Miltefosine
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Kiran Singh, M.D. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING: EMBRYO-FETAL TOXICITY
See full prescribing information for complete Boxed Warning.
IMPAVIDO may cause fetal harm. Fetal death and teratogenicity occurred in animals administered miltefosine at doses lower than the recommended human dose. Do not administer IMPAVIDO to pregnant women. Obtain a serum or urine pregnancy test in females of reproductive potential prior to prescribing IMPAVIDO. Females of reproductive potential should be advised to use effective contraception during IMPAVIDO therapy and for 5 months after therapy.
|
Overview
Miltefosine is an antimicrobial drug that is FDA approved for the treatment of visceral leishmaniasis, cutaneous leishmaniasis,mucosal leishmaniasis. There is a Black Box Warning for this drug as shown here. Common adverse reactions include diarrhea,vomiting,asthenia, abdominal Pain, malaise, pyrexia, dizziness, headache.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
Miltefosine capsules are indicated in adults for the treatment of:
- Visceral leishmaniasis caused by Leishmania donovani.
- Cutaneous leishmaniasis caused by Leishmania braziliensis, Leishmania guyanensis, and Leishmania panamensis.
- Mucosal leishmaniasis caused by Leishmania braziliensis.
Limitations of Use:
- Leishmania species studied in clinical trials evaluating miltefosine were based on epidemiologic data.
- There may be geographic variation in clinical response of the same Leishmania species to miltefosine.
- The efficacy of miltefosine in the treatment of other Leishmania species has not been evaluated.
Dosage
- The treatment duration is 28 consecutive days. Administer with food to ameliorate gastrointestinal adverse reactions.

DOSAGE FORMS AND STRENGTHS
- Miltefosine oral capsules are opaque, red, hard gelatin capsules with “PLB” imprinted on the capsule body and “MILT 50” imprinted on the cap using a white ink. Each capsule contains 50 mg miltefosine.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Miltefosine in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Miltefosine in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Indications
Miltefosine capsules are indicated adolescents ≥12 years of age weighing ≥ 30 kg for the treatment of:
- Visceral leishmaniasis caused by Leishmania donovani.
- Cutaneous leishmaniasis caused by Leishmania braziliensis, Leishmania guyanensis, and Leishmania panamensis.
- Mucosal leishmaniasis caused by Leishmania braziliensis.
Limitations of Use:
- Leishmania species studied in clinical trials evaluating miltefosine were based on epidemiologic data.
- There may be geographic variation in clinical response of the same Leishmania species to miltefosine.
- The efficacy of miltefosine in the treatment of other Leishmania species has not been evaluated.
Dosage
- The treatment duration is 28 consecutive days. Administer with food to ameliorate gastrointestinal adverse reactions.

DOSAGE FORMS AND STRENGTHS
- Miltefosine oral capsules are opaque, red, hard gelatin capsules with “PLB” imprinted on the capsule body and “MILT 50” imprinted on the cap using a white ink. Each capsule contains 50 mg miltefosine.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Miltefosine in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Miltefosine in pediatric patients.
Contraindications
Pregnancy
Miltefosine may cause fetal harm. Miltefosine is contraindicated in pregnant women. Obtain a urine or serum pregnancy test prior to prescribing Miltefosine.
Sjögren-Larsson-Syndrome
Miltefosine is contraindicated in patients who have Sjögren-Larsson-Syndrome.
Hypersensitivity
Miltefosine is contraindicated in patients who are hypersensitive to miltefosine or any miltefosine excipients.
Warnings
|
WARNING: EMBRYO-FETAL TOXICITY
See full prescribing information for complete Boxed Warning.
IMPAVIDO may cause fetal harm. Fetal death and teratogenicity occurred in animals administered miltefosine at doses lower than the recommended human dose. Do not administer IMPAVIDO to pregnant women. Obtain a serum or urine pregnancy test in females of reproductive potential prior to prescribing IMPAVIDO. Females of reproductive potential should be advised to use effective contraception during IMPAVIDO therapy and for 5 months after therapy.
|
Embryo-Fetal Toxicity
- Miltefosine may cause fetal harm. Embryo-fetal toxicity, including death and teratogenicity, was observed in animals administered miltefosine prior to mating, during early pregnancy, and during organogenesis at doses lower than the maximum recommended human dose (MRHD). Do not use miltefosine in pregnant women. Obtain a urine or serum pregnancy test prior to prescribing miltefosine to females of reproductive potential. Advise females of reproductive potential to use effective contraception during miltefosine therapy and for 5 months after completion of therapy.
Reproductive Effects
Females
- Miltefosine caused impaired fertility in rats and reversible follicular atresia and diestrus in dogs at doses approximately 1.0 and 0.2 times respectively the MRHD based on body surface area comparisons. Effects on human female fertility have not been formally studied.
Males
- Miltefosine caused reduced viable sperm counts and impaired fertility in rats at doses approximately 0.4 times the MRHD. A higher dose in rats, approximately 1.0 times the MRHD, caused testicular atrophy and impaired fertility that did not fully reverse 10 weeks after drug administration ended.
- Scrotal pain and decreased or absent ejaculation during therapy have been reported during miltefosine therapy. The effects of miltefosine on human male fertility have not been adequately studied.
- Advise women and men of the animal fertility findings, and that the potential for impaired fertility with miltefosine therapy in humans has not been adequately evaluated.
Renal Effects
Elevations of serum creatinine (Cr) were noted in clinical trials evaluating miltefosine in the treatment of cutaneous, mucosal and visceral leishmaniasis. Monitor renal function weekly in patients receiving miltefosine during therapy and for 4 weeks after end of therapy.
Hepatic Effects
Elevations in liver transaminases (ALT, AST) and bilirubin were noted in clinical trials evaluating miltefosine in the treatment of visceral leishmaniasis. Monitor liver transaminases (ALT, AST) and bilirubin during therapy in patients receiving miltefosine.
Gastrointestinal Effects
Vomiting and/or diarrhea commonly occur during miltefosine administration and may result in volume depletion. Encourage fluid intake to avoid volume depletion.
Thrombocytopenia
Thrombocytopenia during therapy has been reported in patients treated for visceral leishmaniasis. Monitor platelet count during therapy for visceral leishmaniasis.
Absorption of Oral Contraceptives
- Vomiting and/or diarrhea occurring during miltefosine therapy may affect the absorption of oral contraceptives, and therefore compromise their efficacy. If vomiting and/or diarrhea occur during miltefosine therapy, advise females to use additional non-hormonal or alternative method(s) of effective contraception.
Stevens-Johnson Syndrome
Stevens-Johnson syndrome has been reported during miltefosine therapy.Discontinue miltefosine if an exfoliative or bullous rash is noted during therapy.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
Clinical Trials Experience
Visceral Leishmaniasis
- One Phase 3 trial was conducted in patients ≥ 12 years of age in India. Two-hundred and ninety-nine (299) patients (211 men and 88 women) received oral miltefosine at a target dose of 2.5 mg/kg/day for 28 days (50 mg capsule once daily if weight was less than 25 kg and 50 mg capsule twice daily if weight was 25 kg or greater). Patients ranged between 12 and 64 years of age. Weight ranged between 15 and 67 kg (mean weight 38.6 kg) and BMI ranged between 8.2 and 24 (mean 16.1). Ninety-nine (99) patients received 1 mg/kg/day amphotericin B deoxycholate intravenously every other day for 15 doses. A statistically significant higher percentage of men received miltefosine compared to amphotericin B.
- Less than 1% of patients who received miltefosine died (2/299) and no patient who received amphotericin B died. Serious adverse reactions were reported in 2% of miltefosine recipients (6/299) and 1% of amphotericin B recipients (1/99). Approximately 3% of patients discontinued treatment in each treatment arm due to an adverse reaction. Serious adverse reactions and adverse reactions leading to drug discontinuation that were thought to be related or possibly related to miltefosine included Stevens-Johnson syndrome, melena and thrombocytopenia, arthritis and skin rash, CTCAE1 Grade 4 diarrhea (≥10 stools per day) and CTCAE Grade 4 hyperbilirubinemia (≥10x upper limit of normal ULN).

- In this study, creatinine (Cr) elevations ≥ 1.5 times above baseline occurred in approximately 10% of miltefosine recipients and in 40% of amphotericin B recipients at the end of therapy. Ten percent of subjects in each arm had Cr elevations ≥1.5 times above baseline at 6 months follow up. No miltefosine recipient discontinued therapy due to Cr elevation.
- Elevations of transaminases during therapy occurred in up to half of miltefosine recipients and up to a third of amphotericin B recipients. The elevations were mild (< 3x ULN) or moderate (3-5x ULN) in 94% and 6% respectively of miltefosine-treated patients who experienced an elevation. No patient discontinued therapy due to elevations in transaminases.
- At the end of therapy, 62% and 2.4% of miltefosine recipients and 54% and 2% of amphotericin B recipients had platelet count < 150,000 and < 50,000 respectively.
Cutaneous Leishmaniasis
- The efficacy of miltefosine in the treatment of cutaneous leishmaniasis was evaluated in one placebo-controlled trial conducted in Colombia and Guatemala and in two comparative trials conducted in Bolivia and Brazil respectively. In the placebo-controlled trial, eighty-nine (89) patients ≥12 years of age received a target miltefosine dose of 2.5 mg/kg/day for 28 days and forty-four (44) received placebo. In the comparative trials, one hundred and twenty (120) patients ≥12 years of age received a target miltefosine dose of 2.5 mg/kg/day for 28 days and fifty eight (58) patients received 20 mg/kg/day pentavalent antimony (meglumine) parenterally for 20 days.


- In the placebo controlled trial, 12/89 (13.4%) miltefosine subjects had Cr increases of 1.5-3 times above baseline, compared to 2/44 (4.5%) placebo subjects at end of therapy. In the comparative trial, a similar percentage of subjects who received miltefosine or pentavalent antimony had Cr elevations above baseline at 3 and 6 months after therapy (approximately 5%). Approximately 25% of miltefosine subjects and 11% of pentavalent antimony subjects had Cr elevations 1.5-3 times above baseline at the end of therapy in the two active controlled trials. The frequency of AST and ALT increase above upper limit of normal at end of therapy was similar in miltefosine and placebo recipients (approximately 5%).
- Other adverse events seen at <2% incidence in the miltefosine group included anemia, lymphadenopathy, abdominal distension, constipation, dysphagia, flatulence, fatigue, malaise, abscess, cellulitis, ecthyma, paresthesia, testicular pain, testicular swelling, Stevens-Johnson syndrome, urticaria, rash, pyoderma.
Postmarketing Experience
- The following adverse reactions have been identified during use of miltefosine worldwide. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Blood and Lymphatics Disorders: thrombocytopenia, agranulocytosis
- Gastrointestinal Disorders: melena
- General Disorders: generalized edema, peripheral edema
- Hepatobiliary Disorders: jaundice
- Nervous System Disorders: seizure
- Reproductive System and Breast Disorders: scrotal pain, decreased ejaculate volume, absent ejaculation.
- Vascular Disorders: epistaxis
Drug Interactions
- In vitro and animal metabolism studies showed that miltefosine did not markedly induce or inhibit the activity of the major human cytochrome P450 enzymes. The potential of miltefosine to interact with drug transporters has not been evaluated.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): Pregnancy Category D
Risk Summary
- miltefosine may cause fetal harm. Human pregnancy data are not available, however, embryo-fetal toxicity including death and teratogenicity, was observed in embryo-fetal studies in rats and rabbits administered oral miltefosine during organogenesis at doses that were respectively 0.06 and 0.2 times the maximum recommended human dose (MRHD), based on body surface area (BSA) comparison. Numerous visceral and skeletal fetal malformations were observed in a fertility study in female rats administered miltefosine prior to mating through day 7 of pregnancy at doses 0.3 times the MRHD. Do not administer miltefosine to pregnant women.
Clinical Considerations
- During pregnancy, visceral leishmaniasis may be life-threatening for the mother and may result in adverse fetal outcomes, including spontaneous abortion, congenital disease due to vertical transmission, small for gestational age newborn, and severe anemia. During pregnancy, cutaneous leishmaniasis may manifest with larger and atypical appearing lesions and may be associated with increased risk for adverse fetal outcomes, including preterm births and stillbirths.
Animal Data
- Miltefosine administration in rat embryo-fetal toxicity studies during early embryonic development (Day 6 to Day 15 of gestation) caused embryo-fetal toxicity including death and teratogenicity at dosages of ≥ 1.2 mg/kg/day (0.06 times the MRHD based on BSA comparison). Teratogenic effects included undeveloped cerebrum, hemorrhagic fluid filling the lumina of the skull, cleft palate and generalized edema. Embryo-fetal toxicity was also observed in rabbits after oral administration of miltefosine during organogenesis (Day 6 to Day 18 of gestation) at doses ≥ 2.4 mg/kg/day (0.2 times the MRHD based on BSA comparison). In both rats and rabbits, there were no viable litters at miltefosine doses ≥ 6.0 mg/kg/day (0.3 or 0.6 times the MRHD based on BSA comparisons for rats and rabbits respectively).
- In a separate female fertility study in rats, miltefosine doses ≥ 6.81 mg/kg/day (0.3 times the MRHD based on BSA comparison) administered for four weeks before mating and up to Day 7 of pregnancy produced numerous visceral (misshapen cerebral structures, dilated ventricles filled with brown masses, misshapen spinal cord, misshapen and malpositioned eyes, hypophysis, and absent inner ear) and skeletal (cleft palate, dumbbell-shaped ossification of thoracic vertebral centers, markedly enlarged skull bones, and markedly dilated suturae) fetal malformations.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Miltefosine in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Miltefosine during labor and delivery.
Nursing Mothers
- It is not known whether miltefosine is present in human milk. Because many drugs are present in human milk and because of the potential for serious adverse reactions in nursing infants from miltefosine, a decision should be made whether to discontinue nursing or discontinue the drug, taking into account the importance of the drug to the mother. Breastfeeding should be avoided for 5 months after miltefosine therapy.
Pediatric Use
- Safety and effectiveness in pediatric patients < 12 years have not been established. Juvenile rats were more sensitive to the miltefosine-induced effects, especially retinal and kidney effects, than adult rats.
Geriatic Use
- Clinical studies of miltefosine did not include sufficient numbers of subjects 65 years of age and over to determine if they respond differently than younger subjects.
Gender
Contraception
- Miltefosine may cause fetal harm when used during pregnancy. Advise females of reproductive potential to use effective contraception during miltefosine therapy and for 5 months after therapy is completed.
- Vomiting and/or diarrhea occurring during miltefosine therapy may affect absorption of oral contraceptives and therefore may compromise their efficacy. Advise females who use oral contraceptives to use additional non-hormonal or alternative method(s) of effective contraception during miltefosine therapy if vomiting and/or diarrhea occurs during therapy.
Infertility
Females
- Miltefosine caused impaired fertility in rats and caused reversible follicular atresia and diestrus in dogs at doses approximately 1.0 and 0.2 times respectively the MRHD. The effects of miltefosine on human female fertility have not been formally studied.
Males
- Miltefosine caused reduced viable sperm counts and impaired fertility in rats at doses approximately 0.4 times the MRHD . A higher dose in rats, approximately 1.0 times the MRHD, caused testicular atrophy and impaired fertility that did not fully reverse 10 weeks after drug administration ended. The effects of miltefosine on human male fertility have not been adequately studied.
- Advise women and men of the animal fertility findings, and that the potential for impaired fertility with miltefosine therapy has not been adequately evaluated.
Race
There is no FDA guidance on the use of Miltefosine with respect to specific racial populations.
Renal Impairment
- Patients with serum creatinine or BUN levels ≥1.5 times the upper limit of normal were excluded from the clinical studies. Miltefosine pharmacokinetics have not been studied in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Miltefosine in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Miltefosine in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Miltefosine in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
Monitoring
There is limited information regarding Monitoring of Miltefosine in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Miltefosine in the drug label.
Overdosage
- The common adverse effects of vomiting, diarrhea, and abdominal pain are likely in case of overdose. Institute adequate hydration to prevent the risk of impaired renal function, and replace electrolytes as necessary. Because miltefosine is only slightly excreted in the urine, forced diuresis will not increase miltefosine excretion. Gastrointestinal lavage is of unknown value. A specific antidote to treat miltefosine overdose is not known.
Pharmacology

Mechanism of Action
- The specific mode of action of miltefosine against Leishmania species is unknown. The mechanism of action of miltefosine is likely to involve interaction with lipids (phospholipids and sterols), including membrane lipids, inhibition of cytochrome c oxidase (mitochondrial function), and apoptosis-like cell death.
Structure
- Miltefosine capsules contain the active ingredient miltefosine, an antileishmanial agent. The chemical name of miltefosine is 2-(hexadecyloxy)hydroxyphosphenyloxy-N,N,N-trimethylethylammonium inner salt. Miltefosine is a white powder that is freely soluble in water, 0.1 N HCl or NaOH, methanol, and ethanol. It has the empirical formula of C21H46NO4P with a molecular weight of 407.6 and the following structural formula:

- The inactive ingredients are colloidal silicon dioxide, microcrystalline cellulose, lactose monohydrate, talc, and magnesium stearate. The capsule shell contains gelatin, titanium dioxide, ferric oxide, and purified water.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Miltefosine in the drug label.
Pharmacokinetics
- The pharmacokinetic parameters of miltefosine in patients with visceral and cutaneous leishmaniasis treated for 28 days with miltefosine are listed in Table 5. Due to the long half-life of miltefosine (> 6 days), trough plasma concentrations did not appear to reach a steady state at the end of treatment (i.e., Day 28).

Absorption
- Absolute bioavailability of miltefosine has not been determined. In patients with visceral leishmaniasis, maximum miltefosine concentrations following oral administration of miltefosine capsules were reached right before the next dose in many patients, indicating that the absorption of miltefosine may proceed throughout the dosing interval.
Distribution
- The distribution of miltefosine has not been studied in humans. Human plasma protein binding of miltefosine, evaluated by an ultracentrifugation method, was 98% over the drug concentration range from 0.1 to 10 µg/mL. In rats, radioactivity of [14C] miltefosine is widely distributed after both single and repeated oral administration with highest uptake of radioactivity in kidney, liver, and spleen. Placental transfer and excretion into milk have not been investigated.
Metabolism and Excretion
- No in vitro oxidative metabolism by 15 different human cytochrome P450 enzymes (1A1, 1A2, 1B1, 2A6, 2B6, 2C8, 2C9, 2C18, 2C19, 2D6, 2E1, 3A4, 3A5, 3A7, and 4A1) was observed.
- A slow metabolic breakdown could be shown in human hepatocytes, resulting in the release of choline by phospholipase D-like cleavage of the miltefosine molecule. The fatty alcohol-containing fragment of miltefosine can enter the metabolism of fatty acids after being oxidized to palmitic acid. This oxidation is blocked in patients with Sjögren-Larsson syndrome, which is caused by a genetic defect in fatty aldehyde dehydrogenase activity. Miltefosine is contraindicated in patients who have Sjögren-Larsson-Syndrome.
- There was little or no evidence of time or metabolism dependent inhibition of the cytochrome P450 enzymes examined at up to approximately 40 µg/mL miltefosine.
- Oral administration of miltefosine did not markedly induce the content of hepatic CYP3A assayed by demethylation activity of erythromycin in rats.
- In visceral leishmaniasis patients, <0.2% of the administered dose was excreted into the urine.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Mutagenicity/Carcinogenicity: Miltefosine tested negative in the AMES-Salmonella test, DNA-amplification test, chromosomal aberration test in vitro, UDS-test in vivo/in vitro, and oral mouse micronucleus test in vivo. The V 79 mammalian cell HPRT gene mutation test showed an increase in mutant frequency without dose dependency. In view of all mutagenicity test results, the single positive finding in the V 79 HPRT test is considered to be not of toxicological relevance with respect to a mutagenic risk to humans.
- Carcinogenicity studies were not performed. In a 52-week oral rat toxicity study, testicular Leydig cell adenoma was observed in 3 of 30 male rats with daily administration of 21.5 mg/kg/day miltefosine (1.0 times the MRHD based on BSA comparison). The carcinogenic potential of miltefosine in humans is unknown.
- In a Segment I fertility study in male rats, testicular atrophy, reduced numbers of viable sperm, and impaired fertility were observed in rats following daily oral doses of ≥ 8.25 mg/kg (0.4 times the MRHD based on BSA comparison). These findings were reversible within a recovery period of 10 weeks except at the highest dose tested, 21.5 mg/kg/day (1.0 times the MRHD based on BSA comparison), where effects were not fully reversible.
- In a female fertility study in rats, estrus cycle arrest in the metestrus or diestrus phases occurred with the high-dose of 21.5 mg/kg (1.0 times the MRHD based on BSA comparison). At doses of 6.81 and 21.5 mg/kg (0.3 and 1.0 times the MRHD respectively based on BSA comparison) increased numbers of embryonic and fetal resorptions and dead fetuses were observed. In a 52-week toxicology study in dogs, increased numbers of atretic follicles in the ovaries, and cycle arrest in the uterus, vagina, and mammary gland with morphology consistent with anestrus or diestrus was observed at doses ≥ 1 mg/kg/day (0.2 times the MRHD based on BSA comparison). The effects in dogs were fully reversible after a recovery period of 6 weeks.
Animal Toxicology and/or Pharmacology
- Toxicological studies with miltefosine have been performed in mice, rats, dogs, and rabbits. Adverse reactions not observed in clinical studies but seen in animals at exposure levels similar to clinical exposure levels and with possible relevance to clinical use were as follows:
- Acute and chronic toxicity: The oral administration of miltefosine in rats was associated with lesions affecting the eyes (retinal degeneration). Retinal degeneration was observed after 8-weeks treatment at doses of 10 mg/kg/day (0.5 times the MRHD based on BSA comparison). Juvenile rats were more sensitive to the miltefosine-induced effects, especially on eyes and kidneys, than adult rats with retinal degeneration occurring at doses ≥ 2.15 mg/kg/day (0.1 times the MRHD based on BSA comparison), and reversible damage to proximal tubule epithelium occurring at doses ≥ 4.64 mg/kg/day (0.2 times the MRHD based on BSA comparison).
Clinical Studies
Treatment of Visceral Leishmaniasis
- One randomized, open-label, active-controlled study was conducted to evaluate the efficacy of miltefosine in the treatment of visceral leishmaniasis in Bihar, India, an area where L. donovani is known epidemiologically to be the prevalent infecting species. Patients ≥ 12 years of age with clinical signs and symptoms compatible with visceral leishmaniasis (fever, splenomegaly, and cytopenia) confirmed by the presence of Leishmania amastigotes in aspirates of spleen or bone marrow were randomized to receive oral miltefosine or intravenous amphotericin B deoxycholate in a 3:1 ratio. Patients weighing ≥ 25 kg received an miltefosine 50 mg capsule with meals twice a day. Patients weighing < 25 kg received an miltefosine 50 mg capsule with meals once a day in the morning. Weight ranged between 15 and 67 kg (mean weight 38.6 kg) and BMI ranged between 8.2 and 24 (mean 16.1). No patient weighed more than 70kg. Amphotericin B was administered intravenously over 6 continuous hours at 1 mg/kg every other day for 15 doses. Patients were hospitalized for the duration of therapy.
- Exclusion criteria included platelet count <50 × 109/L, white cell count <1 × 109/L, hemoglobin <6 g/100 mL, AST or ALT ≥3 times upper limit of the normal range, bilirubin ≥2 times upper limit of the normal range, serum creatinine or BUN >1.5 times upper limit of the normal range, prothrombin time >5 seconds above control, and any non-compensated or uncontrolled condition including human immunodeficiency virus (HIV) infection. Women of reproductive potential were required to use effective contraception for the duration of therapy and for 2 months post therapy.
- Final cure was defined as initial cure at end of therapy plus absence of signs and symptoms of visceral leishmaniasis at 6 months follow up. Initial cure at the end of therapy was evaluated by repeat spleen or bone marrow aspiration. Patients with initial parasitologic cure were followed for 6 months; patients without absence of clinical signs and symptoms of visceral leishmaniasis were to be evaluated with repeat spleen or bone marrow aspiration to determine final cure.
- Two hundred and ninety nine (299) patients received miltefosine and 99 patients received amphotericin B. Approximately, 70% of patients in each arm had previously failed treatment with pentavalent antimony. Initial cure was achieved in 98% of patients in each treatment arm. At 6 months after therapy, 88 (29.5%) miltefosine recipients and 12 (12.1%) amphotericin B recipients continued to have signs and symptoms suggestive of visceral leishmaniasis. These signs or symptoms were attributed to alternative diagnosis in 73 patients. The remaining 27 patients, all in the miltefosine arm, underwent evaluation with splenic or bone marrow aspiration, and 9 (3.0%) were positive for Leishmania amastigotes, indicating relapse. The final cure rates for miltefosine and amphotericin B were 94% and 97%, respectively.

Treatment of Cutaneous Leishmaniasis
- A placebo controlled study was performed in Colombia where L. panamensis and L. braziliensis are epidemiologically known to be the prevalent infecting Leishmania species, and in Guatemala where L. braziliensis is epidemiologically known to be the prevalent infecting species. The study included male and female patients older than 12 years of age who had newly diagnosed or relapsing cutaneous leishmaniasis without mucosal involvement, parasitologically confirmed, presenting with at least one skin ulcer with minimum area of 50 mm2. Exclusion criteria were AST or ALT ≥2 times upper limit of normal range, bilirubin ≥1.5 times upper limit of normal range, and serum creatinine or BUN >1.5 times upper limit of normal range. Women of reproductive potential were required to use effective contraception for the duration of therapy and for 2 months post therapy.
- Patients were randomized to receive miltefosine or placebo in a 2:1 allocation. Patients who weighed < 45 kg received miltefosine 50 mg capsule twice a day, and patients who weighed ≥45 kg received miltefosine 50 mg capsule three times a day. No patient weighed more than 84 kg. Definite cure was defined as apparent (complete epithelialization of all lesions) or partial cure (incomplete epithelialization, no enlargement by > 50% in lesions, no appearance of new lesions, and negative parasitology if done) at 2 weeks after end of therapy and complete epithelialization of all ulcers at 6 months after end of therapy. The definite cure rate for miltefosine was statistically significantly higher than the cure rate for placebo.
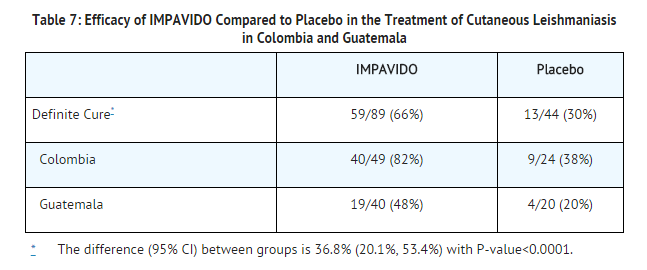
- An additional study of miltefosine was conducted in Bahia and Manaus, two regions in Brazil where respectively L. braziliensis and L. guyanensis are epidemiologically the prevalent infecting pathogens. Adolescent/adult patients aged 12-65 years received miltefosine orally for 28 days. Miltefosine target dose was 2.5 mg/kg/day: patients weighing 15-29 kg received 50 mg once daily, patients weighing 30-45 kg received 50 twice mg daily and patients weighing > 46 kg received 50 mg three times daily. The efficacy criteria were initial cure (complete re-epithelialization of the ulcer at 2 months after the end of therapy) followed by definite cure (complete re-epithelialization at 6 months after the end of therapy). Definite cure rate in patients aged ≥12 years was 27/40 (67.5%) for Manaus, Brazil and 34/40 (85%) for Bahia, Brazil.
Treatment of Mucosal Leishmaniasis
- A single arm study was conducted to evaluate the efficacy of miltefosine capsules for the treatment of mucosal leishmaniasis. The study was conducted in Bolivia where L. braziliensis is epidemiologically the prevalent species.
- Seventy nine (79) patients ≥18 years of age with a cutaneous leishmaniasis scar plus parasites observed or cultured from lesion material or a positive skin test, and no clinically significant concomitant disease received miltefosine at a target dose of 2.5 mg/kg/day for 28 days. By 12 months after the end of therapy, 49 of the patients (62%) had complete resolution of edema, erythema, infiltration and erosion from the involved mucosal sites.
How Supplied
- Each miltefosine capsule contains 50 mg miltefosine in an opaque, red, hard gelatin capsule. Miltefosine capsules are supplied in a folded peel/push-through blister card. Each blister card contains 14 capsules. Each carton contains two blister cards (NDC 61744-050-01).
Storage
- Store at 20-25 °C (68-77 °F); excursions permitted to 15-30 °C (59-86 °F). [See USP Controlled Room Temperature]. Protect from moisture.
- Dispense only in the original carton.
Images
Drug Images
{{#ask: Page Name::Miltefosine |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Miltefosine |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Dosing Instructions'
- Miltefosine is administered with food to ameliorate gastrointestinal side effects.
Instruct the patient to swallow the capsule whole and not to chew it or break it apart. Instruct the patient to complete the full course of therapy.
- Inform the patient that abdominal pain, nausea, vomiting, and diarrhea are common side effects of therapy with miltefosine and instruct the patient to inform their healthcare provider if these gastrointestinal side effects are severe or persistent. Instruct the patient to consume sufficient fluids to avoid dehydration and, consequently, the risk of kidney injury.
Females and Males of Reproductive Potential
- Advise women of reproductive potential to use effective contraception during miltefosine therapy and for 5 months after therapy ends.
- Advise women who use oral contraceptives to use additional non-hormonal or alternative method(s) of effective contraception during miltefosine therapy if vomiting and/or diarrhea occurs .
Advise nursing mothers not to breastfeed during miltefosine therapy and for 5 months after therapy is completed.
- Advise women and men that miltefosine caused infertility in male rats, impaired fertility in female rats, and caused atresia in ovarian follicles in female dogs. Advise patients that the potential of impaired fertility in humans has not been adequately evaluated.
Precautions with Alcohol
- Alcohol-Miltefosine interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- IMPAVIDO
Look-Alike Drug Names
- A® — B®[1]
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Miltefosine
|Pill Name=Miltefosine image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Miltefosine |Label Name=Miltefosine ingredients and appearance.png
}}
{{#subobject:
|Label Page=Miltefosine |Label Name=Miltefosine medication guide.png
}}