in the Fracture Intervention Trial, treatment with Fosamax 5 mg/day for two years followed by 10 mg/day for the third year (plus calcium and vitamin D) resulted in approximately 50% reductions in fractures of the spine, hip and wrist compared to the control group taking placebos plus calcium and vitamin D.
Uses
- Prophylaxis and treatment of female osteoporosis
- Treatment of male osteoporosis
- Prevention and treatment of corticosteroid-associated osteoporosis together with supplements of calcium and vitamin D
- Paget's disease
Contraindications and precautions
- Acute inflammations of the gastrointestinal tract (esophagitis, gastritis, ulcerations)
- Clinically manifest osteomalacia
- Certain malformations and malfunctions of the esophagus (strictures, achalasia)
- Inability to stand, walk, or sit for 30 minutes after oral administration
- Renal impairment with a creatinine clearance below 30ml/min
- Hypersensitivity to alendronate or another ingredient
- Hypocalcemia
- Pregnancy and breastfeeding
- Patients below 18 yrs. of age, because no clinical data exists
Side-effects
- GI tract: A severe side effect is an ulceration of the esophagus caused by alendronate, which may require hospitalization and intensive treatment. Gastric and duodenal ulceration.
- General: infrequent cases of skin rash, rarely manifesting as Stevens-Johnson syndrome and toxic epidermal necrolysis, eye problems (uveitis, scleritis) and generalized muscle, joint, and bone pain (rarely severe) have been seen. In laboratory tests decreased calcium and phosphate values may be obtained but reflect action of the drug and are harmless.
- Cases of osteonecrosis of the jaw have been reported in the medical literature, but relationship to alendronate is unknown.
- Osteonecrosis of the Jaw- Deterioration of the TM Joint can also result while on this drug if dental work of any kind 'needs' to be done. Although not common, this may result when patients of on the intravenous alendronate and most cases have been reported in cancer patients.
- Rare instances of auditory hallucinations and visual disturbances have been associated with alendronate and other bisphosphonates.[2]
Interactions
- Milk, diet and drugs containing high amounts of calcium, magnesium or aluminium (antacids): the resorption of alendronate is decreased. At least half an hour should pass after intake of alendronate before taking the supplement or drug.
- Highly active vitamin D analogues or fluorides: no data is available. Concomitant treatment should be avoided.
- The additional beneficial effect of HRT (hormone replacement therapy) with estrogens/progestins or raloxifene in postmenopausal women remains to be elucidated, but no interactions have been seen. The combination is therefore possible.
- Intravenous ranitidine increases the oral bioavailability of alendronate. No clinical consequences are known.
- The combination of NSAIDs and alendroate may increase the risk of gastric ulcers. Both these drugs have the potential to irritate the upper gastro-intestinal mucosa.
Dosage
- Prophylaxis of osteoporosis in women: 5-10mg daily or 35-70mg weekly.
- Therapy of osteoporosis in women and men : 10mg daily or 70mg weekly.
- Osteoporosis under corticosteroids: 5-10mg daily or 35-70 mg weekly in men and premenopausal women or those receiving concomitant HRT. In postmenopausal women not receiving HRT the recommended dose is 10mg daily or 70mg weekly.
- Paget's Disease: 40mg daily for 6 months.
The drug is to be taken only upon rising for the day with three swallows of water, not to exceed 6-8 oz. Stand, walk or sit and remain fasting for 30-45 minutes afterwards, then eat breakfast. Lying down or reclining prior to eating breakfast may cause gastroesophageal reflux and esophageal irritation. At least 30 minutes should be waited before meals or other beverages than water are taken in.
Dosage forms
- Fosamax® solution 70mg/75ml
- Fosamax® tablets 5mg, 10mg, 35mg, 40mg, and 70mg
Patent Remarks (USA)
Its patent is set to expire in 2008 and Merck has lost a series of appeals to block a generic version of the drug from being certified by the US FDA.
Bis-phossy Jaw
The term given by scientists to the link between bisphosphonates and jaw necrosis is 'bis-phossy jaw.' This is derived from the 19th-century term phossy jaw, given its name after workers in match factories working with white phosphorus developed osteonecrosis of the jaw.
External links
- Fosamax Information, Side Effects, ONJ etc...
- Fosamax information from Merck
- RxList page
- MedlinePlus listing
- Fosamax Side Effects
- Pharmacotherapy 24(6):799-802, 2004
- Pages with script errors
- Drugs with non-standard legal status
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- Bisphosphonates
- Merck
- Endocrinology