Ibuprofen lysine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ammu Susheela, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Ibuprofen lysine is a non-steroidal anti-inflammatory agents that is FDA approved for the treatment of patent ductus arteriosus (PDA) in premature infants weighing between 500 and 1500 g, who are no more than 32 weeks gestational age. Common adverse reactions include gastrointestinal perforation and necrotizing enterocolitis.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
There is limited information regarding FDA-Label Guideline-Supported Use of Ibuprofen lysine in adult patients.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ibuprofen lysine in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Ibuprofen lysine in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Dosage
- Ibuprofen lysine is indicated to close a clinically significant patent ductus arteriosus (PDA) in premature infants weighing between 500 and 1500 g, who are no more than 32 weeks gestational age when usual medical management (e.g., fluid restriction, diuretics, respiratory support, etc.) is ineffective. The clinical trial was conducted among infants with an asymptomatic PDA. However, the consequences beyond 8 weeks after treatment have not been evaluated; therefore, treatment should be reserved for infants with clear evidence of a clinically significant PDA.
Dosage
- A course of therapy is three doses of ibuprofen lysine administered intravenously (administration via an umbilical arterial line has not been evaluated). An initial dose of 10 mg per kilogram is followed by two doses of 5 mg per kilogram each, after 24 and 48 hours. All doses should be based on birth weight. If anuria or marked oliguria (urinary output <0.6 mL/kg/hr) is evident at the scheduled time of the second or third dose of ibuprofen lysine, no additional dosage should be given until laboratory studies indicate that renal function has returned to normal.
- If the ductus arteriosus closes or is significantly reduced in size after completion of the first course of ibuprofen lysine, no further doses are necessary.
- If during continued medical management the ductus arteriosus fails to close or reopens, then a second course of ibuprofen lysine, alternative pharmacological therapy, or surgery may be necessary.
Directions for Use
- For intravenous administration only. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever solution and container permit. Do not use ibuprofen lysine if particulate matter is observed.
- After the first withdrawal from the vial, any solution remaining must be discarded because ibuprofen lysine contains no preservative.
- For administration, ibuprofen lysine should be diluted to an appropriate volume with dextrose or saline. Ibuprofen lysine should be prepared for infusion and administered within 30 minutes of preparation and infused continuously over a period of 15 minutes. The drug should be administered via the IV port that is nearest the insertion site. After the first withdrawal from the vial, any solution remaining must be discarded because ibuprofen lysine contains no preservative.
- Since ibuprofen lysine is potentially irritating to tissues, it should be administered carefully to avoid extravasation.
- Ibuprofen lysine should not be simultaneously administered in the same intravenous line with Total Parenteral Nutrition (TPN). If necessary, TPN should be interrupted for a 15-minute period prior to and after drug administration. Line patency should be maintained by using dextrose or saline.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ibuprofen lysine in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Ibuprofen lysine in pediatric patients.
Contraindications
- Ibuprofen lysine is contraindicated in:
- Preterm infants with proven or suspected infection that is untreated;
- Preterm infants with congenital heart disease in whom patency of the PDA is necessary for satisfactory pulmonary or systemic blood flow (e.g., pulmonary atresia, severe tetralogy of Fallot, severe coarctation of the aorta);
- Preterm infants who are bleeding, especially those with active intracranial hemorrhage or gastrointestinal bleeding;
- Preterm infants with thrombocytopenia;
- Preterm infants with coagulation defects;
- Preterm infants with or who are suspected of having necrotizing enterocolitis;
- Preterm infants with significant impairment of renal function.
Warnings
General
- There are no long-term evaluations of the infants treated with ibuprofen at durations greater than the 36 weeks post-conceptual age observation period.
- Ibuprofen’s effects on neurodevelopmental outcome and growth as well as disease processes associated with prematurity (such as retinopathy of prematurity and chronic lung disease) have not been assessed.
Infection
- Ibuprofen lysine may alter the usual signs of infection. The physician must be continually on the alert and should use the drug with extra care in the presence of controlled infection and in infants at risk of infection.
Platelet Aggregation
- Ibuprofen lysine, like other non-steroidal anti-inflammatory agents, can inhibit platelet aggregation. Preterm infants should be observed for signs of bleeding. Ibuprofen has been shown to prolong bleeding time (but within the normal range) in normal adult subjects. This effect may be exaggerated in patients with underlying hemostatic defects.
Bilirubin Displacement
- Ibuprofen has been shown to displace bilirubin from albumin binding-sites; therefore, it should be used with caution in patients with elevated total bilirubin.
Administration
- Ibuprofen lysine should be administered carefully to avoid extravascular injection or leakage, as solution may be irritating to tissue.
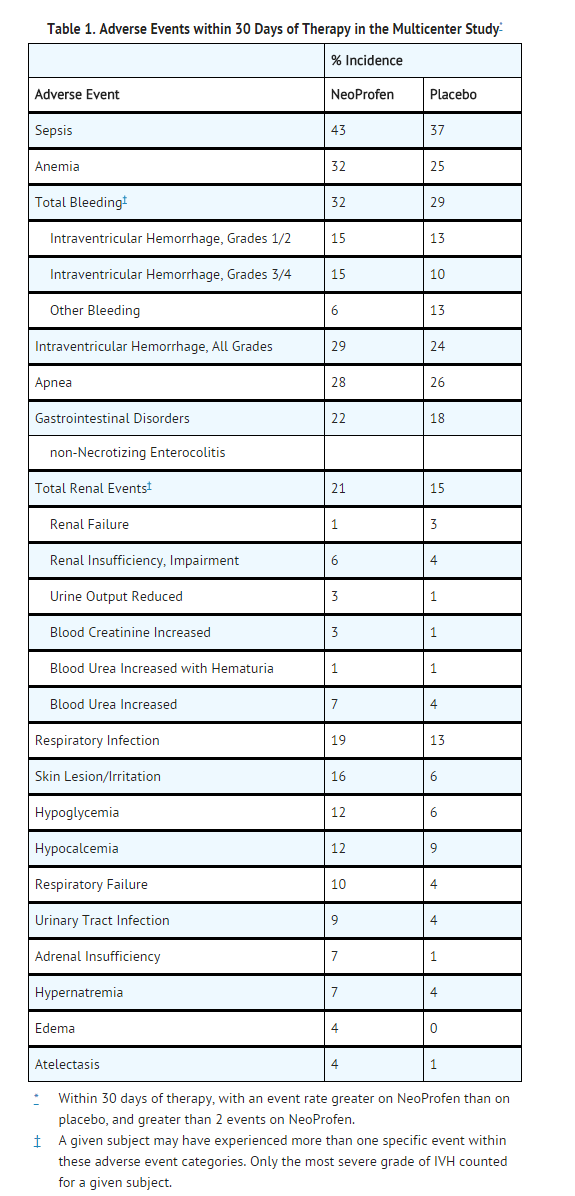
Adverse Reactions
Clinical Trials Experience
Renal Function
- Compared to placebo, there was a small decrease in urinary output in the ibuprofen group on days 2-6 of life, with a compensatory increase in urine output on day 9. In other studies, adverse events classified as renal insufficiency including oliguria, elevated BUN, elevated creatinine, or renal failure were reported in ibuprofen treated infants.
Additional Adverse Events
- The adverse events reported in the multicenter study and of unknown association include tachycardia, cardiac failure, abdominal distension, gastroesophageal reflux, gastritis, ileus, inguinal hernia, injection site reactions, cholestasis, various infections, feeding problems, convulsions, jaundice, hypotension, and various laboratory abnormalities including neutropenia, thrombocytopenia, and hyperglycemia.
Postmarketing Experience
- The following adverse reactions have been identified from spontaneous post-marketing reports or published literature: gastrointestinal perforation and necrotizing enterocolitis. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency, or establish a causal relationship to drug exposure.
Drug Interactions
- Drug interactions of ibuprofen lysine in neonates have not been assessed.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
There is no FDA guidance on usage of Ibuprofen lysine in women who are pregnant.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Ibuprofen lysine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Ibuprofen lysine during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Ibuprofen lysine in women who are nursing.
Pediatric Use
There is no FDA guidance on the use of Ibuprofen lysine in pediatric settings.
Geriatic Use
There is no FDA guidance on the use of Ibuprofen lysine in geriatric settings.
Gender
There is no FDA guidance on the use of Ibuprofen lysine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Ibuprofen lysine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Ibuprofen lysine in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Ibuprofen lysine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Ibuprofen lysine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Ibuprofen lysine in patients who are immunocompromised.
Administration and Monitoring
Administration
Monitoring
There is limited information regarding Ibuprofen lysine Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Ibuprofen lysine and IV administrations.
Overdosage
- The following signs and symptoms have occurred in individuals (not necessarily in premature infants) following an overdose of oral ibuprofen: breathing difficulties, coma, drowsiness, irregular heartbeat, kidney failure, low blood pressure, seizures, and vomiting. There are no specific measures to treat acute overdosage with ibuprofen lysine. The patient should be followed for several days because gastrointestinal ulceration and hemorrhage may occur.
Pharmacology
There is limited information regarding Ibuprofen lysine Pharmacology in the drug label.
Mechanism of Action
- The mechanism of action through which ibuprofen causes closure of a patent ductus arteriosus (PDA) in neonates is not known. In adults, ibuprofen is an inhibitor of prostaglandin synthesis.
Structure
- Ibuprofen lysine® is a clear sterile preservative-free solution of the L-lysine salt of (±)-ibuprofen which is the active ingredient. (±)-Ibuprofen is a nonsteroidal anti-inflammatory agent (NSAID). L-lysine is used to create a water-soluble drug product salt suitable for intravenous administration. Each mL of ibuprofen lysine contains 17.1 mg of ibuprofen lysine (equivalent to 10 mg of (±)-ibuprofen) in Water for Injection, USP. The pH is adjusted to 7.0 with sodium hydroxide or hydrochloric acid.
- The structural formula is:
- Ibuprofen lysine is designated chemically as α-methyl-4-(2-methyl propyl) benzeneacetic acid lysine salt. Its molecular weight is 352.48. Its empirical formula is C19H32N2O4. It occurs as a white crystalline solid which is soluble in water and slightly soluble in ethanol.
Pharmacodynamics
There is limited information regarding Ibuprofen lysine Pharmacodynamics in the drug label.
Pharmacokinetics
- The pharmacokinetic data were obtained from 54 ibuprofen lysine-treated premature infants included in a double-blind, placebo-controlled, randomized, multicenter study. Infants were less than 30 weeks gestational age, weighed between 500 and 1000 g, and exhibited asymptomatic PDA with evidence of echocardiographic documentation of ductal shunting. Dosing was initially 10 mg/kg followed by 5 mg/kg at 24 and 48 hours.
- The population average clearance and volume of distribution values of racemic ibuprofen for premature infants at birth were 3 mL/kg/h and 320 mL/kg, respectively. Clearance increased rapidly with post-natal age (an average increase of approximately 0.5 mL/kg/h per day). Inter-individual variability in clearance and volume of distribution were 55% and 14%, respectively. In general, the half-life in infants is more than 10 times longer than in adults.
- The metabolism and excretion of ibuprofen in premature infants have not been studied.
- In adults, renal elimination of unchanged ibuprofen accounts for only 10-15% of the dose. The excretion of ibuprofen and metabolites occurs rapidly in both urine and feces. Approximately 80% of the dose administered orally is recovered in urine as hydroxyl and carboxyl metabolites, respectively, as a mixture of conjugated and unconjugated forms. Ibuprofen is eliminated primarily by metabolism in the liver where CYP2C9 mediates the 2- and 3-hydroxylations of R- and S-ibuprofen. Ibuprofen and its metabolites are further conjugated to acyl glucuronides.
- In neonates, renal function and the enzymes associated with drug metabolism are underdeveloped at birth and substantially increase in the days after birth.
Nonclinical Toxicology
There is limited information regarding Ibuprofen lysine Nonclinical Toxicology in the drug label.
Clinical Studies
- In a double-blind, multicenter clinical study premature infants of birth weight between 500 and 1000 g, less than 30 weeks post-conceptional age, and with echocardiographic evidence of a PDA were randomized to placebo or ibuprofen lysine. These infants were asymptomatic from their PDA at the time of enrollment. The primary efficacy parameter was the need for rescue therapy (indomethacin, open-label ibuprofen, or surgery) to treat a hemodynamically significant PDA by study day 14. An infant was rescued if there was clinical evidence of a hemodynamically significant PDA that was echocardiographically confirmed. A hemodynamically significant PDA was defined by three of the following five criteria ― bounding pulse, hyperdynamic precordium, pulmonary edema, increased cardiac silhouette, or systolic murmur ― or hemodynamically significant ductus as determined by a neonatologist.
- One hundred and thirty-six premature infants received either placebo or ibuprofen lysine (10 mg/kg on the first dose and 5 mg/kg at 24 and 48 hours). Mean birth age was 1.5 days (range: 4.6 – 73.0 hours), mean gestational age was 26 weeks (range: 23 – 30 weeks), and mean weight was 798 g (range: 530 – 1015 g). All infants had a documented PDA with evidence of ductal shunting. As shown in TABLE 2, 25% of infants on ibuprofen lysine required rescue therapy versus 48% of infants on placebo (p=0.003 from logistic regression controlling for site).
- Of the infants requiring rescue within the first 14 days after the first dose of study drug, no statistically significant difference was observed between the ibuprofen lysine and placebo groups for mean age at start of first rescue treatment (8.7 days, range 4-15 days, for the ibuprofen lysine group and 6.9 days, range 2-15 days, for the placebo group).
- The groups were similar in the number of deaths by day 14, the number of patients on a ventilator or requiring oxygenation at day 1, 4 and 14, the number of patients requiring surgical ligation of their PDA (12%), the number of cases of pulmonary hemorrhage and pulmonary hypertension by day 14, and bronchopulmonary dysplasia at day 28. In addition, no significant differences were noted in the incidences of Stage 2 and 3 necrotizing enterocolitis, Grades 3 and 4 intraventricular hemorrhage, periventricular leukomalacia and retinopathy of prematurity between groups as determined at 36±1 weeks adjusted gestational age.
- Two supportive studies also determined that ibuprofen, either prophylactically (n=433, weight range: 400 – 2165 g) or as treatment (n=210, weight range: 400 – 2370 g), was superior to placebo (or no treatment) in preventing the need for rescue therapy for a symptomatic PDA.
How Supplied
- Ibuprofen lysine injection is dispensed in clear glass single-use vials, each containing 2 mL of sterile solution (NDC 55292-122- 52). The solution is not buffered and contains no preservatives. Each milliliter contains 17.1 mg/mL (±)-ibuprofen L-lysine [equivalent to 10 mg/mL (±)-ibuprofen] dissolved in Water for Injection, USP. Ibuprofen lysine is supplied in a carton containing 3 single-use vials.
Storage
- Store at 20 – 25°C (68 – 77°F); excursions permitted 15 – 30°C (59 – 86°F). Protect from light. Store vials in carton until contents have been used.
Images
Drug Images
{{#ask: Page Name::Ibuprofen lysine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel


{{#ask: Label Page::Ibuprofen lysine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
General
- Patients’ caregivers should be informed that the effects of ibuprofen on infants’ neurodevelopmental outcome, growth and disease process with prematurity have not been assessed in long-term studies.
Infection
- Ibuprofen lysine may alter signs of infection. Patients’ caregivers should be informed that the infant will be carefully monitored for any signs of infection.
Platelet Aggregation
- Patients’ caregivers should be informed that like other NSAIDS, ibuprofen lysine can inhibit clot formation therefore their infant will be monitored for any signs of bleeding.
Bilirubin Displacement
- Patients’ caregivers should be informed that the infants’ blood will be tested for increased levels of total bilirubin.
Administration
- Patients’ caregivers should be informed that the infants’ skin and tissues will be monitored as leakage from administration may be irritating to tissue.
Precautions with Alcohol
Alcohol-Ibuprofen lysine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- NeoProfen®[1]
Look-Alike Drug Names
There is limited information regarding Ibuprofen lysine Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.