Trimetrexate
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Ammu Susheela, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
BOXED WARNING
See full prescribing information for complete Boxed Warning.
* TRIMETREXATE GLUCURONATE FOR INJECTION MUST BE USED WITH CONCURRENT LEUCOVORIN (LEUCOVORIN PROTECTION) TO AVOID POTENTIALLY SERIOUS OR LIFE-THREATENING TOXICITIES.
|
Overview
Trimetrexate is an antibiotic that is FDA approved for the treatment of moderate-to-severe Pneumocystis carinii pneumonia (PCP) in immunocompromised patients, including patients with the acquired immunodeficiency syndrome (AIDS), who are intolerant of, or are refractory to, trimethoprim-sulfamethoxazole therapy or for whom trimethoprim-sulfamethoxazole is contraindicated. There is a Black Box Warning for this drug as shown here. Common adverse reactions include hypersensitivity.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
- Trimetrexate glucuronate for injection with concurrent leucovorin administration (leucovorin protection) is indicated as an alternative therapy for the treatment of moderate-to-severe Pneumocystis carinii pneumonia (PCP) in immunocompromised patients, including patients with the acquired immunodeficiency syndrome (AIDS), who are intolerant of, or are refractory to, trimethoprim-sulfamethoxazole therapy or for whom trimethoprim-sulfamethoxazole is contraindicated.
- This indication is based on the results of a randomized, controlled double-blind trial comparing Neutrexin with concurrent leucovorin protection (TMTX/LV) to trimethoprim‑sulfamethoxazole (TMP/SMX) in patients with moderate-to-severe Pneumocystis carinii pneumonia, as well as results of a Treatment IND. These studies are summarized below:
Neutrexin Comparative Study with TMP/SMX
- This double-blind, randomized trial initiated by the AIDS Clinical Trials Group (ACTG) in 1988 was designed to compare the safety and efficacy of TMTX/LV to that of TMP/SMX for the treatment of histologically confirmed, moderate-to-severe PCP, defined as (A-a) baseline gradient >30 mmHg, in patients with AIDS.
- Of the 220 patients with histologically confirmed PCP, 109 were randomized to receive TMTX/LV and 111 to TMP/SMX. Study patients randomized to TMTX/LV treatment were to receive 45 mg/m2 of TMTX daily for 21 days plus 20 mg/m2 of LV every 6 hours for 24 days. Those randomized to TMP/SMX were to receive 5 mg/kg TMP plus 25 mg/kg SMX four times daily for 21 days.
- Response to therapy, defined as alive and off ventilatory support at completion of therapy, with no change in anti-pneumocystis therapy, or addition of supraphysiologic doses of steroids, occurred in fifty percent of patients in each treatment group.
- The observed mortality in the TMTX/LV treatment group was approximately twice that in the TMP/SMX treatment group (95% CI: 0.99 - 4.11). Thirty of 109 (27%) patients treated with TMTX/LV and 18 of 111 (16%) patients receiving TMP/SMX died during the 21-day treatment course or 4-week follow-up period. Twenty-seven of 30 deaths in the TMTX/LV arm were attributed to PCP; all 18 deaths in the TMP/SMX arm were attributed to PCP.
- A significantly smaller proportion of patients who received TMTX/LV compared to TMP/SMX failed therapy due to toxicity (10% vs. 25%), and a significantly greater proportion of patients failed due to lack of efficacy (40% vs. 24%). Six patients (12%) who responded to TMTX/LV relapsed during the one-month follow-up period; no patient responding to TMP/SMX relapsed during this period. Information is not available as to whether these patients received prophylaxis therapy for PCP.
Treatment IND
- The FDA granted a Treatment IND for Neutrexin with leucovorin protection in February 1988 to make trimetrexate therapy available to HIV-infected patients with histologically confirmed PCP who had disease refractory to or who were intolerant of TMP/SMX and/or intravenous pentamidine.
- Over 500 physicians in the United States participated in the Treatment IND. Of the first 753 patients enrolled, 577 were evaluable for efficacy. Of these, 227 patients were intolerant of both TMP/SMX and pentamidine (IST - patients intolerant of both standard therapies), 146 were intolerant of one therapy and refractory to the other (RIST - patients refractory to one therapy and intolerant of the other) and 204 were refractory to both therapies (RST - refractory to both standard therapies). This was a very ill patient population; 38% required ventilatory support at entry (Table 1). These studies did not have concurrent control groups.
- Caution: Trimetrexate must be administered with concurrent leucovorin (leucovorin protection) to avoid potentially serious or life-threatening toxicities. Leucovorin therapy must extend for 72 hours past the last dose of Neutrexin.
- Trimetrexate glucuronate for injection is administered at a dose of 45 mg/m2 once daily by intravenous infusion over 60 minutes. Leucovorin must be administered daily during treatment with trimetrexate and for 72 hours past the last dose of trimetrexate. Leucovorin may be administered intravenously at a dose of 20 mg/m2 over 5 to 10 minutes every 6 hours for a total daily dose of 80 mg/m2, or orally as 4 doses of 20 mg/m2 spaced equally throughout the day. The oral dose should be rounded up to the next higher 25 mg increment. The recommended course of therapy is 21 days of trimetrexate and 24 days of leucovorin.
- Trimetrexate and leucovorin may alternatively be dosed on a mg/kg basis, depending on the patient’s body weight, using the conversion factors shown in the table below:
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Trimetrexate in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Trimetrexate in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Trimetrexate in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Trimetrexate in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Trimetrexate in pediatric patients.
Contraindications
- (trimetrexate glucuronate for injection) is contraindicated in patients with clinically significant sensitivity to trimetrexate, leucovorin, or methotrexate.
Warnings
|
BOXED WARNING
See full prescribing information for complete Boxed Warning.
* TRIMETREXATE GLUCURONATE FOR INJECTION MUST BE USED WITH CONCURRENT LEUCOVORIN (LEUCOVORIN PROTECTION) TO AVOID POTENTIALLY SERIOUS OR LIFE-THREATENING TOXICITIES.
|
- Trimetrexate glucuronate for injection must be used with concurrent leucovorin to avoid potentially serious or life-threatening complications including bone marrow suppression, oral and gastrointestinal mucosal ulceration, and renal and hepatic dysfunction.
- Leucovorin therapy must extend for 72 hours past the last dose of Neutrexin.
- Patients should be informed that failure to take the recommended dose and duration of leucovorin can lead to fatal toxicity. Patients should be closely monitored for the development of serious hematologic adverse reactions.
- Trimetrexate can cause fetal harm. Trimetrexate when administered to a pregnant woman. Trimetrexate has been shown to be fetotoxic and teratogenic in rats and rabbits. Rats administered 1.5 and 2.5 mg/kg/day intravenously on gestational days 6-15 showed substantial postimplantation loss and severe inhibition of maternal weight gain.
- Trimetrexate administered intravenously to rats at 0.5 and 1.0 mg/kg/day on gestational days 6-15 retarded normal fetal development and was teratogenic. Rabbits administered trimetrexate intravenously at daily doses of 2.5 and 5.0 mg/kg/day on gestational days 6-18 resulted in significant maternal and fetal toxicity. In rabbits, trimetrexate at 0.1 mg/kg/day was teratogenic in the absence of significant maternal toxicity. These effects were observed using doses 1/20 to 1/2 the equivalent human therapeutic dose based on a mg/m2 basis. Teratogenic effects included skeletal, visceral, ocular, and cardiovascular abnormalities. If trimetrexate is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus. Women of childbearing potential should be advised to avoid becoming pregnant.
Adverse Reactions
Clinical Trials Experience
- Because many patients who participated in clinical trials of trimetrexate had complications of advanced HIV disease, it is difficult to distinguish adverse events caused by trimetrexate from those resulting from underlying medical conditions.
- Table 3 lists the adverse events that occurred in ≥ 1% of the patients who participated in the Comparative Study of trimetrexate plus leucovorin versus TMP/SMX.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Trimetrexate in the drug label.
Drug Interactions
- Drug
- Description
Use in Specific Populations
Pregnancy
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Trimetrexate in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Trimetrexate during labor and delivery.
Nursing Mothers
- It is not known if trimetrexate is excreted in human milk. Because many drugs are excreted in human milk and because of the potential for serious adverse reactions in nursing infants from trimetrexate, it is recommended that breast feeding be discontinued if the mother is treated with trimetrexate.
Pediatric Use
- The safety and effectiveness of trimetrexate for the treatment of histologically confirmed PCP has not been established for patients under 18 years of age. Two children, ages 15 months and 9 months, were treated with trimetrexate and leucovorin using a dose of 45 mg/m2 of trimetrexate per day for 21 days and 20 mg/m2 of leucovorin every 6 hours for 24 days. There were no serious or unexpected adverse effects.
Geriatic Use
There is no FDA guidance on the use of Trimetrexate with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Trimetrexate with respect to specific gender populations.
Race
There is no FDA guidance on the use of Trimetrexate with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Trimetrexate in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Trimetrexate in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Trimetrexate in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Trimetrexate in patients who are immunocompromised.
Administration and Monitoring
Administration
Monitoring
There is limited information regarding Monitoring of Trimetrexate in the drug label.
- Description
IV Compatibility
There is limited information regarding IV Compatibility of Trimetrexate in the drug label.
Overdosage
- Trimetrexate administered without concurrent
leucovorin can cause lethal complications. There has been no extensive experience in humans receiving single intravenous doses of trimetrexate greater than 90 mg/m2/day with concurrent leucovorin. The toxicities seen at this dose were primarily hematologic. In the event of overdose, trimetrexate should be stopped and leucovorin should be administered at a dose of 40 mg/m2 every 6 hours for 3 days. The LD50 of intravenous trimetrexate in mice is 62 mg/kg (186 mg/m2).
Pharmacology
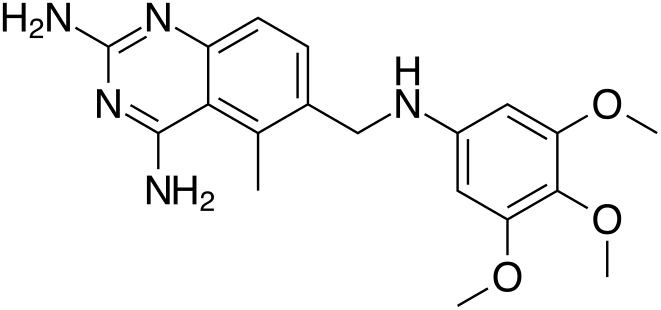
| |
Trimetrexate
| |
| Systematic (IUPAC) name | |
| 5-methyl-6-[(3,4,5-trimethoxyphenyl) aminomethyl] quinazoline-2,4-diamine | |
| Identifiers | |
| CAS number | |
| ATC code | P01 |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 369.418 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | VD: 20-30 Liters |
| Metabolism | Oxidative O-demethylation, followed by conjugation with glucuronide or sulfate |
| Half life | 11 to 12 hours |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
- In vitro studies have shown that trimetrexate is a competitive inhibitor of dihydrofolate reductase (DHFR) from bacterial, protozoan, and mammalian sources. DHFR catalyzes the reduction of intracellular dihydrofolate to the active coenzyme tetrahydrofolate. Inhibition of DHFR results in the depletion of this coenzyme, leading directly to interference with thymidylate biosynthesis, as well as inhibition of folate-dependent formyltransferases, and indirectly to inhibition of purine biosynthesis. The end result is disruption of DNA, RNA, and protein synthesis, with consequent cell death. Leucovorin (folinic acid) is readily transported into mammalian cells by an active, carrier‑mediated process and can be assimilated into cellular folate pools following its metabolism. In vitro studies have shown that leucovorin provides a source of reduced folates necessary for normal cellular biosynthetic processes. Because the Pneumocystis carinii organism lacks the reduced folate carrier-mediated transport system, leucovorin is prevented from entering the organism. Therefore, at concentrations achieved with therapeutic doses of trimetrexate plus leucovorin, the selective transport of trimetrexate, but not leucovorin, into the Pneumocystis carinii organism allows the concurrent administration of leucovorin to protect normal host cells from the cytotoxicity of trimetrexate without inhibiting the antifolate's inhibition of Pneumocystis carinii. It is not known if considerably higher doses of leucovorin would affect trimetrexate's effect on Pneumocystis carinii.
Structure
- Neutrexin (trimetrexate glucuronate for injection) administered without concurrent leucovorin can cause lethal complications. There has been no extensive experience in humans receiving single intravenous doses of trimetrexate greater than 90 mg/m2/day with concurrent leucovorin.
- The toxicities seen at this dose were primarily hematologic. In the event of overdose, trimetrexate should be stopped and leucovorin should be administered at a dose of 40 mg/m2 every 6 hours for 3 days. The LD50 of intravenous trimetrexate in mice is 62 mg/kg (186 mg/m2).
- The empirical formula for trimetrexate glucuronate is C19H23N5O3• C6H10O7 with a molecular weight of 563.56. The active ingredient, trimetrexate free base, has an empirical formula of C19H23N5O3 with a molecular weight of 369.42. Trimetrexate glucuronate for injection is a pale greenish-yellow powder or cake. Trimetrexate glucuronate is soluble in water (>50 mg/mL), whereas trimetrexate free base is practically insoluble in water (<0.1 mg/mL). The pKa of trimetrexate free base in 50% methanol/water is 8.0. The logarithm10 of the partition coefficient of trimetrexate free base between octanol and water is 1.63.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Trimetrexate in the drug label.
Pharmacokinetics
- Trimetrexate pharmacokinetics were assessed in six patients with acquired immunodeficiency syndrome (AIDS) who had Pneumocystis carinii pneumonia (4 patients) or toxoplasmosis (2 patients). Trimetrexate was administered intravenously as a bolus injection at a dose of 30 mg/m2/day along with leucovorin 20 mg/m2 every 6 hours for 21 days. Trimetrexate clearance (mean ± SD) was 38 ± 15 mL/min/m2 and volume of distribution at steady state (Vdss) was 20 ± 8 L/m2. The plasma concentration time profile declined in a biphasic manner over 24 hours with a terminal half-life of 11 ± 4 hours.
- The pharmacokinetics of trimetrexate without the concomitant administration of leucovorin have been evaluated in cancer patients with advanced solid tumors using various dosage regimens. The decline in plasma concentrations over time has been described by either biexponential or triexponential equations. Following the single-dose administration of 10 to 130 mg/m2 to 37 patients, plasma concentrations were obtained for 72 hours. Nine plasma concentration time profiles were described as biexponential. The alpha phase half-life was 57 ± 28 minutes, followed by a terminal phase with a half-life of 16 ± 3 hours. The plasma concentrations in the remaining patients exhibited a triphasic decline with half-lives of 8.6 ± 6.5 minutes, 2.4 ± 1.3 hours, and 17.8 ± 8.2 hours.
- Trimetrexate clearance in cancer patients has been reported as 53 ± 41 mL/min (14 patients) and 32 ± 18 mL/min/m2 (23 patients) following single-dose administration. After a five-day infusion of trimetrexate to 16 patients, plasma clearance was 30 ± 8 mL/min/m2.
- Renal clearance of trimetrexate in cancer patients has varied from about 4 ± 2 mL/min/m2 to 10 ± 6 mL/min/m2. Ten to 30% of the administered dose is excreted unchanged in the urine. Considering the free fraction of trimetrexate, active tubular secretion may possibly contribute to the renal clearance of trimetrexate. Renal clearance has been associated with urine flow, suggesting the possibility of tubular reabsorption as well.
- The Vdss of trimetrexate in cancer patients after single-dose administration and for whom plasma concentrations were obtained for 72 hours was 36.9 ± 17.6 L/m2 (n=23) and 0.62 ± 0.24 L/kg (n=14). Following a constant infusion of trimetrexate for five days, Vdss was 32.8 ± 16.6 L/m2. The volume of the central compartment has been estimated as 0.17 ± 0.08 L/kg and 4.0 ± 2.9 L/m2.
- There have been inconsistencies in the reporting of trimetrexate protein binding. The in vitro plasma protein binding of trimetrexate using ultrafiltration is approximately 95% over the concentration range of 18.75 to 1000 ng/mL. There is a suggestion of capacity limited binding (saturable binding) at concentrations greater than about 1000 ng/mL, with free fraction progressively increasing to about 9.3% as concentration is increased to 15 μg/mL. Other reports have declared trimetrexate to be greater than 98% bound at concentrations of 0.1 to 10 μg/mL; however, specific free fractions were not stated. The free fraction of trimetrexate also has been reported to be about 15 to 16% at a concentration of 60 ng/mL, increasing to about 20% at a trimetrexate concentration of 6 μg/mL.
- Trimetrexate metabolism in man has not been characterized. Preclinical data strongly suggest that the major metabolic pathway is oxidative O-demethylation, followed by conjugation to either glucuronide or the sulfate. N-demethylation and oxidation is a related minor pathway. Preliminary findings in humans indicate the presence of a glucuronide conjugate with DHFR inhibition and a demethylated metabolite in urine.
- The presence of metabolite(s) in human plasma following the administration of trimetrexate is suggested by the differences seen in trimetrexate plasma concentrations when measured by HPLC and a nonspecific DHFR inhibition assay. The profiles are similar initially, but diverge with time; concentrations determined by DHFR being higher than those determined by HPLC. This suggests the presence of one or more metabolites with DHFR inhibition activity. After intravenous administration of trimetrexate to humans, urinary recovery averaged about 40%, using a DHFR assay, in comparison to 10% urinary recovery as determined by HPLC, suggesting the presence of one or more metabolites that retain inhibitory activity against DHFR. Fecal recovery of trimetrexate over 48 hours after intravenous administration ranged from 0.09 to 7.6% of the dose as determined by DHFR inhibition and 0.02 to 5.2% of the dose as determined by HPLC.
- The pharmacokinetics of trimetrexate have not been determined in patients with renal insufficiency or hepatic dysfunction.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Trimetrexate in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Trimetrexate in the drug label.
How Supplied
- Trimetrexate is supplied as a sterile lyophilized powder in either 5 mL or 30 mL vials. Each 5 mL vial contains trimetrexate glucuronate equivalent to 25 mg of trimetrexate. Each 30 mL vial contains trimetrexate glucuronate equivalent to 200 mg of trimetrexate. The 5 mL vials are packaged and available in two market presentations as listed below:
- 10 Pack - 10 vials in a white chip-board carton (NDC 58178-020-10)
- 50 Pack - 2 trays of 25 vials per shrink-wrapped tray (NDC 58178-020-50)
- The 30 mL vials are packaged and available as listed below:
- Single Pack - 1 vial (NDC 58178-021-01)
Storage
Store at controlled room temperature 20° to 25°C (68° to 77°F). Protect from exposure to light.
Images
Drug Images
{{#ask: Page Name::Trimetrexate |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Trimetrexate |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Patient Counseling Information of Trimetrexate in the drug label.
Precautions with Alcohol
- Alcohol-Trimetrexate interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Neutrexin®[1]
Look-Alike Drug Names
There is limited information regarding Trimetrexate Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Trimetrexate |Label Name=DailyMed - NEUTREXIN- trimetrexate glucuronate injection, powder, lyophilized, for solution .png
}}



