Phenylacetic acid
| Template:Chembox header| Phenylacetic acid | |
|---|---|
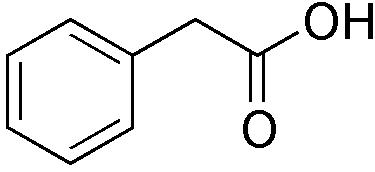
| |
| Systematic name | Phenylacetic acid |
| Chemical formula | C8H8O2 |
| Molecular mass | 136.15 g/mol |
| Density | 1.0809 g/cm3 |
| Melting point | 76-77 °C |
| Boiling point | 265.5 °C |
| CAS number | 103-82-2 |
| SMILES | c1ccccc1CC(=O)O |
| Template:Chembox header | Disclaimer and references | |
Please Take Over This Page and Apply to be Editor-In-Chief for this topic: There can be one or more than one Editor-In-Chief. You may also apply to be an Associate Editor-In-Chief of one of the subtopics below. Please mail us [1] to indicate your interest in serving either as an Editor-In-Chief of the entire topic or as an Associate Editor-In-Chief for a subtopic. Please be sure to attach your CV and or biographical sketch.
Phenylacetic acid (abr. PAA and synonyms are: α-toluic acid, benzeneacetic acid, alpha tolylic acid, 2-phenylacetic acid) is an organic compound containing a phenyl functional group and an acetic acid functional group. It is a white solid with a disagreeable odor. Because it is used in the illicit production of phenylacetone, it is subject to controls in the United States.
Significance
Phenylacetic acid has been found to be an active auxin (a type of plant hormone) molecule, predominantly found in fruits. However its effect is much weaker than the effect of the basic auxin molecule indole-3-acetic acid.
Phenylacetic acid is used in some perfumes, possessing a honey-like odour in low concentrations, and is also used in penicillin G production.
Its refractive index is 1.5025.
See also
External links
- Synthesis of Phenylacetic Acid
- Synthesis of Phenylacetic Acid (Rhodium mirror)
- MSDS for phenylacetic acid
de:Phenylessigsäure
lv:Feniletiķskābe
fi:Fenyyliasetaattihappo