Hereditary nonpolyposis colorectal cancer pathophysiology
|
Hereditary Nonpolyposis Colorectal Cancer Microchapters |
|
Differentiating Hereditary Nonpolyposis Colorectal Cancer from other Diseases |
|---|
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Hereditary nonpolyposis colorectal cancer pathophysiology On the Web |
|
American Roentgen Ray Society Images of Hereditary nonpolyposis colorectal cancer pathophysiology |
|
FDA on Hereditary nonpolyposis colorectal cancer pathophysiology |
|
CDC on Hereditary nonpolyposis colorectal cancer pathophysiology |
|
Hereditary nonpolyposis colorectal cancer pathophysiology in the news |
|
Blogs on Hereditary nonpolyposis colorectal cancer pathophysiology |
|
Directions to Hospitals Treating Hereditary nonpolyposis colorectal cancer |
|
Risk calculators and risk factors for Hereditary nonpolyposis colorectal cancer pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]Associate Editor(s)-in-Chief: Maria Fernanda Villarreal, M.D. [2]
Overview
HNPCC is an autosomal dominant genetic disease characterized by the early onset of colon cancer, endometrial cancer and other tumors caused by a genetic mutation within DNA mismatch repair (MMR) genes.[1] Two-thirds of hereditary nonpolyposis colorectal cancers occur in the proximal colon.
Pathogenesis
HNPCC defects in DNA mismatch repair lead to microsatellite instability, also known as MSI-H, which is a hallmark of HNPCC. Three major groups of MSI-H cancers can be recognized by histopathological criteria:
- (1) right-sided poorly differentiated cancers
- (2) right-sided mucinous cancers
- (3) adenocarcinomas in any location showing any measurable level of intraepithelial lymphocyte (TIL)
MSI is identifiable in cancer specimens in the pathology laboratory. In hereditary nonpolyposis colorectal cancer, colon malignancies usually occur from a single or a few adenomas, and mostly occurs in the proximal colon.
Genetics
HNPCC is known to be associated with mutations in genes involved in the DNA mismatch repair pathway
| OMIM name | Genes implicated in HNPCC | Frequency of mutations in HNPCC families | Locus | First publication |
|---|---|---|---|---|
| HNPCC1 (120435) | MSH2 | approximately 60% | 2p22 | Fishel 1993[2] |
| HNPCC2 (609310) | MLH1 | approximately 30% | 3p21 | Papadopoulos 1994[3] |
| HNPCC5 | MSH6 | 7-10% | 2p16 | Miyaki 1997[4] |
| HNPCC4 | PMS2 | relatively infrequent | 7p22 [5] | Nicolaides 1994 |
| HNPCC3 | PMS1 | case report[6] | 2q31-q33 | Nicolaides 1994 |
| HNPCC6 | TGFBR2 | case report[7] | 3p22 | |
| HNPCC7 | MLH3 | disputed[8] | 14q24.3 |
Up to 39% of families with mutations in an HNPCC gene do not meet the Amsterdam criteria. Therefore, families found to have a deleterious mutation in an HNPCC gene should be considered to have HNPCC regardless of the extent of the family history. This also means that the Amsterdam criteria fail to identify many patients at risk for Lynch syndrome. Improving the criteria for screening is an active area of research, as detailed in the Screening Strategies section of this article.
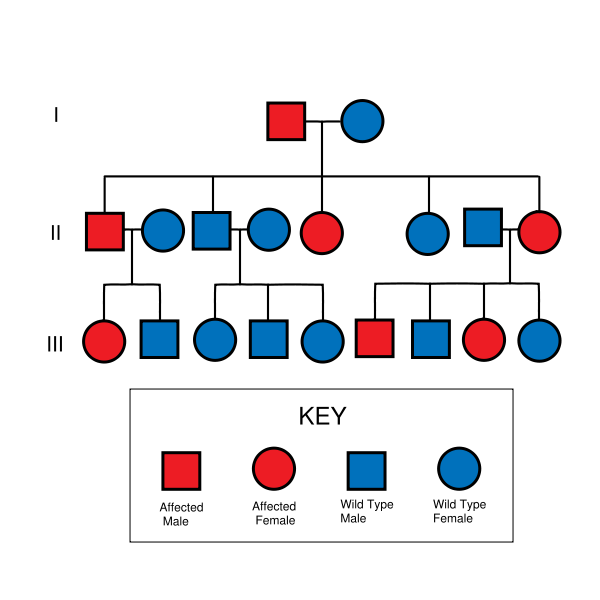
HNPCC is inherited in an autosomal dominant manner. Most people with HNPCC inherit the condition from a parent. However, due to incomplete penetrance, variable age of cancer diagnosis, cancer risk reduction, or early death, not all patients with an HNPCC gene mutation have a parent who had cancer. Some patients develop HNPCC de-novo in a new generation, without inheriting the gene. These patients are often only identified after developing an early-life colon cancer. Parents with HNPCC have a 50% chance to pass the gene on to each child.
Associated Conditions
- Colorectal carcinoma
- Endometrial carcinoma
- Non-endometrioid endometrial carcinoma,[9] e.g. endometrial clear cell carcinoma.[10]
- Endometrioid endometrial carcinoma[11]
- Suggestive features: lower uterine segment, tumor infiltrating lymphocytes.[12]
- Stomach carcinoma,[13] intestinal-type.[14]
- Biliary tree carcinoma[13]
- Pancreatic carcinoma[13]
- Urinary system carcinoma[13]
Gross Pathology
On gross pathology, there are no characteristic findings of HNPCC.
Microscopic Pathology
On microscopic histopathological analysis, HNPCC is more likely to be poorly differentiated, abundant in extracellular mucin, and distinguished by a lymphoid (peritumoral lymphocytes) host response to the tumor.
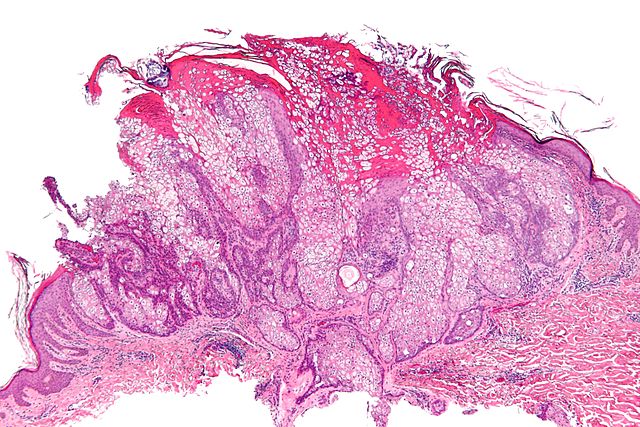
Gallery
-
Micrograph of a sebaceous adenoma, as may be seen in Muir-Torre syndrome.
References
- ↑ Si JW, Wang L, Ba XJ, Zhang X, Dong Y, Zhang JX, Li WT, Li T (2015). "[Clinicopathological screening of Lynch syndrome: a report of 2 cases and literature review]". Beijing Da Xue Xue Bao (in Chinese). 47 (5): 858–64. PMID 26474631.
- ↑ Fishel R, Lescoe MK, Rao MR, Copeland NG, Jenkins NA, Garber J, Kane M, Kolodner R (Dec 1993). "The human mutator gene homolog MSH2 and its association with hereditary nonpolyposis colon cancer". Cell. 75 (5): 1027–38. doi:10.1016/0092-8674(93)90546-3. PMID 8252616.
- ↑ Papadopoulos N, Nicolaides NC, Wei YF, Ruben SM, Carter KC, Rosen CA, Haseltine WA, Fleischmann RD, Fraser CM, Adams MD (Mar 1994). "Mutation of a mutL homolog in hereditary colon cancer". Science. 263 (5153): 1625–9. doi:10.1126/science.8128251. PMID 8128251.
- ↑ Miyaki M, Konishi M, Tanaka K, Kikuchi-Yanoshita R, Muraoka M, Yasuno M, Igari T, Koike M, Chiba M, Mori T (Nov 1997). "Germline mutation of MSH6 as the cause of hereditary nonpolyposis colorectal cancer". Nature Genetics. 17 (3): 271–2. doi:10.1038/ng1197-271. PMID 9354786.
- ↑ Nicolaides NC, Papadopoulos N, Liu B, Wei YF, Carter KC, Ruben SM, Rosen CA, Haseltine WA, Fleischmann RD, Fraser CM (Sep 1994). "Mutations of two PMS homologues in hereditary nonpolyposis colon cancer". Nature. 371 (6492): 75–80. doi:10.1038/371075a0. PMID 8072530.
- ↑ Nicolaides NC, Papadopoulos N, Liu B, Wei YF, Carter KC, Ruben SM, Rosen CA, Haseltine WA, Fleischmann RD, Fraser CM (Sep 1994). "Mutations of two PMS homologues in hereditary nonpolyposis colon cancer". Nature. 371 (6492): 75–80. doi:10.1038/371075a0. PMID 8072530.
- ↑ Lu SL, Kawabata M, Imamura T, Akiyama Y, Nomizu T, Miyazono K, Yuasa Y (May 1998). "HNPCC associated with germline mutation in the TGF-beta type II receptor gene". Nature Genetics. 19 (1): 17–8. doi:10.1038/ng0598-17. PMID 9590282.
- ↑ Ou J, Rasmussen M, Westers H, Andersen SD, Jager PO, Kooi KA, Niessen RC, Eggen BJ, Nielsen FC, Kleibeuker JH, Sijmons RH, Rasmussen LJ, Hofstra RM (Apr 2009). "Biochemical characterization of MLH3 missense mutations does not reveal an apparent role of MLH3 in Lynch syndrome". Genes, Chromosomes & Cancer. 48 (4): 340–50. doi:10.1002/gcc.20644. PMID 19156873.
- ↑ Okuda T, Sekizawa A, Purwosunu Y; et al. (2010). "Genetics of endometrial cancers". Obstet Gynecol Int. 2010: 984013. doi:10.1155/2010/984013. PMC 2852605. PMID 20396392.
- ↑ Garg, K.; Soslow, RA. (2009). "Lynch syndrome (hereditary non-polyposis colorectal cancer) and endometrial carcinoma". J Clin Pathol. 62 (8): 679–84. doi:10.1136/jcp.2009.064949. PMID 19638537. Unknown parameter
|month=ignored (help) - ↑ Lax, SF. (2002). "[Dualistic model of molecular pathogenesis in endometrial carcinoma]". Zentralbl Gynakol. 124 (1): 10–6. doi:10.1055/s-2002-20303. PMID 11873308. Unknown parameter
|month=ignored (help) - ↑ Garg, K.; Soslow, RA. (2009). "Lynch syndrome (hereditary non-polyposis colorectal cancer) and endometrial carcinoma". J Clin Pathol. 62 (8): 679–84. doi:10.1136/jcp.2009.064949. PMID 19638537. Unknown parameter
|month=ignored (help) - ↑ 13.0 13.1 13.2 13.3 Online Mendelian Inheritance in Man (OMIM) 120435
- ↑ Cristofaro, G.; Lynch, HT.; Caruso, ML.; Attolini, A.; DiMatteo, G.; Giorgio, P.; Senatore, S.; Argentieri, A.; Sbano, E. (1987). "New phenotypic aspects in a family with Lynch syndrome II". Cancer. 60 (1): 51–8. PMID 3581033. Unknown parameter
|month=ignored (help) - ↑ Barrow, E.; Robinson, L.; Alduaij, W.; Shenton, A.; Clancy, T.; Lalloo, F.; Hill, J.; Evans, DG. (2009). "Cumulative lifetime incidence of extracolonic cancers in Lynch syndrome: a report of 121 families with proven mutations". Clin Genet. 75 (2): 141–9. doi:10.1111/j.1399-0004.2008.01125.x. PMID 19215248. Unknown parameter
|month=ignored (help) - ↑ Renkonen-Sinisalo, L.; Sipponen, P.; Aarnio, M.; Julkunen, R.; Aaltonen, LA.; Sarna, S.; Järvinen, HJ.; Mecklin, JP. (2002). "No support for endoscopic surveillance for gastric cancer in hereditary non-polyposis colorectal cancer". Scand J Gastroenterol. 37 (5): 574–7. PMID 12059060. Unknown parameter
|month=ignored (help) - ↑ Crockett, DG.; Wagner, DG.; Holmäng, S.; Johansson, SL.; Lynch, HT. (2011). "Upper urinary tract carcinoma in Lynch syndrome cases". J Urol. 185 (5): 1627–30. doi:10.1016/j.juro.2010.12.102. PMID 21419447. Unknown parameter
|month=ignored (help) - ↑ 18.0 18.1 Hartmann, A.; Dietmaier, W.; Hofstädter, F.; Burgart, LJ.; Cheville, JC.; Blaszyk, H. (2003). "Urothelial carcinoma of the upper urinary tract: inverted growth pattern is predictive of microsatellite instability". Hum Pathol. 34 (3): 222–7. doi:10.1053/hupa.2003.22. PMID 12673555. Unknown parameter
|month=ignored (help) - ↑ van der Post, RS.; Kiemeney, LA.; Ligtenberg, MJ.; Witjes, JA.; Hulsbergen-van de Kaa, CA.; Bodmer, D.; Schaap, L.; Kets, CM.; van Krieken, JH. (2010). "Risk of urothelial bladder cancer in Lynch syndrome is increased, in particular among MSH2 mutation carriers". J Med Genet. 47 (7): 464–70. doi:10.1136/jmg.2010.076992. PMID 20591884. Unknown parameter
|month=ignored (help)