Temocillin
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
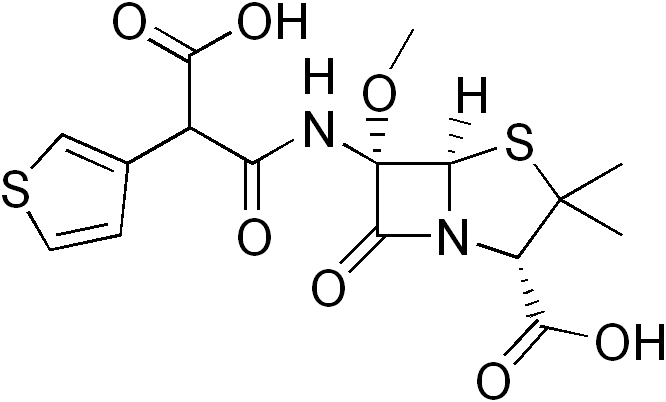
| Formula | C16H18N2O7S2 |
| Molar mass | 414.453 g/mol |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
|
WikiDoc Resources for Temocillin |
|
Articles |
|---|
|
Most recent articles on Temocillin |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Temocillin at Clinical Trials.gov Clinical Trials on Temocillin at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Temocillin
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Temocillin Discussion groups on Temocillin Patient Handouts on Temocillin Directions to Hospitals Treating Temocillin Risk calculators and risk factors for Temocillin
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Temocillin |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Temocillin is a β-lactamase-resistant penicillin[1][2] introduced by Beecham, marketed by Eumedica Pharmaceuticals as Negaban. It is used primarily for the treatment of multiple drug-resistant, Gram-negative bacteria. It is a carboxypenicillin.[3]
Pharmacology
Temocillin is a β-lactamase-resistant penicillin. It is not active against Gram-positive bacteria or bacteria with altered penicillin-binding proteins.
It is normally active against Moraxella catarrhalis, Brucella abortus, Burkholderia cepacia, Citrobacter species, Escherichia coli, Haemophilus influenzae, Klebsiella pneumoniae, Pasteurella multocida, Proteus mirabilis, Salmonella typhimurium, and Yersinia enterocolitica. It is also active against some Enterobacter species, Morganella morganii, and Serratia species. Temocillin has no useful activity against Acinetobacter species or Pseudomonas aeruginosa.
Its primary use is against Enterobacteriaceae, and in particular against strains producing extended-spectrum β-lactamase or AmpC β-lactamase.[4]
Dosage
The common dose is 2 g intravenously every 12 hours. Theoretical reasons exist for giving temocillin as a continuous intravenous infusion in severe disease:[5] a single loading dose of 2 g is given intravenously followed by a 4-g infusion over 24 hours. Temocillin for intravenous injection is diluted in 20 ml of sterile water; it is diluted in less than 2.7 ml of sterile water when being prepared for intramuscular injection; the continuous infusion is diluted in 48 ml of sterile water for ease of administration (1 ml per half hour). To reduce pain, the intramuscular injection may be made up using sterile 1% lignocaine instead of sterile water.
Temocillin may be given to patients with impaired renal function. No adjustment needs to be made to the dose in mild to moderate renal impairment (creatinine clearance greater than 30 ml/min). Temocillin is cleared by haemodialysis, so in dialysis patients, the dose should be given after dialysis.
No oral preparation of temocillin is licensed.
Adverse effects
The undesirable effects of temocillin are those of any β-lactam antibiotic. In particular, it has been associated with angioedema and anaphylaxis in penicillin-allergic patients. Animal studies have not shown any induction of Clostridium difficile infection.[6] As with any other penicillin, convulsions can occur if very high doses are given.
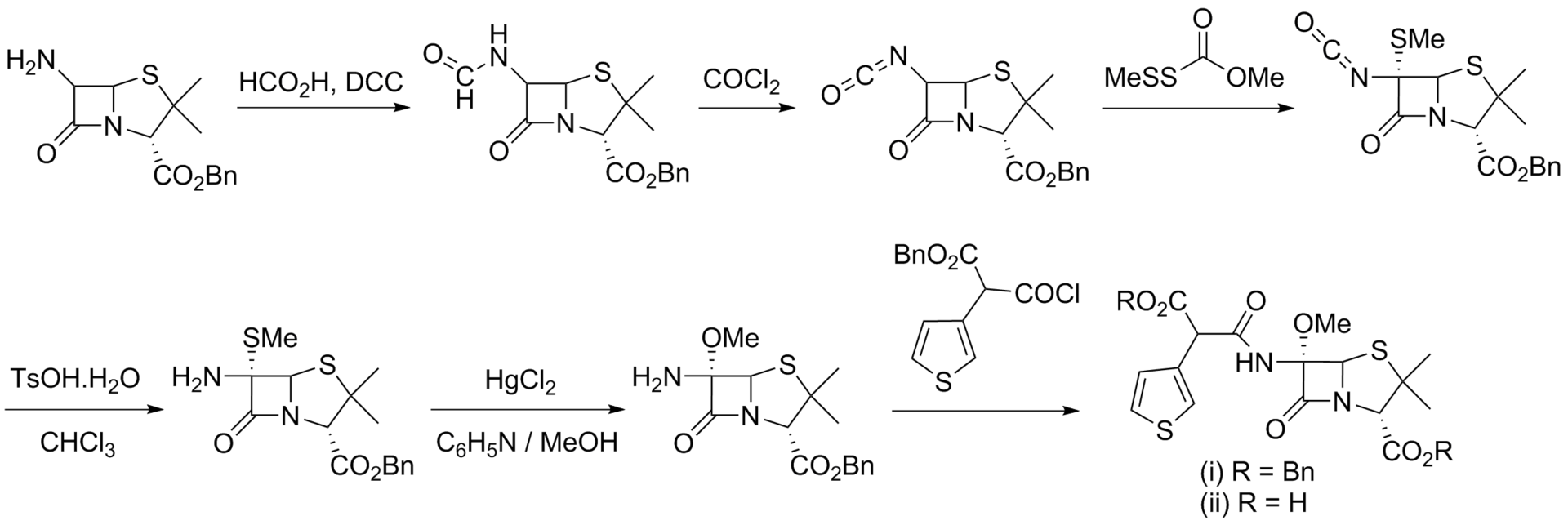
Synthesis
References
- ↑ Andrews JM, Jevons G, Walker R, Ashby J, Fraise AP (July 2007). "Temocillin susceptibility by BSAC methodology". J. Antimicrob. Chemother. 60 (1): 185–7. doi:10.1093/jac/dkm179. PMID 17550891.
- ↑ Van Landuyt HW, Pyckavet M, Lambert A, Boelaert J (October 1982). "In vitro activity of temocillin (BRL 17421), a novel beta-lactam antibiotic". Antimicrob. Agents Chemother. 22 (4): 535–40. doi:10.1128/aac.22.4.535. PMC 183789. PMID 7181470.
- ↑ Chanal M, Sirot J, Cluzel M, Joly B, Glanddier Y (June 1983). "[In vitro study of the bacteriostatic and bactericidal activity of temocillin (BRL 17421)]". Pathol. Biol. (in French). 31 (6): 467–70. PMID 6348653.
- ↑ Livermore DM et al. (2006) Activity of temocillin vs. prevalent ESBL- and AmpC-producing Enterobacteriaceae from SE England. J Antimicrob Chemother. 2006 May;57(5):1012-4.
- ↑ De Jongh R et al. (2008) Continuous versus intermittent infusion of temocillin, a directed spectrum penicillin for intensive care patients with nosocomial pneumonia: stability, compatibility, population pharmacokinetic studies and breakpoint selection. J Antimicrob Chemother. 2008 Feb;61(2):382-8.
- ↑ Boon RJ; et al. (1985). "Studies with temocillin in a hamster model of antibiotic-associated colitis". Antimicrob Agents Chemother. 27 (6): 980–1. doi:10.1128/aac.27.6.980. PMC 180203. PMID 3875312.
- ↑ Template:Cite doi
Further reading
- Livermore DM, Tulkens PM (February 2009). "Temocillin revived". Journal of Antimicrobial Chemotherapy. 63 (2): 243–5. doi:10.1093/jac/dkn511. PMID 19095679.
- Pages with script errors
- CS1 maint: Multiple names: authors list
- CS1 maint: Unrecognized language
- CS1 maint: Explicit use of et al.
- Template:drugs.com link with non-standard subpage
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Chemical pages without DrugBank identifier
- Drugs with no legal status
- Drugboxes which contain changes to watched fields
- Beta-lactam antibiotics
- Thiophenes
- Drug