Pyrimethamine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Gloria Picoy [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Pyrimethamine is a folic acid antagonist that is FDA approved for the treatment of toxoplasmosis, acute malaria and for the prophylaxis of malaria. Common adverse reactions include rash.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Treatment of Toxoplasmosis
- Pyrimethamine is indicated for the treatment of toxoplasmosis when used conjointly with a sulfonamide, since synergism exists with this combination.
- Concurrent administration of folinic acid is strongly recommended in all patients.
- Starting dose is 50 to 75 mg of the drug daily, together with 1 to 4 g daily of a sulfonamide of the sulfapyrimidine type, e.g. sulfadoxine; for 1 to 3 weeks.
- The dosage may then be reduced to about one half that previously given for each drug and continued for an additional 4 to 5 weeks.
Treatment of Acute Malaria
- Pyrimethamine is also indicated for the treatment of acute malaria. It should not be used alone to treat acute malaria. Fast-acting schizonticides such as chloroquine or quinine are indicated and preferable for the treatment of acute malaria. However, conjoint use of pyrimethamine with a sulfonamide (e.g., sulfadoxine) will initiate transmission control and suppression of susceptible strains of plasmodia.
- Dosage: 25 mg daily for 2 days with a sulfonamide will initiate transmission control and suppression of non-falciparum malaria.
Chemoprophylaxis of Malaria
- Pyrimethamine is indicated for the chemoprophylaxis of malaria due to susceptible strains of plasmodia. However, resistance to pyrimethamine is prevalent worldwide. It is not suitable as a prophylactic agent for travelers to most areas.
- Dosage: 25 mg (1 tablet) once weekly
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Pyrimethamine in adult patients.
Non–Guideline-Supported Use
- Prophylaxis of Pneumocystis jiroveci pneumonia infection in HIV-infected adults
- Prophylaxis of Toxoplasma gondii encephalitis in adults and adolescents with human immunodeficiency virus
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Treatment of Toxoplasmosis
- Pyrimethamine is indicated for the treatment of toxoplasmosis when used conjointly with a sulfonamide, since synergism exists with this combination.
- Concurrent administration of folinic acid is strongly recommended in all patients.
- Dosage:
- 1 mg/kg/day divided into 2 equal daily doses for 2 to 4 days
- This dose may be reduced to one half and continued for approximately 1 month.
Treatment of Acute Malaria
- Pyrimethamine is also indicated for the treatment of acute malaria. It should not be used alone to treat acute malaria. Fast-acting schizonticides such as chloroquine or quinine are indicated and preferable for the treatment of acute malaria. However, conjoint use of pyrimethamine with a sulfonamide (e.g., sulfadoxine) will initiate transmission control and suppression of susceptible strains of plasmodia.
- Dosage for children 4 through 10 years old: may be given 25 mg daily for 2 days
Chemoprophylaxis of Malaria
- Pyrimethamine is indicated for the chemoprophylaxis of malaria due to susceptible strains of plasmodia. However, resistance to pyrimethamine is prevalent worldwide. It is not suitable as a prophylactic agent for travelers to most areas.
- Dosage:
- Pediatric patients over 10 years: 25 mg (1 tablet) once weekly
- Children 4 through 10 years: 12.5 mg (1/2 tablet) once weekly
- Infants and children under 4 years: 6.25 mg (1/4 tablet) once weekly.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Pyrimethamine in pediatric patients.
Non–Guideline-Supported Use
- Prophylaxis of Toxoplasma gondii encephalitis in adults and adolescents with human immunodeficiency virus.
Contraindications
- Use of pyrimethamine is contraindicated in patients with known hypersensitivity to pyrimethamine or to any component of the formulation.
- Use of the drug is also contraindicated in patients with documented megaloblastic anemia due to folate deficiency.
Warnings
- The dosage of pyrimethamine required for the treatment of toxoplasmosis is 10 to 20 times the recommended antimalaria dosage and approaches the toxic level. If signs of folate deficiency develop, reduce the dosage or discontinue the drug according to the response of the patient. Folinic acid (leucovorin) should be administered in a dosage of 5 to 15 mg daily (orally, IV, or IM) until normal hematopoiesis is restored.
- Data in 2 humans indicate that pyrimethamine may be carcinogenic; a 51-year-old female who developed chronic granulocytic leukemia after taking pyrimethamine for 2 years for toxoplasmosis and a 56-year-old patient who developed reticulum cell sarcoma after 14 months of pyrimethamine for toxoplasmosis.
- Pyrimethamine has been reported to produce a significant increase in the number of lung tumors in mice when given intraperitoneally at doses of 25 mg/kg.
- Pyrimethamine susceptible to adverse effects from an overdose. Deaths in pediatric patients have been reported after accidental ingestion.
Adverse Reactions
Clinical Trials Experience
Hypersensitivity reactions, occasionally severe (such as Stevens-Johnson syndrome, toxic epidermal necrolysis, erythema multiforme, and anaphylaxis), and hyperphenylalaninemia, can occur particularly when pyrimethamine is administered concomitantly with a sulfonamide. Consult the complete prescribing information for the relevant sulfonamide for sulfonamide-associated adverse events. With doses of pyrimethamine used for the treatment of toxoplasmosis, anorexia and vomiting may occur. Vomiting may be minimized by giving the medication with meals; it usually disappears promptly upon reduction of dosage. Doses used in toxoplasmosis may produce megaloblastic anemia, leukopenia, thrombocytopenia, pancytopenia, atrophic glossitis, hematuria, and disorders of cardiac rhythm. Hematologic effects, however, may also occur at low doses in certain individuals.
Pulmonary eosinophilia has been reported rarely.
Postmarketing Experience
There is limited information regarding Pyrimethamine Postmarketing Experience in the drug label.
Drug Interactions
- Pyrimethamine may be used with sulfonamides, quinine and other antimalarials, and with other antibiotics. However, the concomitant use of other antifolic drugs or agents associated with myelosuppression including sulfonamides or trimethoprim-sulfamethoxazole combinations, proguanil, zidovudine, or cytostatic agents (e.g., methotrexate), while the patient is receiving pyrimethamine, may increase the risk of bone marrow suppression.
- If signs of folate deficiency develop, pyrimethamine should be discontinued. Folinic acid (leucovorin) should be administered until normal hematopoiesis is restored.
- Mild hepatotoxicity has been reported in some patients when lorazepam and pyrimethamine were administered concomitantly.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): C Pyrimethamine has been shown to be teratogenic in rats when given in oral doses 7 times the human dose for chemoprophylaxis of malaria or 2.5 times the human dose for treatment of toxoplasmosis. At these doses in rats, there was a significant increase in abnormalities such as cleft palate, brachygnathia, oligodactyly, and microphthalmia. Pyrimethamine has also been shown to produce terata such as meningocele in hamsters and cleft palate in miniature pigs when given in oral doses 170 and 5 times the human dose, respectively, for chemoprophylaxis of malaria or for treatment of toxoplasmosis.
There are no adequate and well-controlled studies in pregnant women. pyrimethamine should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Concurrent administration of folinic acid is strongly recommended when used for the treatment of toxoplasmosis during pregnancy.
Pregnancy Category (AUS): B3
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Pyrimethamine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Pyrimethamine during labor and delivery.
Nursing Mothers
Pyrimethamine is excreted in human milk. Because of the potential for serious adverse reactions in nursing infants from pyrimethamine and from concurrent use of a sulfonamide with pyrimethamine for treatment of some patients with toxoplasmosis, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
Pediatric Use
There is no FDA guidance on the use of Pyrimethamine in pediatric settings.
Geriatic Use
Clinical studies of pyrimethamine did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experience has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Gender
There is no FDA guidance on the use of Pyrimethamine with respect to specific gender populations.
Race
There is no FDA guidance on the use of Pyrimethamine with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Pyrimethamine in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Pyrimethamine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Pyrimethamine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Pyrimethamine in patients who are immunocompromised.
Administration and Monitoring
Administration
Oral
Monitoring
In patients receiving high dosage, as for the treatment of toxoplasmosis, semiweekly blood counts, including platelet counts, should be performed.
IV Compatibility
There is limited information regarding the compatibility of Pyrimethamine and IV administrations.
Overdosage
Following the ingestion of 300 mg or more of pyrimethamine, gastrointestinal and/or central nervous system signs may be present, including convulsions. The initial symptoms are usually gastrointestinal and may include abdominal pain, nausea, severe and repeated vomiting, possibly including hematemesis. Central nervous system toxicity may be manifest by initial excitability, generalized and prolonged convulsions which may be followed by respiratory depression, circulatory collapse, and death within a few hours. Neurological symptoms appear rapidly (30 minutes to 2 hours after drug ingestion), suggesting that in gross overdosage pyrimethamine has a direct toxic effect on the central nervous system.
The fatal dose is variable, with the smallest reported fatal single dose being 375 mg. There are, however, reports of pediatric patients who have recovered after taking 375 to 625 mg.
There is no specific antidote to acute pyrimethamine poisoning. In the event of overdosage, symptomatic and supportive measures should be employed. Gastric lavage is recommended and is effective if carried out very soon after drug ingestion. Parenteral diazepam may be used to control convulsions. Folinic acid should be administered within 2 hours of drug ingestion to be most effective in counteracting the effects on the hematopoietic system. Due to the long half-life of pyrimethamine, daily monitoring of peripheral blood counts is recommended for up to several weeks after the overdose until normal hematologic values are restored.
Pharmacology
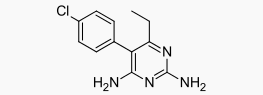
| |
Pyrimethamine
| |
| Systematic (IUPAC) name | |
| 5-(4-chlorophenyl)-6-ethyl- 2,4-pyrimidinediamine | |
| Identifiers | |
| CAS number | |
| ATC code | P01 Template:ATCvet (combinations) |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 248.71 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | well-absorbed |
| Protein binding | 87% |
| Metabolism | Hepatic |
| Half life | 96 hours |
| Excretion | Renal |
| Therapeutic considerations | |
| Pregnancy cat. | |
| Legal status |
Prescription Only (S4)(AU) ?(CA) POM(UK) [[Prescription drug|Template:Unicode-only]](US) |
| Routes | Oral |
Mechanism of Action
Binds to and reversibly inhibits the protozoal enzyme dihydrofolate reductase, selectively blocking conversion of dihydrofolic acid to its functional form, tetrahydrofolic acid. This depletes folate, an essential cofactor in the biosynthesis of nucleic acids, resulting in interference with protozoal nucleic acid and protein production.
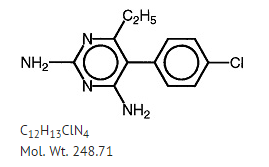
Structure
Pyrimethamine, known chemically as 5-(4-chlorophenyl)-6-ethyl-2,4-pyrimidinediamine, has the following structural formula:
Pharmacodynamics
Pyrimethamine is a folic acid antagonist and the rationale for its therapeutic action is based on the differential requirement between host and parasite for nucleic acid precursors involved in growth. This activity is highly selective against plasmodia and Toxoplasma gondii.
Pyrimethamine possesses blood schizonticidal and some tissue schizonticidal activity against malaria parasites of humans. However, the 4-amino-quinoline compounds are more effective against the erythrocytic schizonts. It does not destroy gametocytes, but arrests sporogony in the mosquito.
The action of pyrimethamine against Toxoplasma gondii is greatly enhanced when used in conjunction with sulfonamides. This was demonstrated by Eyles and Coleman1 in the treatment of experimental toxoplasmosis in the mouse. Jacobs et al2 demonstrated that combination of the 2 drugs effectively prevented the development of severe uveitis in most rabbits following the inoculation of the anterior chamber of the eye with toxoplasma.
Pharmacokinetics
Pyrimethamine is well absorbed with peak levels occurring between 2 to 6 hours following administration. It is eliminated slowly and has a plasma half-life of approximately 96 hours. Pyrimethamine is 87% bound to human plasma proteins.
Nonclinical Toxicology
Carcinogenesis
Data in 2 humans indicate that pyrimethamine may be carcinogenic; a 51-year-old female who developed chronic granulocytic leukemia after taking pyrimethamine for 2 years for toxoplasmosis and a 56-year-old patient who developed reticulum cell sarcoma after 14 months of pyrimethamine for toxoplasmosis.
Pyrimethamine has been reported to produce a significant increase in the number of lung tumors in mice when given intraperitoneally at doses of 25 mg/kg.
Mutagenesis
- Pyrimethamine has been shown to be nonmutagenic in the following in vitro assays: the Ames point mutation assay, the Rec assay, and the E. coli WP2 assay. It was positive in the L5178Y/TK +/- mouse lymphoma assay in the absence of exogenous metabolic activation.6 Human blood lymphocytes cultured in vitro had structural chromosome aberrations induced by pyrimethamine.
In vivo, chromosomes analyzed from the bone marrow of rats dosed with pyrimethamine showed an increased number of structural and numerical aberrations.
Clinical Studies
There is limited information regarding Pyrimethamine Clinical Studies in the drug label.
How Supplied
- Pyrimethamine 25 mg tablets
- Bottles of 100
- NDC 52054-330-10 and NDC 52054-330-95
Storage
Store at 15° to 25°C (59° to 77°F)
Images
Drug Images
{{#ask: Page Name::Pyrimethamine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
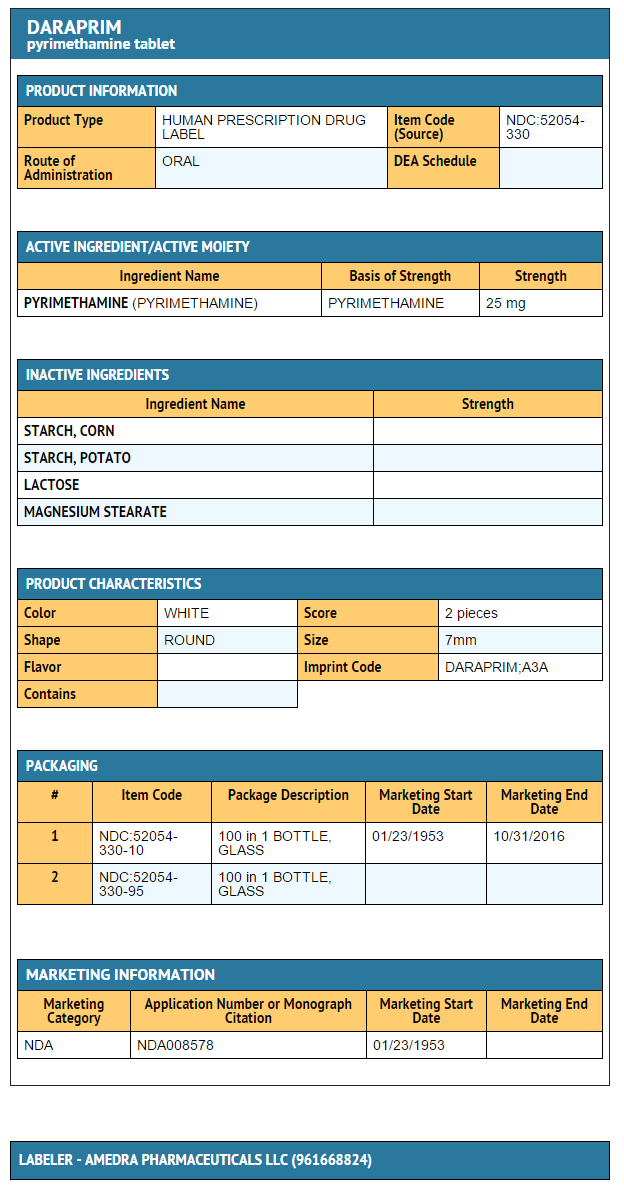
{{#ask: Label Page::Pyrimethamine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Patients should be warned that at the first appearance of a skin rash they should stop use of pyrimethamine and seek medical attention immediately. Patients should also be warned that the appearance of sore throat, pallor, purpura, or glossitis may be early indications of serious disorders which require treatment with pyrimethamine to be stopped and medical treatment to be sought.
- Women of childbearing potential who are taking pyrimethamine should be warned against becoming pregnant. Patients should be warned to keep pyrimethamine out of the reach of children. Patients should be advised not to exceed recommended doses. Patients should be warned that if anorexia and vomiting occur, they may be minimized by taking the drug with meals.
- Concurrent administration of folinic acid is strongly recommended when used for the treatment of toxoplasmosis in all patients.
Precautions with Alcohol
Alcohol-Pyrimethamine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Daraprim [1]
Look-Alike Drug Names
There is limited information regarding Pyrimethamine Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Pyrimethamine |Label Name=Pyrimethamine 25 mg.png
}}