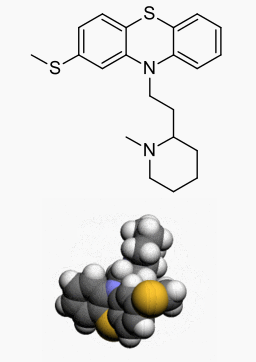
Thioridazine hydrochloride
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Gloria Picoy [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
Prolong the QTc interval and Increased Mortality in Elderly Patients with Dementia-Related Psychosis
See full prescribing information for complete Boxed Warning.
Thioridazine has been shown to prolong the QTc interval in a dose related manner, and drugs with this potential, including thioridazine, have been associated with Torsades de pointes type arrhythmias and sudden death. Due to its potential for significant, possibly life threatening, proarrhythmic effects, thioridazine should be reserved for use in the treatment of schizophrenic patients who fail to show an acceptable response to adequate courses of treatment with other antipsychotic drugs, either because of insufficient effectiveness or the inability to achieve an effective dose due to intolerable adverse effects from those drugs.
Increased Mortality in Elderly Patients with Dementia-Related Psychosis |
Overview
Thioridazine hydrochloride is an antipsychotic that is FDA approved for the treatment of schizophrenic patients who fail to respond adequately to treatment with other antipsychotic drugs. There is a Black Box Warning for this drug as shown here. Common adverse reactions include hypotension, orthostatic hypotension, diminished sweating, constipation, xerostomia, akathisia, dizziness, dystonia, extrapyramidal disease, parkinsonian, somnolence, tardive dyskinesia, blurred vision, epithelial keratopathy, retinitis pigmentosa, urinary retention and nasal congestion.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Thioridazine hydrochloride is indicated for the management of schizophrenic patients who fail to respond adequately to treatment with other antipsychotic drugs.
- Dosage: 50 to 100 mg three times a day, with a gradual increment to a maximum of 800 mg daily if necessary.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Thioridazine hydrochloride in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Thioridazine hydrochloride in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Thioridazine hydrochloride is indicated for the management of schizophrenic patients who fail to respond adequately to treatment with other antipsychotic drugs.
- Dosage:
- The recommended initial dose is 0.5 mg/kg/day given in divided doses.
- Dosage may be increased gradually until optimum therapeutic effect is obtained or the maximum dose of 3 mg/kg/day has been reached.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Thioridazine hydrochloride in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Thioridazine hydrochloride in pediatric patients.
Contraindications
Thioridazine hydrochloride tablet use should be avoided in combination with other drugs that are known to prolong the QTc interval and in patients with congenital long QT syndrome or a history of cardiac arrhythmias.
Reduced cytochrome P450 2D6 isozyme activity drugs that inhibit this isozyme (e.g., fluoxetine and paroxetine) and certain other drugs (e.g., fluvoxamine, propranolol, and pindolol) appear to appreciably inhibit the metabolism of thioridazine. The resulting elevated levels of thioridazine would be expected to augment the prolongation of the QTc interval associated with thioridazine and may increase the risk of serious, potentially fatal, cardiac arrhythmias, such as Torsades de pointes type arrhythmias. Such an increased risk may result also from the additive effect of coadministering thioridazine with other agents that prolong the QTc interval. Therefore, thioridazine is contraindicated with these drugs as well as in patients, comprising about 7% of the normal population, who are known to have a genetic defect leading to reduced levels of activity of P450 2D6.
In common with other phenothiazines, thioridazine is contraindicated in severe central nervous system depression or comatose states from any cause including drug induced central nervous system depression. It should also be noted that hypertensive or hypotensive heart disease of extreme degree is a contraindication of phenothiazine administration.
Warnings
|
Prolong the QTc interval and Increased Mortality in Elderly Patients with Dementia-Related Psychosis
See full prescribing information for complete Boxed Warning.
Thioridazine has been shown to prolong the QTc interval in a dose related manner, and drugs with this potential, including thioridazine, have been associated with Torsades de pointes type arrhythmias and sudden death. Due to its potential for significant, possibly life threatening, proarrhythmic effects, thioridazine should be reserved for use in the treatment of schizophrenic patients who fail to show an acceptable response to adequate courses of treatment with other antipsychotic drugs, either because of insufficient effectiveness or the inability to achieve an effective dose due to intolerable adverse effects from those drugs.
Increased Mortality in Elderly Patients with Dementia-Related Psychosis |
Potential for Proarrhythmic Effects
A crossover study in nine healthy males comparing single doses of thioridazine 10 mg and 50 mg with placebo demonstrated a dose related prolongation of the QTc interval. The mean maximum increase in QTc interval following the 50 mg dose was about 23 msec; greater prolongation may be observed in the clinical treatment of unscreened patients.
Prolongation of the QTc interval has been associated with the ability to cause Torsades de pointes type arrhythmias, a potentially fatal polymorphic ventricular tachycardia, and sudden death. There are several published case reports of Torsades de pointes and sudden death associated with thioridazine treatment. A causal relationship between these events and thioridazine therapy has not been established but, given the ability of thioridazine to prolong the QTc interval, such a relationship is possible.
Certain circumstances may increase the risk of Torsades de pointes and/or sudden death in association with the use of drugs that prolong the QTc interval, including 1) bradycardia, 2) hypokalemia, 3) concomitant use of other drugs that prolong the QTc interval, 4) presence of congenital prolongation of the QT interval, and 5) for thioridazine in particular, its use in patients with reduced activity of P450 2D6 or its coadministration with drugs that may inhibit P450 2D6 or by some other mechanism interfere with the clearance of thioridazine.
It is recommended that patients being considered for thioridazine treatment have a baseline ECG performed and serum potassium levels measured. Serum potassium should be normalized before initiating treatment and patients with a QTc interval greater than 450 msec should not receive thioridazine treatment. It may also be useful to periodically monitor ECG's and serum potassium during thioridazine treatment, especially during a period of dose adjustment. Thioridazine should be discontinued in patients who are found to have a QTc interval over 500 msec.
Patients taking thioridazine who experience symptoms that may be associated with the occurrence of Torsades de pointes (e.g., dizziness, palpitations, or syncope) may warrant further cardiac evaluation; in particular, Holter monitoring should be considered.
Tardive Dyskinesia
Tardive dyskinesia, a syndrome consisting of potentially irreversible, involuntary, dyskinetic movements may develop in patients treated with antipsychotic drugs. Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to rely upon prevalence estimates to predict, at the inception of antipsychotic treatment, which patients are likely to develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown.
Both the risk of developing the syndrome and the likelihood that it will become irreversible are believed to increase as the duration of treatment and the total cumulative dose of antipsychotic drugs administered to the patient increase. However, the syndrome can develop, although much less commonly, after relatively brief treatment periods at low doses.
There is no known treatment for established cases of tardive dyskinesia, although the syndrome may remit, partially or completely, if antipsychotic treatment is withdrawn. Antipsychotic treatment itself, however, may suppress (or partially suppress) the signs and symptoms of the syndrome and thereby may possibly mask the underlying disease process. The effect that symptomatic suppression has upon the long-term course of the syndrome is unknown.
Given these considerations, antipsychotics should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that, 1) is known to respond to antipsychotic drugs, and, 2) for whom alternative, equally effective, but potentially less harmful treatments are not available or appropriate. In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a satisfactory clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive dyskinesia appear in a patient on antipsychotics, drug discontinuation should be considered. However, some patients may require treatment despite the presence of the syndrome.
It has been suggested in regard to phenothiazines in general, that people who have demonstrated a hypersensitivity reaction (e.g., blood dyscrasias, jaundice) to one may be more prone to demonstrate a reaction to others. Attention should be paid to the fact that phenothiazines are capable of potentiating central nervous system depressants (e.g., anesthetics, opiates, alcohol, etc.) as well as atropine and phosphorus insecticides. Physicians should carefully consider benefit versus risk when treating less severe disorders.
Reproductive studies in animals and clinical experience to date have failed to show a teratogenic effect with thioridazine. However, in view of the desirability of keeping the administration of all drugs to a minimum during pregnancy, thioridazine should be given only when the benefits derived from treatment exceed the possible risks to mother and fetus.
Neuroleptic Malignant Syndrome (NMS)
A potentially fatal symptom complex sometimes referred to as Neuroleptic Malignant Syndrome (NMS) has been reported in association with antipsychotic drugs. Clinical manifestations of NMS are hyperpyrexia, muscle rigidity, altered mental status, and evidence of autonomic instability (irregular pulse or blood pressure, tachycardia, diaphoresis, and cardiac dysrhythmias).
The diagnostic evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to identify cases where the clinical presentation includes both serious medical illness (e.g., pneumonia, systemic infection, etc.) and untreated or inadequately treated extrapyramidal signs and symptoms (EPS). Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system (CNS) pathology.
The management of NMS should include, 1) immediate discontinuation of antipsychotic drugs and other drugs not essential to concurrent therapy, 2) intensive symptomatic treatment and medical monitoring, and 3) treatment of any concomitant serious medical problems for which specific treatments are available. There is no general agreement about specific pharmacological treatment regimens for uncomplicated NMS.
If a patient requires antipsychotic drug treatment after recovery from NMS, the potential reintroduction of drug therapy should be carefully considered. The patient should be carefully monitored, since recurrences of NMS have been reported.
Central Nervous System Depressants
As in the case of other phenothiazines, thioridazine is capable of potentiating central nervous system depressants (e.g., alcohol, anesthetics, barbiturates, narcotics, opiates, other psychoactive drugs, etc.) as well as atropine and phosphorus insecticides. Severe respiratory depression and respiratory arrest have been reported when a patient was given a phenothiazine and a concomitant high dose of a barbiturate.
Leukopenia, Neutropenia and Agranulocytosis
In clinical trial and post-marketing experience, events of leukopenia/neutropenia and agranulocytosis have been reported temporally related to antipsychotic agents.
Possible risk factors for leukopenia/neutropenia include preexisting low white blood cell count (WBC) and history of drug induced leukopenia/neutropenia. Patients with a preexisting low WBC or a history of drug induced leukopenia/neutropenia should have their complete blood count (CBC) monitored frequently during the first few months of therapy and should discontinue thioridazine hydrochloride tablets, USP at the first sign of a decline in WBC in the absence of other causative factors.
Patients with neutropenia should be carefully monitored for fever or other symptoms or signs of infection and treated promptly if such symptoms occur. Patients with severe neutropenia (absolute neutrophil count < 1000 mm3) should discontinue thioridazine hydrochloride tablets, USP and have their WBC followed until recovery.
Adverse Reactions
Clinical Trials Experience
In the recommended dosage ranges with thioridazine hydrochloride most side effects are mild and transient.
Central Nervous System
Drowsiness may be encountered on occasion, especially where large doses are given early in treatment. Generally, this effect tends to subside with continued therapy or a reduction in dosage. Pseudoparkinsonism and other extrapyramidal symptoms may occur but are infrequent. Nocturnal confusion, hyperactivity, lethargy, psychotic reactions, restlessness, and headache have been reported but are extremely rare.
Autonomic Nervous System
Dryness of mouth, blurred vision, constipation, nausea, vomiting, diarrhea, nasal stuffiness, and pallor have been seen.
Endocrine System
Galactorrhea, breast engorgement, amenorrhea, inhibition of ejaculation, and peripheral edema have been described.
Skin
Dermatitis and skin eruptions of the urticarial type have been observed infrequently. Photosensitivity is extremely rare.
Cardiovascular System
Thioridazine produces a dose related prolongation of the QTc interval, which is associated with the ability to cause Torsades de pointes type arrhythmias, a potentially fatal polymorphic ventricular tachycardia, and sudden death. Both Torsades de pointes type arrhythmias and sudden death have been reported in association with thioridazine. A causal relationship between these events and thioridazine therapy has not been established but, given the ability of thioridazine to prolong the QTc interval, such a relationship is possible. Other ECG changes have been reported.
Other
Rare cases described as parotid swelling have been reported following administration of thioridazine.
Postmarketing Experience
Drug Interactions
Reduced cytochrome P450 2D6 isozyme activity, drugs which inhibit this isozyme (e.g., fluoxetine and paroxetine), and certain other drugs (e.g., fluvoxamine, propranolol, and pindolol) appear to appreciably inhibit the metabolism of thioridazine. The resulting elevated levels of thioridazine would be expected to augment the prolongation of the QTc interval associated with thioridazine and may increase the risk of serious, potentially fatal, cardiac arrhythmias, such as Torsades de pointes type arrhythmias. Such an increased risk may result also from the additive effect of coadministering thioridazine with other agents that prolong the QTc interval. Therefore, thioridazine is contraindicated with these drugs as well as in patients, comprising about 7% of the normal population, who are known to have a genetic defect leading to reduced levels of activity of P450 2D6.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): Neonates exposed to antipsychotic drugs, during the third trimester of pregnancy are at risk for extrapyramidal and/or withdrawal symptoms following delivery. There have been reports of agitation, hypertonia, hypotonia, tremor, somnolence, respiratory distress and feeding disorder in these neonates. These complications have varied in severity; while in some cases symptoms have been self-limited, in other cases neonates have required intensive care unit support and prolonged hospitalization.
Thioridazine hydrochloride should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Pregnancy Category (AUS): C
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Thioridazine hydrochloride in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Thioridazine hydrochloride during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Thioridazine hydrochloride in women who are nursing.
Pediatric Use
For pediatric patients with schizophrenia who are unresponsive to other agents, the recommended initial dose is 0.5 mg/kg/day given in divided doses. Dosage may be increased gradually until optimum therapeutic effect is obtained or the maximum dose of 3 mg/kg/day has been reached.
Geriatic Use
There is no FDA guidance on the use of Thioridazine hydrochloride in geriatric settings.
Gender
There is no FDA guidance on the use of Thioridazine hydrochloride with respect to specific gender populations.
Race
There is no FDA guidance on the use of Thioridazine hydrochloride with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Thioridazine hydrochloride in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Thioridazine hydrochloride in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Thioridazine hydrochloride in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Thioridazine hydrochloride in patients who are immunocompromised.
Administration and Monitoring
Administration
Oral
Monitoring
There is limited information regarding Thioridazine hydrochloride Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Thioridazine hydrochloride and IV administrations.
Overdosage
Many of the symptoms observed are extensions of the side effects described under ADVERSE REACTIONS. Thioridazine can be toxic in overdose, with cardiac toxicity being of particular concern. Frequent ECG and vital sign monitoring of overdosed patients is recommended. Observation for several days may be required because of the risk of delayed effects.
Signs and Symptoms
Effects and clinical complications of acute overdose involving phenothiazines may include:
- Cardiovascular: Cardiac arrhythmias, hypotension, shock, ECG changes, increased QT and PR intervals, non-specific ST and T wave changes, bradycardia, sinus tachycardia, atrioventricular block, ventricular tachycardia, ventricular fibrillation, Torsades de pointes, myocardial depression.
- Central Nervous System: Sedation, extrapyramidal effects, confusion, agitation, hypothermia, hyperthermia, restlessness, seizures, areflexia, coma.
- Autonomic Nervous System: Mydriasis, miosis, dry skin, dry mouth, nasal congestion, urinary retention, blurred vision.
- Respiratory: Respiratory depression, apnea, pulmonary edema.
- Gastrointestinal: Hypomotility, constipation, ileus.
- Renal: Oliguria, uremia.
Toxic dose and blood concentration ranges for the phenothiazines have not been firmly established. It has been suggested that the toxic blood concentration range for thioridazine begins at 1 mg/dL, and 2 to 8 mg/dL is the lethal concentration range.
Treatment
An airway must be established and maintained. Adequate oxygenation and ventilation must be ensured.
Cardiovascular monitoring should commence immediately and should include continuous electrocardiographic monitoring to detect possible arrhythmias. Treatment may include one or more of the following therapeutic interventions: correction of electrolyte abnormalities and acid-base balance, lidocaine, phenytoin, isoproterenol, ventricular pacing, and defibrillation. Disopyramide, procainamide, and quinidine may produce additive QT-prolonging effects when administered to patients with acute overdosage of thioridazine and should be avoided (see WARNINGS and CONTRAINDICATIONS). Caution must be exercised when administering lidocaine, as it may increase the risk of developing seizures.
Treatment of hypotension may require intravenous fluids and vasopressors. Phenylephrine, levarterenol, or metaraminol are the appropriate pressor agents for use in the management of refractory hypotension. The potent α adrenergic blocking properties of the phenothiazines makes the use of vasopressors with mixed α and β adrenergic agonist properties inappropriate, including epinephrine and dopamine. Paradoxical vasodilation may result. In addition, it is reasonable to expect that the α adrenergic-blocking properties of bretylium might be additive to those of thioridazine, resulting in problematic hypotension.
In managing overdosage, the physician should always consider the possibility of multiple drug involvement. Gastric lavage and repeated doses of activated charcoal should be considered. Induction of emesis is less preferable to gastric lavage because of the risk of dystonia and the potential for aspiration of vomitus. Emesis should not be induced in patients expected to deteriorate rapidly, or those with impaired consciousness.
Acute extrapyramidal symptoms may be treated with diphenhydramine hydrochloride or benztropine mesylate.
Avoid the use of barbiturates when treating seizures, as they may potentiate phenothiazine-induced respiratory depression.
Forced diuresis, hemoperfusion, hemodialysis and manipulation of urine pH are of unlikely benefit in the treatment of phenothiazine overdose due to their large volume of distribution and extensive plasma protein binding.
Pharmacology
Mechanism of Action
The basic pharmacological activity of thioridazine is similar to that of other phenothiazines, but is associated with minimal extrapyramidal stimulation.
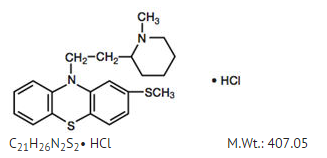
Structure
Thioridazine hydrochloride is 2-methylmercapto-10-[2-(N-methyl-2-piperidyl) ethyl] phenothiazine. Its structural formula, molecular weight and molecular formula are:
Pharmacodynamics
However, thioridazine has been shown to prolong the QTc interval in a dose dependent fashion. This effect may increase the risk of serious, potentially fatal, ventricular arrhythmias, such as Torsades de pointes type arrhythmias. Due to this risk, thioridazine is indicated only for schizophrenic patients who have not been responsive to or cannot tolerate other antipsychotic agents (see WARNINGS and CONTRAINDICATIONS). However, the prescriber should be aware that thioridazine has not been systematically evaluated in controlled trials in treatment refractory schizophrenic patients and its efficacy in such patients is unknown.
Pharmacokinetics
There is limited information regarding Thioridazine hydrochloride Pharmacokinetics in the drug label.
Nonclinical Toxicology
There is limited information regarding Thioridazine hydrochloride Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Thioridazine hydrochloride Clinical Studies in the drug label.
How Supplied
- Thioridazine hydrochloride tablets 10 mg:
- Bottles of 100 (NDC 53489-148-01)
- Bottles of 1000 (NDC 53489-148-10)
- Thioridazine hydrochloride tablets 25 mg:
- Bottles of 100 (NDC 53489-149-01)
- Bottles of 1000 (NDC 53489-149-10)
- Thioridazine hydrochloride tablets 50 mg:
- Bottles of 100 (NDC 53489-150-01)
- Bottles of 1000 (NDC 53489-150-10)
- Thioridazine hydrochloride tablets 100 mg:
- Bottles of 20 (NDC 53489-500-60)
- Bottles of 50 (NDC 53489-500-02)
- Bottles of 60 (NDC 53489-500-06)
- Bottles of 100 (NDC 53489-500-01)
- Bottles of 250 (NDC 53489-500-03)
- Bottles of 500 (NDC 53489-500-05)
- Bottles of 1000 (NDC 53489-500-10)
Storage
Store at 20° to 25°C (68° to 77°F).
Images
Drug Images
{{#ask: Page Name::Thioridazine hydrochloride |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
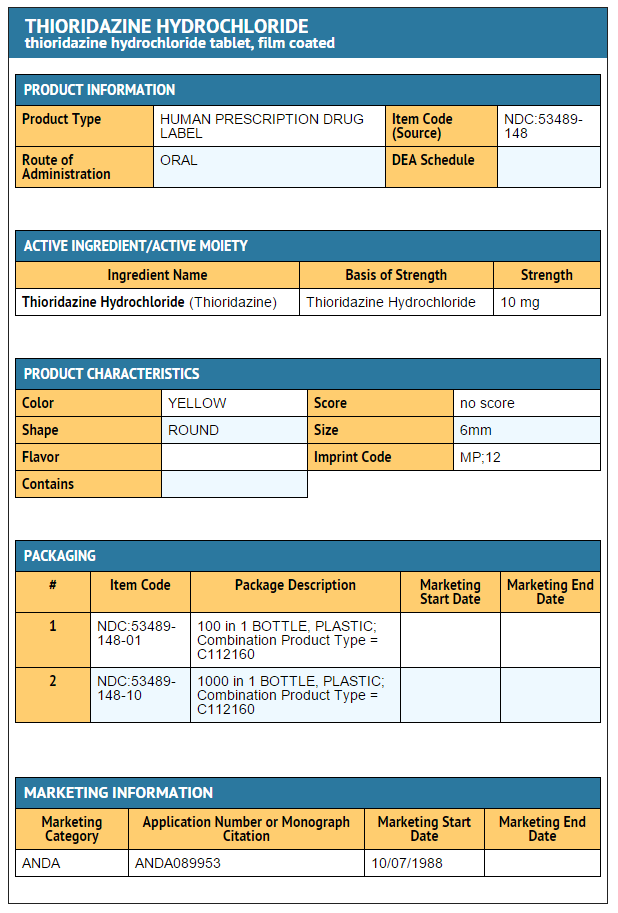
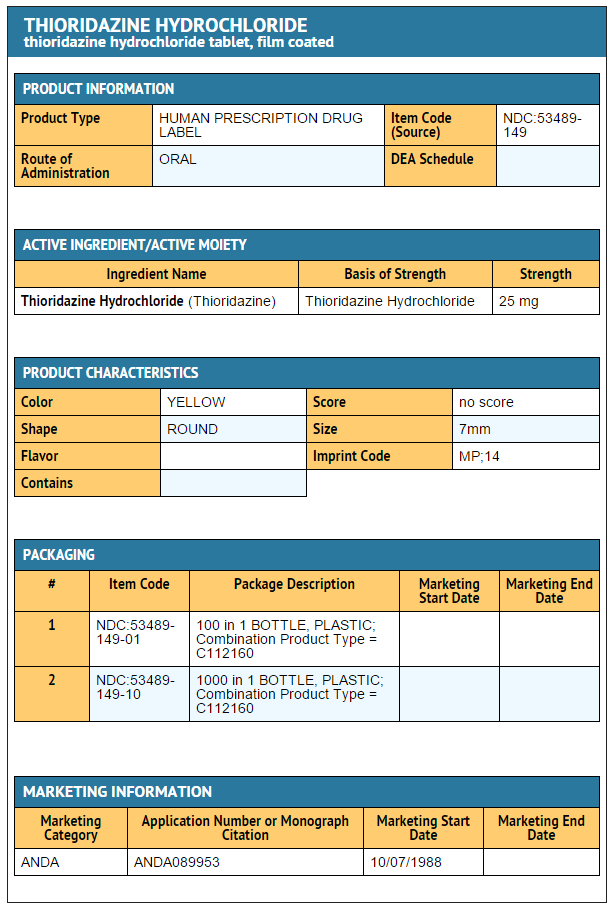
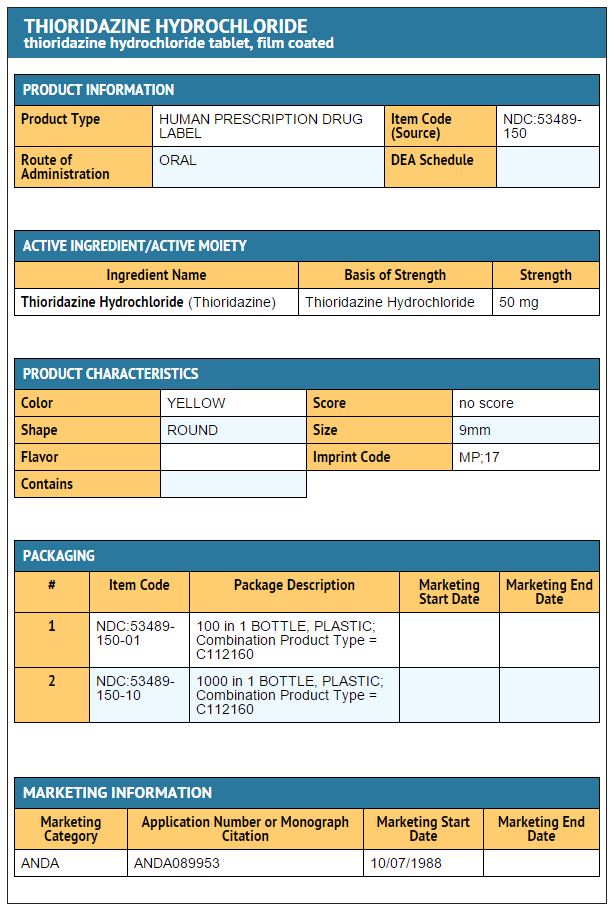
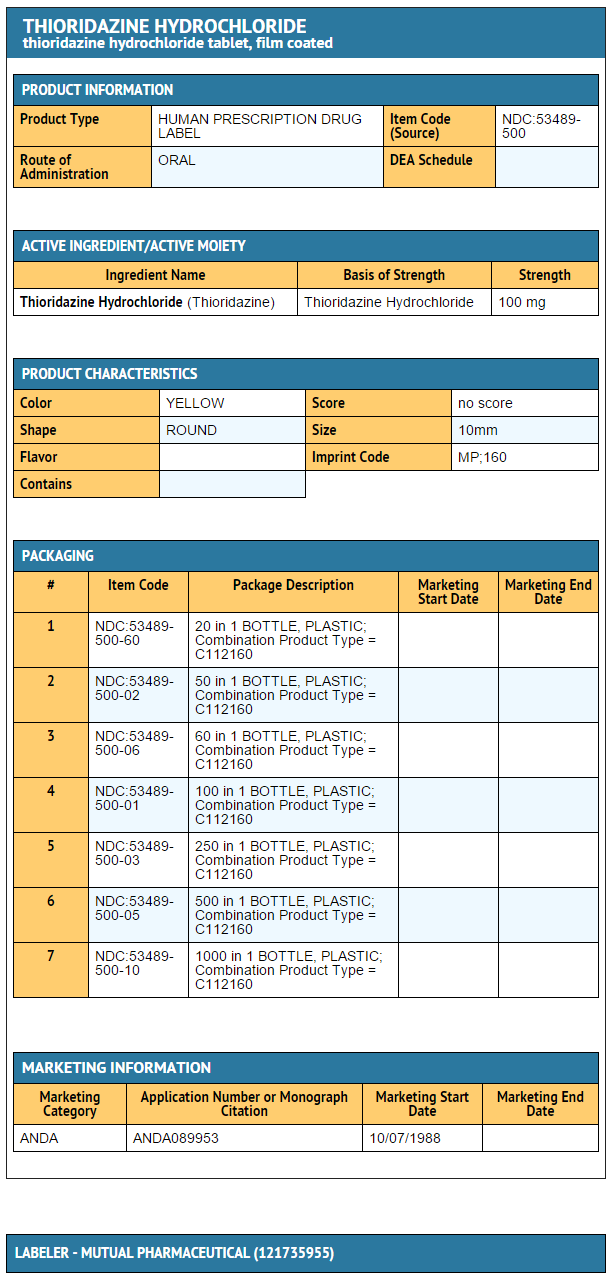
{{#ask: Label Page::Thioridazine hydrochloride |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Patients should be informed that thioridazine has been associated with potentially fatal heart rhythm disturbances. The risk of such events may be increased when certain drugs are given together with thioridazine. Therefore, patients should inform the prescriber that they are receiving thioridazine treatment before taking any new medication.
Given the likelihood that some patients exposed chronically to antipsychotics will develop tardive dyskinesia, it is advised that all patients in whom chronic use is contemplated be given, if possible, full information about this risk. The decision to inform patients and/or their guardians must obviously take into account the clinical circumstances and the competency of the patient to understand the information provided.
Precautions with Alcohol
Alcohol-Thioridazine hydrochloride interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Mellaril [2]
Look-Alike Drug Names
There is limited information regarding Thioridazine hydrochloride Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Shvartsburd, A; Sajadi, C; Morton, V; Mirabi, M; Gordon, J; Smith, RC (August 1984). "Blood levels of haloperidol and thioridazine during maintenance neuroleptic treatment of schizophrenic outpatients". Journal of Clinical Psychopharmacology. 4 (4): 194–198. doi:10.1097/00004714-198408000-00004. PMID 6470190.
- ↑ "FDA LABEL: THIORIDAZINE HYDROCHLORIDE- thioridazine hydrochloride tablet, film coated".
{{#subobject:
|Label Page=Thioridazine hydrochloride |Label Name=Thioridazine hydrochloride 10 mg.png
}}
{{#subobject:
|Label Page=Thioridazine hydrochloride |Label Name=Thioridazine hydrochloride 25 mg.png
}}
{{#subobject:
|Label Page=Thioridazine hydrochloride |Label Name=Thioridazine hydrochloride 50 mg.png
}}
{{#subobject:
|Label Page=Thioridazine hydrochloride |Label Name=Thioridazine hydrochloride 100 mg.png
}}