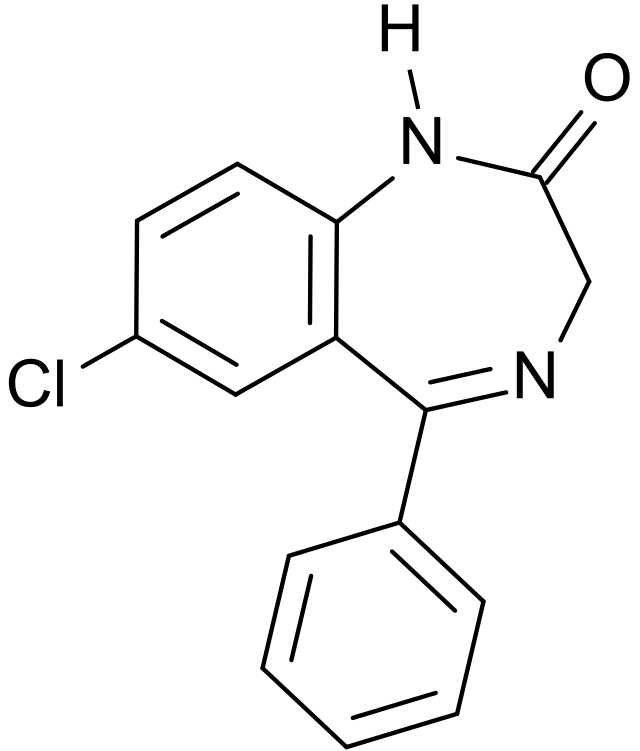
Nordazepam
 | |
 | |
| Clinical data | |
|---|---|
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | Hepatic |
| Elimination half-life | 50-120 hours |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C15H11ClN2O |
| Molar mass | 270.71 |
|
WikiDoc Resources for Nordazepam |
|
Articles |
|---|
|
Most recent articles on Nordazepam |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Nordazepam at Clinical Trials.gov Clinical Trials on Nordazepam at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Nordazepam
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Nordazepam Discussion groups on Nordazepam Patient Handouts on Nordazepam Directions to Hospitals Treating Nordazepam Risk calculators and risk factors for Nordazepam
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Nordazepam |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Nordazepam (marketed under brand names Stilny®, Madar®, Vegesan®, and Calmday®), also known as nordiazepam and desmethyldiazepam, is a 1,4-benzodiazepine derivative. Like other benzodiazepine derivatives, it has anticonvulsant, anxiolytic, muscle relaxant and sedative properties. However, it is used primarily in the treatment of anxiety. It is an active metabolite of diazepam, chlordiazepoxide, clorazepate, prazepam, and medazepam.[1]
Side effects
Common side effects of nordazepam include somnolence, which is more common in elderly patients and/or people on high dose regimens. Hypotonia, which is much less common, is also associated with high doses and/or old age.
Interactions
Benzodiazepines including N-desmethyldiazepam (nordazepam) may inhibit the glucuronidation of morphine leading to increased levels of and prolongation of the effects of morphine.[2]
Abuse
Nordazepam and other sedative hypnotic drugs are detected frequently in cases of people suspected of driving under the influence of drugs. Other benzodiazepines and zolpidem and zopiclone are also found in high numbers of suspected drugged drivers. Many drivers have blood levels far exceeding the therapeutic dose range suggesting a high degree of abuse potential for benzodiazepines and zolpidem and zopiclone.[3]
External links
References
- ↑ Biam. "NORDAZEPAM" (in French). Retrieved 18 October. Unknown parameter
|refyear=ignored (help); Unknown parameter|accessyear=ignored (|access-date=suggested) (help); Check date values in:|accessdate=(help) - ↑ Pacifici GM (1986). "Metabolic interaction between morphine and various benzodiazepines". Acta Pharmacol Toxicol (Copenh). 58 (4): 249–52. PMID 2872767. Unknown parameter
|coauthors=ignored (help); Unknown parameter|month=ignored (help) - ↑ Jones AW (2007). "Concentrations of scheduled prescription drugs in blood of impaired drivers: considerations for interpreting the results". Ther Drug Monit. 29 (2): 248–60. PMID 17417081. Unknown parameter
|coauthors=ignored (help); Unknown parameter|month=ignored (help)
- Pages with script errors
- Pages with citations using unsupported parameters
- CS1 errors: dates
- CS1 maint: Unrecognized language
- Drugs with non-standard legal status
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Articles containing unverified chemical infoboxes
- Drug
- Anxiolytics
- Benzodiazepines