Acute kidney injury pathophysiology
| https://https://www.youtube.com/watch?v=vnTR_y3Sf-k%7C350}} |
|
Acute kidney injury Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Acute kidney injury pathophysiology On the Web |
|
American Roentgen Ray Society Images of Acute kidney injury pathophysiology |
|
Risk calculators and risk factors for Acute kidney injury pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Farima Kahe M.D. [2]
Overview
Acute kidney injury is defined as spontaneous deficit in kidney functions leading to urea retention and electrolyte imbalance. Etiologies of AKI can be divided based on pathophysiologic mechanisms into 3 broad categories: prerenal, intrinsic renal, and postrenal causes. Pre-renal AKI is most common and typically results from hypovolemia. Intrinsic renal is due to damage to renal paranchyma. Post-renal AKI is usually result of an obstruction, may be due to stones or strictures.
Pathophysiology
Physiology
Etiologies of AKI can be divided based on pathophysiologic mechanisms into 3 broad categories: prerenal, intrinsic renal, and postrenal causes.

Prerenal AKI
- Prerenal AKI, known as prerenal azotemia, is by far the most common cause of AKI representing 30-50% of all cases.
- It is provoked by inadequate renal blood flow commonly due to decreased effective circulating blood flow.
- This causes a decrease in the intraglomerular hydrostatic pressure required to achieve proper glomerular filtration.
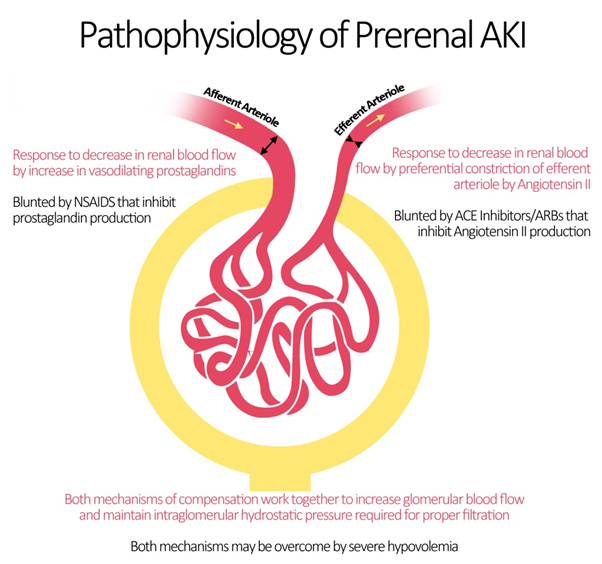
- Blood flow to the kidneys can vary with systemic changes; however, glomerular perfusion pressure and GFR are maintained relatively constant by the kidney itself.
- Under physiologic conditions, minor drops in blood flow to the renal circulation are counteracted by changes in the resistances across the afferent and efferent arterioles of individual glomerular capillary beds.[1]
- The afferent arteriole vasodilates via 2 mechanisms.[2]
- The myogenic reflex, mediating medial smooth muscle relaxation in states of decrease perfusion pressure, vasodilates the afferent arteriole leading to increased blood flow.[3]
- Additionally, intrarenal synthesis of vasodilatory prostaglandins such as prostacyclin and prostaglandin E2 causes further dilation of the afferent arteriole.[4]
- The mechanism explains why the intake of NSAIDs leads to acute kidney injury by inhibiting this autoregulatory mechanism.[5]
- At the level of the efferent arteriole, an increase in resistance is crucial for appropriate maintenance of glomerular hydrostatic pressure.
- This is achieved by an increase in the production of angiotensin II (via the renin-angiotensin-aldosterone system) which acts preferentially on the efferent arteriole leading to vasoconstriction.[6]
- Important medications that target angiotensin II production and action are ACE inhibitors (ACEIs) and angiotensin receptor blockers (ARBs) which may be responsible for renal decompensation in patients dependent on the action of angiotensin II to maintain glomerular perfusion pressure. Such is the case in chronic kidney disease patients, whose autoregulatory mechanisms are typically operating at maximum capacity.[7]
- As such, the pathophysiology of prerenal azotemia entails a drop in renal plasma flow beyond the capacity of autoregulation, a blunted or inadequate renal compensation for an otherwise tolerable change in perfusion, or a combination of both.
- This eventually leads to ischemic renal injury particularly to the medulla which is maintained in hypoxic conditions at baseline.
- Causes of prerenal injury are summarized in the figure below. To note, as prerenal AKI progresses with further ischemia, it transforms into acute tubular necrosis (ATN) crossing into the realm of intrinsic AKI.

Intrinsic Renal AKI
Intrinsic renal AKI generally occurs due to renal parenchymal injury and may be classified according to the site of injury into: glomerular, tubular, interstitial, and vascular.
Tubular AKI
- The most common form of intrinsic renal AKI involves damage to the renal tubules.
- In this context, the most common etiologies are sepsis, nephrotoxins, and ischemia.
- Ischemic AKI is part of a disease continuum involving prerenal AKI and manifests in states of prolonged renal blood flow compromise or renal hypoperfusion with other pre-existing or concomitant renal insults.
- Although sometimes dubbed as acute tubular necrosis (ATN), ATN is non-specific to prerenal disease, and may be induced by sepsis and nephrotoxins.
- ATN is also not a very accurate pathological term, as renal biopsies have rarely shown true tubular necrosis, but rather tubular cell injury & apoptosis with secondary dysfunction are more accurate.
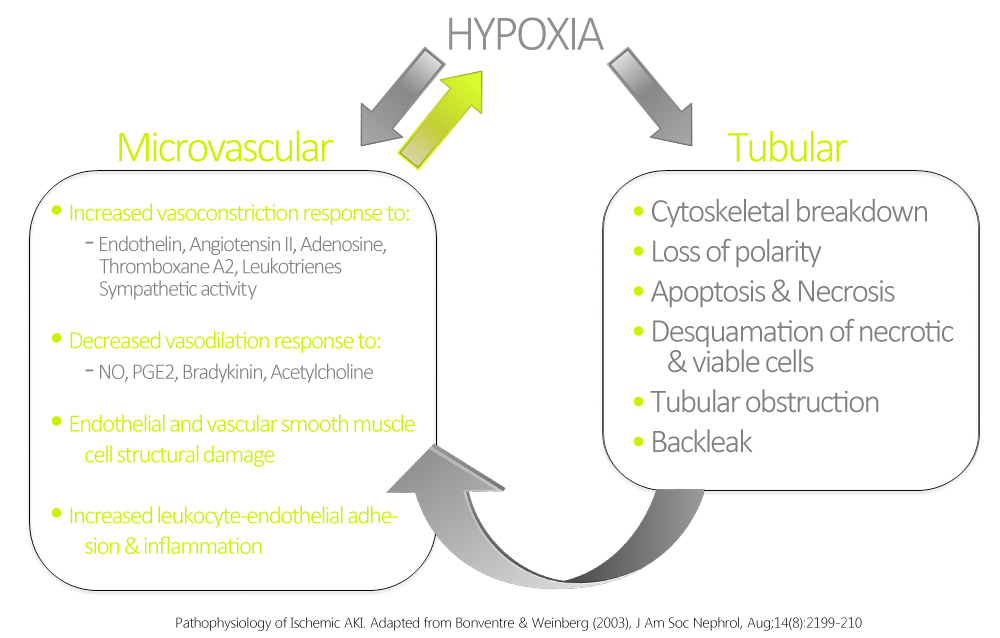
- These pathological manifestations are related to hypoxia and ATP depletion in areas that are physiologically hypoxic such as the renal medulla, and areas that are very metabolically active such as the proximal tubule.
- The response of the renal tubules and the microvasculature are maladaptive leading to a paradoxical increase in hypoxia and further damage and inflammation.[8]
- Ischemia and hypoxia are known to cause increased reactivity to vasoconstrictive agents, and decreased vasodilatory responses in arterioles as compared to normal kidneys.[9]
Sepsis
- AKI is seen in 20 to 25% of cases of sepsis and in 50% of cases of septic shock.
- A decrease in GFR in a septic patient is usually a marker of poor prognosis, and the combination of sepsis and AKI is associated with a mortality rate of 70%.
- Although most cases of AKI occur with severe hemodynamic compromise in septic patients, renal injury may occur without overt hypotension.
- While there is clear tubular damage in sepsis-associated AKI, interstitial inflammation and interstitial edema have also been proposed in the pathogenesis.[10][11]
- The mechanisms of alteration of renal hemodynamics proposed in sepsis include excessive efferent arteriolar vasodilation or generalized renal vasoconstriction secondary to tumor necrosis factor induced release of endothelin.
Nephrotoxins
- Another major cause of intrinsic renal AKI is nephrotoxins.
- These may be either endogenous such as myoglobin, hemoglobin, and myeloma light chains, or exogenous such as contrast agents, antibiotics, and chemotherapeutic agents.
- The kidney is a particularly susceptible organ to toxin injury mainly due to the high blood perfusion and the high concentration of substances in the kidneys destined for excretion.
- Nephrotoxic injury may be secondary to tubular, interstitial, or microvascular damage depending on the nephrotoxin itself.
- Major risk factors for nephrotoxic AKI include old age, pre-existing chronic kidney disease (CKD), and prerenal azotemia.[12]
Contrast-induced Nephropathy
- Contrast induced nephropathy (CIN) recently called contrast induced AKI (CIAKI) is also major cause of intrinsic injury caused by iodinated contrast media used in cardiovascular imaging.
- This entity is virtually non-existent in healthy young individuals.
- Risk factors that increase susceptibility to CIN include advanced age, pre-existing CKD, diabetic nephropathy, severe heart failure, and concomitant exposure to other nephrotoxins.
- The pathophysiology of CIN is not clearly understood; however, several attempts have been made to explain the underlying mechanism.
- It is generally agreed that CIN is due to a combination of several influences brought on by contrast-media infusion rather than a single process.
- The most important mechanism thought to be involved in CIN is a reduction in renal perfusion at the level of the microvasculature leading to tubular damage.
- This is attributed to several alterations in the renal microenvironment including activation of the tubuloglomerular feeback, local vasoactive metabolites including adenosine, prostaglandin, NO, and endothelin as well as increased interstitial pressure.
- Studies have also proposed injury to renal tubular cells may occur via a direct cytotoxic effect of the contrast media and via reactive oxygen species production.[13]

Glomerular AKI
- Glomerular damage causing AKI accounts for a small propotion of cases of AKI.
- Glomerulonephritis leading to AKI is usually seen in rapidly progressive glomerulonephritis (RPGN).
- Other forms of glomerulonephritis progress slowly and generally lead to chronic kidney disease.
- RPGN is characterized by a triad of hematuria, proteinuria, and hypertension progressing to a decrease in GFR and urine output.[14]
- RPGN can be idiopathic or secondary to SLE, Henoch-Schonlein purpura, Wegener’s granulomatosis, and Goodpasture’s syndrome.
- The pathophysiology is almost always related to an autoimmune insult, but specific characteristics depend on the underlying etiologies.[15]
Vascular AKI
- Other causes of AKI of vascular origin include diseases affecting the macro and microvasculature not only confined to the glomerular capillaries.
- Examples include TTP/HUS and DIC associated with microangiopathic hemolytic anemia (MAHA) typically arising from an endothelial cell injury with subsequent leukocyte adhesion, complement consumption, platelet aggregation and eventual ischemic damage.
- Other causes include atheroemboli, calcineurin inhibitors in renal transplant patients via vasoconstriction of the afferent arterioles (although a tubulointerstitial pattern is also seen), and vasculitides.[16][17]
Interstitial AKI
- AKI may be secondary to acute interstitial nephritis caused by an idiosyncratic immune-mediated mechanism.
- Classically, it is associated with a number of medications including penicillins (classically methicillin), cephalosporins, fluoroquinolones, NSAIDs, thiazide and loop diuretics, and allopurinol.[18]
- AIN can also be secondary to an infectious process, or systemic syndromes such as cryoglobulinemia, Sjogren syndrome, sarcoidosis, and primary biliary cirrhosis.
- Clinically, it may be associated with fever, and urinary eosinophilia although it may often be asymptomatic.
- Pathophysiology involves a cell-mediated immune reaction with interstitial infiltrates mostly composed of lymphocytes, macrophages, eosinophils, and plasma cells, with subsequent transformation into areas of interstitial fibrosis.[17]
Postrenal AKI
- Postrenal AKI occurs due to an obstruction in the urinary flow leading to an increase in the intratubular hydrostatic pressure which interferes with proper glomerular filtration.[19]
- Obstructions occurring at the level of the renal pelvis and the ureters must affect both kidneys simultaneously to cause AKI in healthy adults unless only one kidney is functional.
- Causes of upper tract obstructions may be intraluminal such as calculi or blood clots, transmural secondary to neoplastic invasion, or extrinsic compression by retroperitoneal fibrosis, neoplasia, or an abscess.
- The most common cause of postrenal AKI is bladder neck obstruction secondary to benign prostatic hypertrophy and prostate cancer.
- Other etiologies of lower urinary tract obstruction are calculi, and strictures. Patients usually have evident hydronephrosis unless early in the course of obstruction.
Genetics
There is no genetics associated with AKI.
Associated Conditions
Gross Pathology
- On gross pathology, characteristic findings for AKI are not present.
Microscopic Pathology
- On microscopic histopathological analysis, characteristic findings of AKI depends on the etiology of disease.
References
- ↑ Loutzenhiser R, Griffin K, Williamson G, Bidani A (2006). "Renal autoregulation: new perspectives regarding the protective and regulatory roles of the underlying mechanisms". Am J Physiol Regul Integr Comp Physiol. 290 (5): R1153–67. doi:10.1152/ajpregu.00402.2005. PMC 1578723. PMID 16603656.
- ↑ Badr KF, Ichikawa I (1988). "Prerenal failure: a deleterious shift from renal compensation to decompensation". N Engl J Med. 319 (10): 623–9. doi:10.1056/NEJM198809083191007. PMID 3045546.
- ↑ Cupples WA, Braam B (2007). "Assessment of renal autoregulation". Am J Physiol Renal Physiol. 292 (4): F1105–23. doi:10.1152/ajprenal.00194.2006. PMID 17229679.
- ↑ Herbaczynska-Cedro K, Vane JR (1973). "Contribution of intrarenal generation of prostaglandin to autoregulation of renal blood flow in the dog". Circ Res. 33 (4): 428–36. PMID 4355037.
- ↑ Winkelmayer WC, Waikar SS, Mogun H, Solomon DH (2008). "Nonselective and cyclooxygenase-2-selective NSAIDs and acute kidney injury". Am J Med. 121 (12): 1092–8. doi:10.1016/j.amjmed.2008.06.035. PMID 19028206.
- ↑ Arendshorst WJ, Brännström K, Ruan X (1999). "Actions of angiotensin II on the renal microvasculature". J Am Soc Nephrol. 10 Suppl 11: S149–61. PMID 9892156.
- ↑ Abuelo JG (2007). "Normotensive ischemic acute renal failure". N Engl J Med. 357 (8): 797–805. doi:10.1056/NEJMra064398. PMID 17715412.
- ↑ Bonventre JV, Weinberg JM (2003). "Recent advances in the pathophysiology of ischemic acute renal failure". J Am Soc Nephrol. 14 (8): 2199–210. PMID 12874476.
- ↑ Conger JD, Weil JV (1995). "Abnormal vascular function following ischemia-reperfusion injury". J Investig Med. 43 (5): 431–42. PMID 8528754.
- ↑ Devarajan P (2006). "Update on mechanisms of ischemic acute kidney injury". J Am Soc Nephrol. 17 (6): 1503–20. doi:10.1681/ASN.2006010017. PMID 16707563.
- ↑ Bonventre JV (2010). "Pathophysiology of AKI: injury and normal and abnormal repair". Contrib Nephrol. 165: 9–17. doi:10.1159/000313738. PMID 20427950.
- ↑ Choudhury D, Ahmed Z (2006). "Drug-associated renal dysfunction and injury". Nat Clin Pract Nephrol. 2 (2): 80–91. doi:10.1038/ncpneph0076. PMID 16932399.
- ↑ Wong PC, Li Z, Guo J, Zhang A (2012). "Pathophysiology of contrast-induced nephropathy". Int J Cardiol. 158 (2): 186–92. doi:10.1016/j.ijcard.2011.06.115. PMID 21784541.
- ↑ Erwig LP, Rees AJ (1999). "Rapidly progressive glomerulonephritis". J Nephrol. 12 Suppl 2: S111–9. PMID 10688410.
- ↑ Chen YX, Chen N (2013). "Pathogenesis of rapidly progressive glomerulonephritis: what do we learn?". Contrib Nephrol. 181: 207–15. doi:10.1159/000348633. PMID 23689582.
- ↑ Naesens M, Kuypers DR, Sarwal M (2009). "Calcineurin inhibitor nephrotoxicity". Clin J Am Soc Nephrol. 4 (2): 481–508. doi:10.2215/CJN.04800908. PMID 19218475.
- ↑ 17.0 17.1 Ruggenenti P, Noris M, Remuzzi G (2001). "Thrombotic microangiopathy, hemolytic uremic syndrome, and thrombotic thrombocytopenic purpura". Kidney Int. 60 (3): 831–46. doi:10.1046/j.1523-1755.2001.060003831.x. PMID 11532079.
- ↑ Michel D and Kelly C. Acute Interstitial Nephritis. JASN 1998; 9(3): 506-515.
- ↑ Patel TV, Kumar S, Singh AK (2007). "Post-renal acute renal failure". Kidney Int. 72 (7): 890–4. doi:10.1038/sj.ki.5002301. PMID 17495862.