Metipranolol
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Rabin Bista, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Metipranolol is a non-selective beta-adrenergic receptor blocking agent that is FDA approved for the treatment of elevated intraocular pressure in patients with ocular hypertension or open angle glaucoma.. Common adverse reactions include Burning sensation in eye, Blurred vision, Conjunctivitis, Blepharitis, photophobia.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- Metipranolol ophthalmic solution is indicated in the treatment of elevated intraocular pressure in patients with ocular hypertension or open angle glaucoma.
Dosage
- The recommended dose is one drop of metipranolol ophthalmic solution in the affected eye(s) twice a day.
- If the patient's IOP is not at a satisfactory level on this regimen, use of more frequent administration or a larger dose of metipranolol ophthalmic solution is not known to be of benefit. Concomitant therapy to lower intraocular pressure can be instituted.
- In clinical trials, metipranolol ophthalmic solution was safely used during concomitant therapy with pilocarpine, epinephrine, or acetazolamide.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Metipranolol in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Metipranolol in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Metipranolol in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Metipranolol in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Metipranolol in pediatric patients.
Contraindications
- Hypersensitivity to any component of this product.
- Metipranolol ophthalmic solution is contraindicated in patients with bronchial asthma or a history of bronchial asthma, or severe chronic obstructive pulmonary disease; symptomatic sinus bradycardia; greater than a first degree atrioventricular block; cardiogenic shock; or overt cardiac failure.
Warnings
- As with other topically applied ophthalmic drugs, this drug may be absorbed systemically. Thus, the same adverse reactions found with systemic administration of beta-adrenergic blocking agents may occur with topical administration. For example, severe respiratory reactions and cardiac reactions, including death due to bronchospasm in patients with asthma, and rarely, death in association with cardiac failure, have been reported following topical application of beta-adrenergic blocking agents .
- Since metipranolol ophthalmic solution had a minor effect on heart rate and blood pressure in clinical studies, caution should be observed in treating patients with a history of cardiac failure. Treatment with metipranolol ophthalmic solution should be discontinued at the first evidence of cardiac failure.
- Metipranolol ophthalmic solution, or other beta-blockers, should not, in general, be administered to patients with chronic obstructive pulmonary disease (e.g., chronic bronchitis, emphysema) of mild or moderate severity. However, if the drug is necessary in such patients, then it should be administered with caution since it may block bronchodilation produced by endogenous and exogenous catecholamine stimulation of beta2 receptors.
Precautions
General
- Because of potential effects of beta-adrenergic receptor blocking agents relative to blood pressure and pulse, these agents should be used with caution in patients with cerebrovascular insufficiency. If signs or symptoms suggesting reduced cerebral blood flow develop following initiation of therapy with metipranolol ophthalmic solution, alternative therapy should be considered.
- Some authorities recommend gradual withdrawal of beta-adrenergic receptor blocking agents in patients undergoing elective surgery. If necessary during surgery, the effects of beta-adrenergic receptor blocking agents may be reversed by sufficient doses of such agonists as isoproterenol, dopamine, dobutamine or levarterenol.
- While metipranolol ophthalmic solution has demonstrated a low potential for systemic effect, it should be used with caution in patients with diabetes (especially labile diabetes) because of possible masking of signs and symptoms of acute hypoglycemia.
- Beta-adrenergic receptor blocking agents may mask certain signs and symptoms of hyperthyroidism, and their abrupt withdrawal might precipitate a thyroid storm.
- Beta-adrenergic blockade has been reported to potentiate muscle weakness consistent with certain myasthenic symptoms (e.g., diplopia, ptosis, and generalized weakness).
- Risk of anaphylactic reaction: While taking beta-blockers, patients with a history of severe anaphylactic reaction to a variety of allergens may be more reactive to repeated challenge, either accidental, diagnostic, or therapeutic. Such patients may be unresponsive to the usual doses of epinephrine used to treat allergic reaction.
Adverse Reactions
Clinical Trials Experience
- In clinical trials, the use of metipranolol ophthalmic solution has been associated with transient local discomfort.
- Other ocular adverse reactions, such as abnormal vision, blepharitis, blurred vision, browache, conjunctivitis, edema, eyelid dermatitis, photophobia, tearing, and uveitis have been reported in small numbers of patients.
- Other systemic adverse reactions, such as allergic reaction, angina, anxiety, arthritis, asthenia, atrial fibrillation, bradycardia, bronchitis, coughing, depression, dizziness, dyspnea, epistaxis, headache, hypertension, myalgia, myocardial infarct, nausea, nervousness, palpitation, rash, rhinitis, and somnolence have also been reported in small numbers of patients.
Postmarketing Experience
There is limited information regarding Metipranolol Postmarketing Experience in the drug label.
Drug Interactions
- Metipranolol ophthalmic solution should be used with caution in patients who are receiving a beta-adrenergic blocking agent orally, because of the potential for additive effects on systemic beta-blockade.
- Close observation of the patient is recommended when a beta-blocker is administered to patients receiving catecholamine-depleting drugs such as reserpine, because of possible additive effects and the production of hypotension and/or bradycardia.
- Caution should be used in the coadministration of beta-adrenergic receptor blocking agents, such as metipranolol, and oral or intravenous calcium channel antagonists, because of possible precipitation of left ventricular failure, and hypotension. In patients with impaired cardiac function, who are receiving calcium channel antagonists, coadministration should be avoided.
- The concomitant use of beta-adrenergic receptor blocking agents with digitalis and calcium channel antagonists may have additive effects, prolonging atrioventricular conduction time.
- Caution should be used in patients using concomitant adrenergic psychotropic drugs.
- Ocular: In patients with angle-closure glaucoma, the immediate treatment objective is to re-open the angle by constriction of the pupil with a miotic agent. Metipranolol ophthalmic solution has little or no effect on the pupil, therefore, when it is used to reduce intraocular pressure in angle-closure glaucoma, it should be used only with concomitant administration of a miotic agent.
Use in Specific Populations
Pregnancy
- No drug related effects were reported for the segment II teratology study in fetal rats after administration, during organogenesis, to dams of up to 50 mg/kg/day. Metipranolol ophthalmic solution has been shown to increase fetal resorption, fetal death, and delayed development when administered orally to rabbits at 50 mg/kg/day during organogenesis.
- There are no adequate and well-controlled studies in pregnant women. Metipranolol ophthalmic solution should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
Pregnancy Category (AUS):
(Description)
Labor and Delivery
(Description)
Nursing Mothers
- It is not known whether metipranolol ophthalmic solution is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when metipranolol ophthalmic solution is administered in nursing women
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Geriatic Use
No overall differences in safety or effectiveness have been observed between elderly and younger patients.
Gender
(Description)
Race
(Description)
Renal Impairment
(Description)
Hepatic Impairment
(Description)
Females of Reproductive Potential and Males
(Description)
Immunocompromised Patients
(Description)
Others
(Description)
Administration and Monitoring
Administration
- Ophthalmic
Monitoring
There is limited information regarding Monitoring of Metipranolol in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Metipranolol and IV administrations.
Overdosage
- No information is available on overdosage of metipranolol ophthalmic solution in humans. The symptoms which might be expected with an overdose of a systemically administered beta-adrenergic receptor blocking agent are bradycardia, hypotension and acute cardiac failure.
Pharmacology
| |
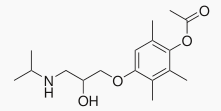
1 : 1 mixture (racemate)Metipranolol
| |
| Systematic (IUPAC) name | |
| (RS)-4-{[-2-hydroxy-3-(isopropylamino)propyl]oxy}-2,3,6-trimethylphenyl acetate | |
| Identifiers | |
| CAS number | |
| ATC code | S01 C07BA68 (WHO) |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 309.401 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
- Metipranolol blocks beta1 and beta2 (non-selective) adrenergic receptors. It does not have significant intrinsic sympathomimetic activity, and has only weak local anesthetic (membrane-stabilizing) and myocardial depressant activity.
- Orally administered beta-adrenergic blocking agents reduce cardiac output in both healthy subjects and patients with heart disease. In patients with severe impairment of myocardial function, beta-adrenergic receptor antagonists may inhibit the sympathetic stimulatory effect necessary to maintain adequate cardiac output.
- Beta-adrenergic receptor blockade in the bronchi and bronchioles may result in significantly increased airway resistance from unopposed para-sympathetic activity. Such an effect is potentially dangerous in patients with asthma or other bronchospastic conditions
Structure
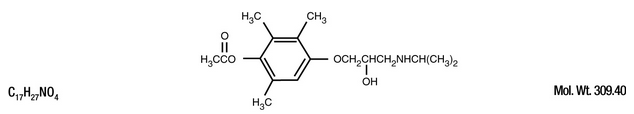
- Metipranolol ophthalmic solution 0.3% is a sterile solution that contains metipranolol, a non-selective beta-adrenergic receptor blocking agent. Metipranolol is a white, odorless, crystalline powder. The chemical name of metipranolol is (±)-1-(4-Hydroxy-2, 3,5-trimethylphenoxy)-3-(isopropylamino)-2-propanol-4-acetate.
- The chemical structural of metipranolol is:
- Each mL of metipranolol ophthalmic solution, for ophthalmic administration, contains 3 mg metipranolol. INACTIVES: povidone, glycerol, hydrochloric acid, sodium chloride, edetate disodium, and purified water. Sodium Hydroxide may be added to adjust pH. PRESERVATIVE ADDED: Benzalkonium chloride 0.004%. DM-00
Pharmacodynamics
(Description)
Pharmacokinetics
- Metipranolol when applied topically in the eye, has the action of reducing elevated as well as normal intraocular pressure (IOP), whether or not accompanied by glaucoma. Elevated intraocular pressure is a major risk factor in the pathogenesis of glaucomatous visual field loss. The higher the level of intraocular pressure, the greater the likelihood of glaucomatous visual field loss and optic nerve damage.
- The primary mechanism of the ocular hypotensive action of metipranolol is most likely due to reduction in aqueous humor production. A slight increase in outflow may be an additional mechanism. Metipranolol reduces IOP with little or no effect on pupil size or accommodation.
- In controlled studies of patients with intraocular pressure greater than 24 mmHg at baseline, metipranolol ophthalmic solution reduced the average intraocular pressure approximately 20 - 26%.
- The onset of action of metipranolol ophthalmic solution, as measured by a reduction in intraocular pressure, occurs within 30 minutes after a single administration. The maximum effect occurs at about 2 hours. A reduction in intraocular pressure can be demonstrated 24 hours after a single dose. Clinical studies in patients with glaucoma treated for up to two years indicate that an intraocular pressure lowering effect is maintained.
ANIMAL PHARMACOLOGY
- In rabbits administered metipranolol in one eye at 2 to 4 fold increased concentrations, multi-focal interstitial nephritis was observed in male animals, and lympho-hystiocytic and heterophilic interstitial pneumonia was observed in female animals. The clinical relevance of these findings is unknown.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Lifetime studies with metipranolol have been conducted in mice at oral doses of 5, 50 and 100 mg/kg/day and in rats at oral doses of up to 70 mg/kg/day. Metipranolol demonstrated no carcinogenic effect. In the mouse study, female animals receiving the low, but not the intermediate or high dose, had an increased number of pulmonary adenomas. The significance of this observation is unknown. In a variety of in vitro and in vivo bacterial and mammalian cell assays, metipranolol was nonmutagenic.
- Reproduction and fertility studies of metipranolol in rats and mice showed no adverse effect on male fertility at oral doses of up to 50 mg/kg/day, and female fertility at oral doses of up to 25 mg/kg/day.
Clinical Studies
There is limited information regarding Metipranolol Clinical Studies in the drug label.
How Supplied
- Metipranolol ophthalmic solution 0.3% is supplied in a plastic bottle with a controlled drop tip and a white plastic screw-top cap as follows:
- 5mL: NDC 61314-447-05
- 10mL: NDC 61314-447-10
Storage
- Store at controlled room temperature, 15°-30°C (59°-86°F). Replace cap immediately after use.
Images
Drug Images
{{#ask: Page Name::Metipranolol |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
PRINCIPAL DISPLAY PANEL
NDC 61314-447-10 Rx Only
FALCON PHARMACEUTICALS®
Metipranolol
Ophthalmic
Solution
0.3%
10 mL STERILE
AFFILIATE OF
ALCON LABORATORIES, INC
QUALITY RX
Ingredients and Appearance
{{#ask: Label Page::Metipranolol |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Patients should be instructed to avoid allowing the tip of the dispensing containers to contact the eye or surrounding structures. Patients should be advised that metipranolol contains benzalkonium chloride which may be absorbed by soft contact lenses. Contact lenses should be removed prior to administration of the solution. Lenses may be reinserted 15 minutes following metipranolol administration.
Precautions with Alcohol
Alcohol-Metipranolol interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Optipranolol®[1]
Look-Alike Drug Names
There is limited information regarding Metipranolol Look-Alike Drug Names in the drug label.
Drug Shortage
Price
References
The contents of this FDA label are provided by the National Library of Medicine.