Rivaroxaban: Difference between revisions
No edit summary |
No edit summary |
||
| Line 12: | Line 12: | ||
<i><span style="color:#FF0000;">SPINAL/EPIDURAL HEMATOMA:</span></i>[[Epidural hematoma|Epidural]] or spinal hematomas have occurred in patients treated with rivaroxaban who are receiving neuraxial anesthesia or undergoing | <i><span style="color:#FF0000;">SPINAL/EPIDURAL HEMATOMA:</span></i>[[Epidural hematoma|Epidural]] or spinal hematomas have occurred in patients treated with rivaroxaban who are receiving neuraxial anesthesia or undergoing spinal puncture. These hematomas may result in long-term or permanent [[paralysis]]. Consider these risks when scheduling patients for spinal procedures. Factors that can increase the risk of developing epidural or spinal hematomas in these patients include: | ||
* Use of indwelling [[epidural catheter]]s | * Use of indwelling [[epidural catheter]]s | ||
* Concomitant use of other drugs that affect [[hemostasis]], such as [[non-steroidal anti-inflammatory drugs]] ([[NSAIDs]]), platelet inhibitors, other [[anticoagulants]] | * Concomitant use of other drugs that affect [[hemostasis]], such as [[non-steroidal anti-inflammatory drugs]] ([[NSAIDs]]), platelet inhibitors, other [[anticoagulants]] | ||
*A history of traumatic or repeated epidural or | *A history of traumatic or repeated epidural or spinal punctures | ||
*A history of spinal deformity or | *A history of spinal deformity or spinal surgery. | ||
*Optimal timing between the administration of rivaroxaban and neuraxial procedures is not known | *Optimal timing between the administration of rivaroxaban and neuraxial procedures is not known | ||
| Line 189: | Line 189: | ||
====Acute PE in Hemodynamically Unstable Patients or Patients Who Require Thrombolysis or Pulmonary Embolectomy==== | ====Acute PE in Hemodynamically Unstable Patients or Patients Who Require Thrombolysis or Pulmonary Embolectomy==== | ||
*Initiation of rivaroxaban is not recommended acutely as an alternative to [[unfractionated heparin]] in patients with [[pulmonary embolism]] who present with [[hemodynamic instability]] or who may receive [[thrombolysis]] or [[pulmonary embolectomy]] | *Initiation of rivaroxaban is not recommended acutely as an alternative to [[unfractionated heparin]] in patients with [[pulmonary embolism]] who present with [[hemodynamic instability]] or who may receive [[thrombolysis]] or [[pulmonary embolectomy]] | ||
|clinicalTrials= | |clinicalTrials=During clinical development for the approved indications, 16326 patients were exposed to rivaroxaban. These included 7111 patients who received rivaroxaban 15 mg or 20 mg orally once daily for a mean of 19 months (5558 for 12 months and 2512 for 24 months) to reduce the risk of [[stroke]] and systemic [[embolism]] in nonvalvular [[atrial fibrillation]] (ROCKET AF); 4728 patients who received either rivaroxaban 15 mg orally twice daily for three weeks followed by 20 mg orally once daily (EINSTEIN DVT, EINSTEIN PE) or 20 mg orally once daily (EINSTEIN Extension) to treat [[DVT]], [[PE]], and to reduce the risk of recurrence of [[DVT]] and of [[PE]]; and 4487 patients who received rivaroxaban 10 mg orally once daily for prophylaxis of DVT following hip or knee replacement surgery (RECORD 1–3). | ||
During clinical development for the approved indications, 16326 patients were exposed to rivaroxaban. These included 7111 patients who received rivaroxaban 15 mg or 20 mg orally once daily for a mean of 19 months (5558 for 12 months and 2512 for 24 months) to reduce the risk of [[stroke]] and systemic [[embolism]] in nonvalvular [[atrial fibrillation]] (ROCKET AF); 4728 patients who received either rivaroxaban 15 mg orally twice daily for three weeks followed by 20 mg orally once daily (EINSTEIN DVT, EINSTEIN PE) or 20 mg orally once daily (EINSTEIN Extension) to treat [[DVT]], [[PE]], and to reduce the risk of recurrence of [[DVT]] and of [[PE]]; and 4487 patients who received rivaroxaban 10 mg orally once daily for prophylaxis of DVT following hip or knee replacement surgery (RECORD 1–3). | |||
====Hemorrhage==== | ====Hemorrhage==== | ||
The most common adverse reactions with rivaroxaban were bleeding complications. | The most common adverse reactions with rivaroxaban were [[bleeding]] complications. | ||
====Nonvalvular Atrial Fibrillation ==== | ====Nonvalvular Atrial Fibrillation==== | ||
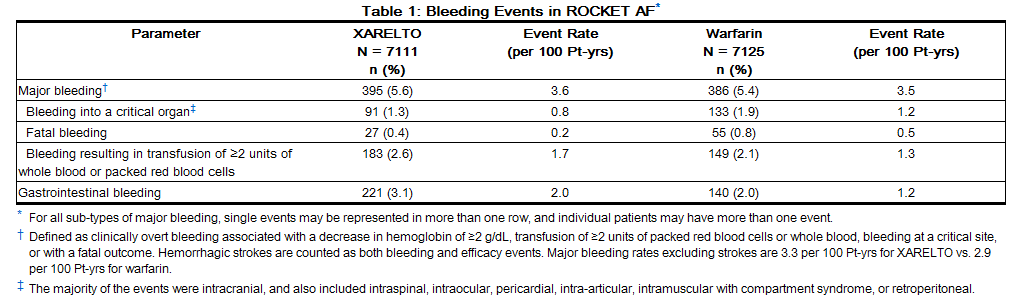
In the ROCKET AF trial, the most frequent adverse reactions associated with permanent drug discontinuation were bleeding events, with incidence rates of 4.3% for rivaroxaban vs. 3.1% for [[warfarin]]. The incidence of discontinuations for non-bleeding adverse events was similar in both treatment groups. | In the ROCKET AF trial, the most frequent adverse reactions associated with permanent drug discontinuation were bleeding events, with incidence rates of 4.3% for rivaroxaban vs. 3.1% for [[warfarin]]. The incidence of discontinuations for non-bleeding adverse events was similar in both treatment groups. | ||
| Line 204: | Line 202: | ||
[[image:riva3.png|600px|left|thumb]] | [[image:riva3.png|600px|left|thumb]] | ||
{{clr}} | {{clr}} | ||
===Treatment of Deep Vein Thrombosis (DVT), Pulmonary Embolism (PE), and to Reduce the Risk of Recurrence of DVT and of PE=== | ====Treatment of Deep Vein Thrombosis (DVT), Pulmonary Embolism (PE), and to Reduce the Risk of Recurrence of DVT and of PE==== | ||
====EINSTEIN DVT and EINSTEIN PE Studies==== | =====EINSTEIN DVT and EINSTEIN PE Studies===== | ||
In the pooled analysis of the EINSTEIN DVT and EINSTEIN PE clinical studies, the most frequent adverse reactions leading to permanent drug discontinuation were bleeding events, with rivaroxaban vs. [[enoxaparin]]/[[Vitamin K antagonist]] (VKA) incidence rates of 1.7% vs. 1.5%, respectively. The mean duration of treatment was 208 days for rivaroxaban-treated patients and 204 days for [[enoxaparin]]/[[VKA]]-treated patients. | In the pooled analysis of the EINSTEIN DVT and EINSTEIN PE clinical studies, the most frequent adverse reactions leading to permanent drug discontinuation were bleeding events, with rivaroxaban vs. [[enoxaparin]]/[[Vitamin K antagonist]] (VKA) incidence rates of 1.7% vs. 1.5%, respectively. The mean duration of treatment was 208 days for rivaroxaban-treated patients and 204 days for [[enoxaparin]]/[[VKA]]-treated patients. | ||
| Line 213: | Line 211: | ||
[[image:riva4.png|600px|left|thumb]] | [[image:riva4.png|600px|left|thumb]] | ||
{{clr}} | {{clr}} | ||
====EINSTEIN Extension Study==== | =====EINSTEIN Extension Study===== | ||
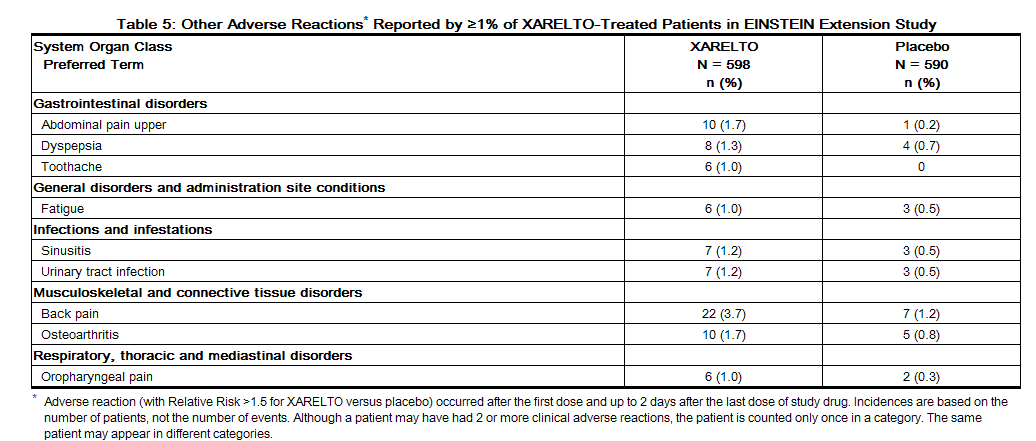
In the EINSTEIN Extension clinical study, the most frequent adverse reactions associated with permanent drug discontinuation were bleeding events, with incidence rates of 1.8% for rivaroxaban vs. 0.2% for placebo treatment groups. The mean duration of treatment was 190 days for both rivaroxaban and placebo treatment groups. | In the EINSTEIN Extension clinical study, the most frequent adverse reactions associated with permanent drug discontinuation were bleeding events, with incidence rates of 1.8% for rivaroxaban vs. 0.2% for placebo treatment groups. The mean duration of treatment was 190 days for both rivaroxaban and placebo treatment groups. | ||
| Line 220: | Line 218: | ||
[[image:riva5.png|600px|left|thumb]] | [[image:riva5.png|600px|left|thumb]] | ||
{{clr}} | {{clr}} | ||
===Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery === | |||
====Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery==== | |||
In the RECORD clinical trials, the overall incidence rate of adverse reactions leading to permanent treatment discontinuation was 3.7% with rivaroxaban. | In the RECORD clinical trials, the overall incidence rate of adverse reactions leading to permanent treatment discontinuation was 3.7% with rivaroxaban. | ||
Revision as of 15:20, 2 July 2014
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Alejandro Lemor, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
(A) PREMATURE DISCONTINUATION OF XARELTO INCREASES THE RISK OF THROMBOTIC EVENTS,
(B) SPINAL/EPIDURAL HEMATOMA See full prescribing information for complete Boxed Warning.
PREMATURE DISCONTINUATION OF RIVAROXABAN INCREASES THE RISK OF THROMBOTIC EVENTS: Premature discontinuation of any oral anticoagulant, including rivaroxaban, increases the risk of thrombotic events. If anticoagulation with rivaroxaban is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant.
Monitor patients frequently for signs and symptoms of neurological impairment. If neurological compromise is noted, urgent treatment is necessary. Consider the benefits and risks before neuraxial intervention in patients anticoagulated or to be anticoagulated for thromboprophylaxis |
Overview
Rivaroxaban is a Factor Xa inhibitor that is FDA approved for the {{{indicationType}}} of reduction in the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation, treatment of deep vein thrombosis (DVT), pulmonary embolism (PE), reduction in the risk of recurrence of DVT and of PE, and for the prophylaxis of DVT, which may lead to PE in patients undergoing knee or hip replacement surgery.. There is a Black Box Warning for this drug as shown here. Common adverse reactions include bleeding.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Reduction of Risk of Stroke and Systemic Embolism in Nonvalvular Atrial Fibrillation
- Rivaroxaban is indicated to reduce the risk of stroke and systemic embolism in patients with nonvalvular atrial fibrillation.
- There are limited data on the relative effectiveness of Rivaroxaban and warfarin in reducing the risk of stroke and systemic embolism when warfarin therapy is well-controlled.
Treatment of Deep Vein Thrombosis
- Rivaroxaban is indicated for the treatment of deep vein thrombosis (DVT).
Treatment of Pulmonary Embolism
- Rivaroxaban is indicated for the treatment of pulmonary embolism (PE).
Reduction in the Risk of Recurrence of Deep Vein Thrombosis and of Pulmonary Embolism
- Rivaroxaban is indicated for the reduction in the risk of recurrence of deep vein thrombosis and of pulmonary embolism following initial 6 months treatment for DVT and/or PE.
Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery
- Rivaroxaban is indicated for the prophylaxis of DVT, which may lead to PE in patients undergoing knee or hip replacement surgery
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
Condition 1
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Non–Guideline-Supported Use
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
Condition 1
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Condition 2
- Developed by: (Organisation)
- Class of Recommendation: (Class) (Link)
- Strength of Evidence: (Category A/B/C) (Link)
- Dosing Information/Recommendation
- (Dosage)
Non–Guideline-Supported Use
Condition 1
- Dosing Information
- (Dosage)
Condition 2
- Dosing Information
- (Dosage)
Condition 3
- Dosing Information
- (Dosage)
Contraindications
- Active pathological bleeding
- Severe hypersensitivity reaction to rivaroxaban
Warnings
|
(A) PREMATURE DISCONTINUATION OF XARELTO INCREASES THE RISK OF THROMBOTIC EVENTS,
(B) SPINAL/EPIDURAL HEMATOMA See full prescribing information for complete Boxed Warning.
PREMATURE DISCONTINUATION OF RIVAROXABAN INCREASES THE RISK OF THROMBOTIC EVENTS: Premature discontinuation of any oral anticoagulant, including rivaroxaban, increases the risk of thrombotic events. If anticoagulation with rivaroxaban is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant.
Monitor patients frequently for signs and symptoms of neurological impairment. If neurological compromise is noted, urgent treatment is necessary. Consider the benefits and risks before neuraxial intervention in patients anticoagulated or to be anticoagulated for thromboprophylaxis |
Increased Risk of Thrombotic Events after Premature Discontinuation
- Premature discontinuation of any oral anticoagulant, including rivaroxaban, in the absence of adequate alternative anticoagulation increases the risk of thrombotic events.
- An increased rate of stroke was observed during the transition from rivaroxaban to warfarin in clinical trials in atrial fibrillation patients.
- If rivaroxaban is discontinued for a reason other than pathological bleeding or completion of a course of therapy, consider coverage with another anticoagulant.
Risk of Bleeding
- Rivaroxaban increases the risk of bleeding and can cause serious or fatal bleeding. In deciding whether to prescribe rivaroxaban to patients at increased risk of bleeding, the risk of thrombotic events should be weighed against the risk of bleeding.
- Promptly evaluate any signs or symptoms of blood loss and consider the need for blood replacement.
- Discontinue rivaroxaban in patients with active pathological hemorrhage. The terminal elimination half-life of rivaroxaban is 5 to 9 hours in healthy subjects aged 20 to 45 years.
- Concomitant use of other drugs that impair hemostasis increases the risk of bleeding. These include aspirin, P2Y12 platelet inhibitors, other antithrombotic agents, fibrinolytic therapy, and non-steroidal anti-inflammatory drugs (NSAIDs).
- Concomitant use of drugs that are combined P-gp and CYP3A4 inhibitors (e.g., ketoconazole and ritonavir) increases rivaroxaban exposure and may increase bleeding risk.
Reversal of Anticoagulant Effect=
- A specific antidote for rivaroxaban is not available. Because of high plasma protein binding, rivaroxaban is not expected to be dialyzable.
- Protamine sulfate and vitamin K are not expected to affect the anticoagulant activity of rivaroxaban.
- Partial reversal of prothrombin time prolongation has been seen after administration of prothrombin complex concentrates (PCCs) in healthy volunteers.
- The use of other procoagulant reversal agents like activated prothrombin complex concentrate (APCC) or recombinant factor VIIa (rFVIIa) has not been evaluated.
Spinal/Epidural Anesthesia or Puncture
- When neuraxial anesthesia (spinal/epidural anesthesia) or spinal puncture is employed, patients treated with anticoagulant agents for prevention of thromboembolic complications are at risk of developing an epidural or spinal hematoma which can result in long-term or permanent paralysis.
- An epidural catheter should not be removed earlier than 18 hours after the last administration of rivaroxaban.
- The next rivaroxaban dose is not to be administered earlier than 6 hours after the removal of the catheter.
- If traumatic puncture occurs, the administration of rivaroxaban is to be delayed for 24 hours.
Use in Patients with Renal Impairment
Nonvalvular Atrial Fibrillation
- Avoid the use of rivaroxaban in patients with CrCl <15 mL/min since drug exposure is increased.
- Periodically assess renal function as clinically indicated (i.e., more frequently in situations in which renal function may decline) and adjust therapy accordingly.
- Discontinue rivaroxaban in patients who develop acute renal failure while on rivaroxaban.
Treatment of Deep Vein Thrombosis (DVT), Pulmonary Embolism (PE), and Reduction in the Risk of Recurrence of DVT and of PE
- Avoid the use of rivaroxaban in patients with CrCl <30 mL/min due to an expected increase in rivaroxaban exposure and pharmacodynamic effects in this patient population.
Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery
- Avoid the use of rivaroxaban in patients with CrCl <30 mL/min due to an expected increase in rivaroxaban exposure and pharmacodynamic effects in this patient population.
- Observe closely and promptly evaluate any signs or symptoms of blood loss in patients with CrCl 30 to 50 mL/min.
- Patients who develop acute renal failure while on rivaroxaban should discontinue the treatment.
Use in Patients with Hepatic Impairment
- No clinical data are available for patients with severe hepatic impairment.
- Avoid use of rivaroxaban in patients with moderate (Child-Pugh B) and severe (Child-Pugh C) hepatic impairment or with any hepatic disease associated with coagulopathy since drug exposure and bleeding risk may be increased.
Use with P-gp and Strong CYP3A4 Inhibitors or Inducers
- Avoid concomitant use of rivaroxaban with combined P-gp and strong CYP3A4 inhibitors (e.g., ketoconazole, itraconazole, lopinavir/ritonavir, ritonavir, indinavir, and conivaptan).
- Avoid concomitant use of rivaroxaban with drugs that are combined P-gp and strong CYP3A4 inducers (e.g., carbamazepine, phenytoin, rifampin, St. John's wort).
Risk of Pregnancy-Related Hemorrhage
- In pregnant women, rivaroxaban should be used only if the potential benefit justifies the potential risk to the mother and fetus. rivaroxaban dosing in pregnancy has not been studied.
- The anticoagulant effect of rivaroxaban cannot be monitored with standard laboratory testing nor readily reversed.
- Promptly evaluate any signs or symptoms suggesting blood loss (e.g., a drop in hemoglobin and/or hematocrit, hypotension, or fetal distress).
Patients with Prosthetic Heart Valves
- The safety and efficacy of rivaroxaban have not been studied in patients with prosthetic heart valves. Therefore, use of rivaroxaban is not recommended in these patients.
Acute PE in Hemodynamically Unstable Patients or Patients Who Require Thrombolysis or Pulmonary Embolectomy
- Initiation of rivaroxaban is not recommended acutely as an alternative to unfractionated heparin in patients with pulmonary embolism who present with hemodynamic instability or who may receive thrombolysis or pulmonary embolectomy
Adverse Reactions
Clinical Trials Experience
During clinical development for the approved indications, 16326 patients were exposed to rivaroxaban. These included 7111 patients who received rivaroxaban 15 mg or 20 mg orally once daily for a mean of 19 months (5558 for 12 months and 2512 for 24 months) to reduce the risk of stroke and systemic embolism in nonvalvular atrial fibrillation (ROCKET AF); 4728 patients who received either rivaroxaban 15 mg orally twice daily for three weeks followed by 20 mg orally once daily (EINSTEIN DVT, EINSTEIN PE) or 20 mg orally once daily (EINSTEIN Extension) to treat DVT, PE, and to reduce the risk of recurrence of DVT and of PE; and 4487 patients who received rivaroxaban 10 mg orally once daily for prophylaxis of DVT following hip or knee replacement surgery (RECORD 1–3).
Hemorrhage
The most common adverse reactions with rivaroxaban were bleeding complications.
Nonvalvular Atrial Fibrillation
In the ROCKET AF trial, the most frequent adverse reactions associated with permanent drug discontinuation were bleeding events, with incidence rates of 4.3% for rivaroxaban vs. 3.1% for warfarin. The incidence of discontinuations for non-bleeding adverse events was similar in both treatment groups.
Table 1 shows the number of patients experiencing various types of bleeding events in the ROCKET AF trial.
Treatment of Deep Vein Thrombosis (DVT), Pulmonary Embolism (PE), and to Reduce the Risk of Recurrence of DVT and of PE
EINSTEIN DVT and EINSTEIN PE Studies
In the pooled analysis of the EINSTEIN DVT and EINSTEIN PE clinical studies, the most frequent adverse reactions leading to permanent drug discontinuation were bleeding events, with rivaroxaban vs. enoxaparin/Vitamin K antagonist (VKA) incidence rates of 1.7% vs. 1.5%, respectively. The mean duration of treatment was 208 days for rivaroxaban-treated patients and 204 days for enoxaparin/VKA-treated patients.
Table 2 shows the number of patients experiencing major bleeding events in the pooled analysis of the EINSTEIN DVT and EINSTEIN PE studies.

EINSTEIN Extension Study
In the EINSTEIN Extension clinical study, the most frequent adverse reactions associated with permanent drug discontinuation were bleeding events, with incidence rates of 1.8% for rivaroxaban vs. 0.2% for placebo treatment groups. The mean duration of treatment was 190 days for both rivaroxaban and placebo treatment groups.
Table 3 shows the number of patients experiencing bleeding events in the EINSTEIN Extension study.

Prophylaxis of Deep Vein Thrombosis Following Hip or Knee Replacement Surgery
In the RECORD clinical trials, the overall incidence rate of adverse reactions leading to permanent treatment discontinuation was 3.7% with rivaroxaban.
The rates of major bleeding events and any bleeding events observed in patients in the RECORD clinical trials are shown in Table 4.

Following rivaroxaban treatment, the majority of major bleeding complications (≥60%) occurred during the first week after surgery.
Other Adverse Reactions
Non-hemorrhagic adverse reactions reported in ≥1% of rivaroxaban-treated patients in the EINSTEIN Extension study are shown in Table 5.
Non-hemorrhagic adverse reactions reported in ≥1% of rivaroxaban-treated patients in RECORD 1–3 studies are shown in Table 6.

Other clinical trial experience: In an investigational study of acute medically ill patients being treated with rivaroxaban 10 mg tablets, cases of pulmonary hemorrhage and pulmonary hemorrhage with bronchiectasis were observed.
Postmarketing Experience
The following adverse reactions have been identified during post-approval use of rivaroxaban. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Blood and lymphatic system disorders
Gastrointestinal disorders
Hepatobiliary disorders
- Jaundice
- Cholestasis
- Cytolytic hepatitis
Immune system disorders
Nervous system disorders
Skin and subcutaneous tissue disorders
Drug Interactions
- Drug 1
- Drug 2
- Drug 3
- Drug 4
- Drug 5
Drug 1
(Description)
Drug 2
(Description)
Drug 3
(Description)
Drug 4
(Description)
Drug 5
(Description)
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
(Description)
Pregnancy Category (AUS):
(Description)
Labor and Delivery
(Description)
Nursing Mothers
(Description)
Pediatric Use
(Description)
Geriatic Use
(Description)
Gender
(Description)
Race
(Description)
Renal Impairment
(Description)
Hepatic Impairment
(Description)
Females of Reproductive Potential and Males
(Description)
Immunocompromised Patients
(Description)
Others
(Description)
Administration and Monitoring
Administration
(Oral/Intravenous/etc)
Monitoring
Condition 1
(Description regarding monitoring, from Warnings section)
Condition 2
(Description regarding monitoring, from Warnings section)
Condition 3
(Description regarding monitoring, from Warnings section)
IV Compatibility
Solution
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Y-Site
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Admixture
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Syringe
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
TPN/TNA
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Overdosage
Acute Overdose
Signs and Symptoms
(Description)
Management
(Description)
Chronic Overdose
Signs and Symptoms
(Description)
Management
(Description)
Pharmacology
| |
Rivaroxaban
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | ? |
| ATC code | ? |
| PubChem | ? |
| Chemical data | |
| Formula | ? |
| Mol. mass | ? |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
(Description)
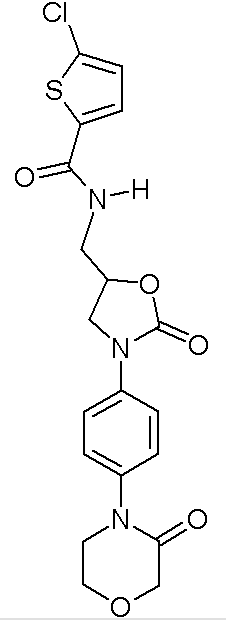
Structure
(Description with picture)
Pharmacodynamics
(Description)
Pharmacokinetics
(Description)
Nonclinical Toxicology
(Description)
Clinical Studies
Condition 1
(Description)
Condition 2
(Description)
Condition 3
(Description)
How Supplied
(Description)
Storage
There is limited information regarding Rivaroxaban Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Rivaroxaban |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Rivaroxaban |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
(Patient Counseling Information)
Precautions with Alcohol
Alcohol-Rivaroxaban interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Rivaroxaban Brand Names in the drug label.
Look-Alike Drug Names
- (Paired Confused Name 1a) — (Paired Confused Name 1b)
- (Paired Confused Name 2a) — (Paired Confused Name 2b)
- (Paired Confused Name 3a) — (Paired Confused Name 3b)
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.