Progabide
 | |
| Clinical data | |
|---|---|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 60% |
| Protein binding | 95% |
| Metabolism | Hepatic |
| Elimination half-life | 4 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C17H16ClFN2O2 |
| Molar mass | 334.772 g/mol |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
|
WikiDoc Resources for Progabide |
|
Articles |
|---|
|
Most recent articles on Progabide |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Progabide at Clinical Trials.gov Clinical Trials on Progabide at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Progabide
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Progabide Discussion groups on Progabide Directions to Hospitals Treating Progabide Risk calculators and risk factors for Progabide
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Progabide |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Progabide (INN) (trade name Gabrene, Sanofi-Aventis) is an analog and prodrug of gamma-aminobutyric acid used in the treatment of epilepsy. It has agonistic activity at the GABAA, GABAB, and GABAρ receptors.
Uses
Progabide is approved in France for either monotherapy or adjunctive use in the treatment of epilepsy—specifically, generalized tonic-clonic, myoclonic, partial, and Lennox-Gastaut syndrome seizures—in both children and adults.
Progabide has been investigated for many diseases besides epilepsy, including Parkinson's disease, schizophrenia, clinical depression, anxiety disorder and spasticity with various levels of success.
In 1987, Bartolini and colleagues reported progabide's actions on dopamine to be contradictory, decreasing dopamine release, dopamine receptor density and postsynaptic receptor responsivity to dopamine while reducing striatal cholinergic activity so as to increase dopaminergic effects.[1] Bartholini and colleagues concluded that it was this that caused Parkinson's patients in human clinical trials to either see an improvement in their Parkinson's with a worsening of L-dopa dyskinesia or an improvement in dyskinesia but with sometimes aggravated Parkinson's symptoms.[1] The cholinergic effect takes only a single injection to achieve in rats; when given with haloperidol, the development of tolerance to haloperidol's cataleptic effects did not develop.[2] It was hoped that this would be effective for tardive dyskinesia. However, Soares, Rathbone and Deeks wrote in the 2004 issue of The Cochrane Database of Systematic Reviews that "Any possible benefits are likely to be outweighed by the adverse effects associated with their [GABAergic agents'] use."[3]
In addition to being tested for antipsychotic-induced tardive dyskinesia, progabide was itself tested as an antipsychotic; as early as 1979, it was obvious that it was ineffective for psychosis.[4] While progabide may have been devoid of antipsychotic effects, it did have the effect in schizoaffective and hebephrenic patients of improving environmental responsiveness and social interactions.[5]
Synthesis
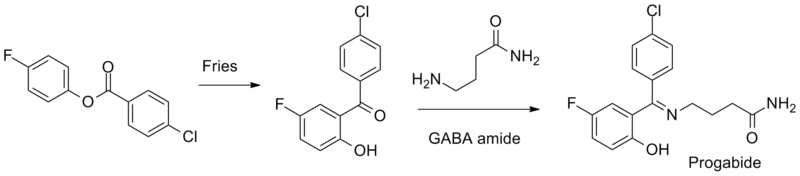
References
- ↑ 1.0 1.1 Bartholini G, Scatton B, Zivkovic B, Lloyd KG. "GABA receptor agonists and extrapyramidal motor function: therapeutic implications for Parkinson's disease." Advances in Neurology. 1987;45:79-83. PMID 3030072
- ↑ Bartholini G.; Scatton B; Zivkovic B. (1980). "Effect of the new gamma-aminobutyric acid agonist SL 76 002 on striatal acetylcholine: relation to neuroleptic-induced extrapyramidal alterations". Advances in Biochemical Psychopharmacology. 24: 207–13. PMID 6105775.
- ↑ Soares, K; Rathbone J; Deeks J. (18 October 2004). Soares-Weiser, Karla, ed. "Gamma-aminobutyric acid agonists for neuroleptic-induced tardive dyskinesia". The Cochrane Database of Systematic Reviews (4): CD000203. doi:10.1002/14651858.CD000203.pub2. PMID 15494993.
- ↑ Bartholini, G. (1979). "[Potential therapeutic activity of GABA-mimetic drugs in neuropsychiatry]". Schweizer Archiv fur Neurologie, Neurochirurgie und Psychiatrie = Archives suisses de neurologie, neurochirurgie et de psychiatrie. 125 (2): 265–9. PMID 45343. (French)
- ↑ Lloyd KG; Morselli PL; Depoortere H; Fournier V; Zivkovic B; Scatton B; Broekkamp C; Worms P; Bartholini G. (June 1983). "The potential use of GABA agonists in psychiatric disorders: evidence from studies with progabide in animal models and clinical trials". Pharmacology, Biochemistry, and Behavior. 18 (6): 957–66. doi:10.1016/S0091-3057(83)80021-5. PMID 6351106.
- Pages with script errors
- Drugs with non-standard legal status
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without KEGG source
- Drugboxes which contain changes to watched fields
- Anticonvulsants
- Phenols
- Organofluorides
- Organochlorides
- Drug