Lung cancer diagnostic study of choice
|
Lung cancer Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Lung cancer diagnostic study of choice On the Web |
|
American Roentgen Ray Society Images of Lung cancer diagnostic study of choice |
|
Risk calculators and risk factors for Lung cancer diagnostic study of choice |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Dildar Hussain, MBBS [2]Kim-Son H. Nguyen M.D., Cafer Zorkun, M.D., Ph.D. [3]. Rim Halaby, M.D. [4], Michael Maddaleni, B.S., Anum Ijaz M.B.B.S., M.D.[5]
Overview
Chest CT scan is the modality of choice in the diagnosis of lung cancer. Findings on CT scan suggestive of lung cancer include a solitary pulmonary nodule, centrally located masses, mediastinal invasion CT scans help stage the lung cancer. A CT scan of the abdomen and brain can help visualize the common sights of metastases such as adrenal glands, liver, and brain. CT scans diagnose lung cancer by providing anatomical detail to locate the tumor, demonstrating proximity to the nearby structures, and deciphering whether lymph nodes are enlarged in the mediastinum.
Diagnostic Study of Choice
Study of Choice
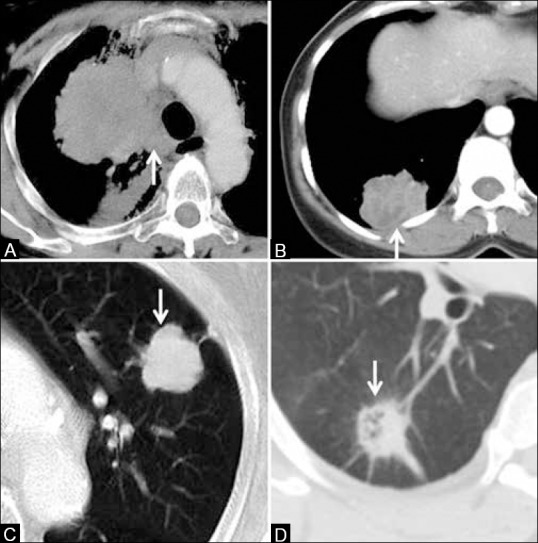
Chest CT scan is the modality of choice in the diagnosis of lung cancer. Findings on CT scan suggestive of lung cancer include:[1]
- Solitary pulmonary nodule
- Centrally located masses
- Mediastinal invasion
- Peripherally situated lesions invading the chest wall
- A ground-glass opacity
- Consolidation
- Mixed density or pure ground glass nodules
- Mixed density or pure ground glass consolidation
 |
 |
 |
 |
 |
 |
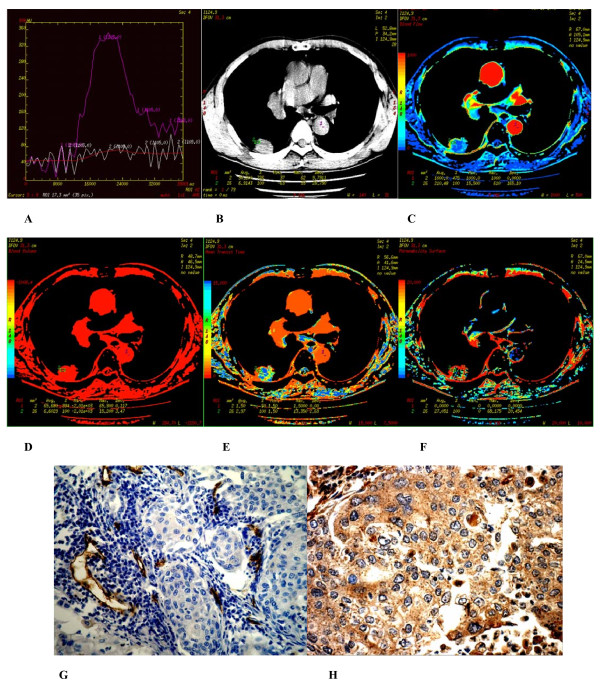
Spiral CT Perfusion Imaging
- Spiral CT perfusion study can be used as a diagnostic method for peripheral pulmonary nodules.
- Spiral CT perfusion study provides non-invasive method for the quantitative assessment of blood flow patterns of peripheral pulmonary nodules.
- Spiral CT perfusion imaging is analyzed and evaluated for:[2]
- TDC (time density curve)
- Perfusion parametric maps
- The respective perfusion parameters
- Immunohistochemical findings of microvessel density (MVD) measurement
- VEGF expression
 |
 |
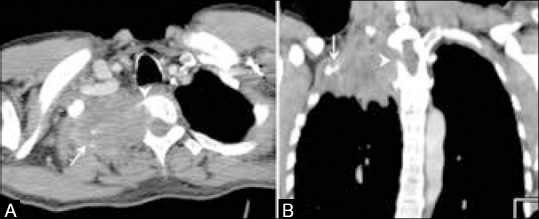
CT Findings of Metastatic disease
- CT scans help stage the lung cancer. A CT scan of the abdomen and brain can help visualize the common sights of metastases such as adrenal glands, liver, and brain.
- The benefits of CT scan in lung cancer patients are the following:[3]
- Provides anatomical detail to locate the tumor
- Demonstrates proximity to nearby structures
- Deciphers whether lymph nodes are enlarged in the mediastinum
 |
 |
 |
- Unfortunately, research has shown that there are a number of false positives associated with CT scanning because CT scan alone cannot determine malignancy.
- A positive result for a tumor using CT scan is typically followed up with a biopsy for confirmation.
Staging
The following is 2017 TNM classification of lung cancer.[4][5][6]
T: Primary Tumor
| T | Description |
| TX |
|
|---|---|
| T0 |
|
| Tis | |
| T1 |
|
| T2 |
|
| T3 |
AND
|
| T4 |
|
N: Regional Lymph Nodes
| T | Description |
| NX |
|
|---|---|
| N0 |
|
| N1 |
|
| N2 |
|
| N3 |
|
M: Distant Metastasis
| T | Description |
| MX |
|
|---|---|
| M0 |
|
| M1 |
|
Classification of Lung Cancer by Staging
| Stage | T | N | M |
| Occult carcinoma | TX | N0 | M0 |
| Stage 0 | Tis | N0 | M0 |
| Stage IA1 | T1(mi)/T1a | N0 | M0 |
| Stage IA2 | T1b | N0 | M0 |
| Stage IA3 | T1c | N0 | M0 |
| Stage IB | T2a | N0 | M0 |
| Stage IIA | T2b | N0 | M0 |
| Stage IIB | T1a | N1 | M0 |
| T1c | N1 | M0 | |
| T2a | N1 | M0 | |
| T2b | N1 | M0 | |
| T3 | N0 | M0 | |
| Stage IIIA | T1a | N2 | M0 |
| T1b | N2 | M0 | |
| T1c | N2 | M0 | |
| T2a | N2 | M0 | |
| T2b | N2 | M0 | |
| T1a | N2 | M0 | |
| T1b | N2 | M0 | |
| T1c | N2 | M0 | |
| T2a | N2 | M0 | |
| T2b | N2 | M0 | |
| T3 | N1 | M0 | |
| T4 | N0 | M0 | |
| T4 | N1 | M0 | |
| Stage IIIB | T1a | N3 | M0 |
| T1b | N3 | M0 | |
| T1c | N3 | M0 | |
| T2a | N3 | M0 | |
| T2b | N3 | M0 | |
| T1a | N3 | M0 | |
| T1b | N3 | M0 | |
| T1c | N3 | M0 | |
| T2a | N3 | M0 | |
| T2b | N3 | M0 | |
| T3 | N2 | M0 | |
| T4 | N2 | M0 | |
| Stage IIIC | T3 | N3 | M0 |
| T4 | N3 | M0 | |
| Stage IVA | Any T | Any N | M1a |
| Any T | Any N | M1b | |
| Stage IVB | Any T | Any N | M1c |
Procedures for Staging Lung Cancer
- There are currently multiple different procedures available to stage lung cancer.
Invasive
- EUS-NA (esophageal endoscopic ultrasound with needle aspiration)
- TBNA (transbronchial needle aspiration)
- EBUS-NA (endobronchial ultrasound with needle aspiration)
- TTNA (transthoracic needle aspiration)
- VATS staging (video assisted thoracic surgery) aka thoracoscopy
- Extended cervical mediastinoscopy
- Chamberlain procedure
Minimally Invasive
- EBUS-FNA (endobronchial ultrasound guided fine needle aspiration)
- EUS-FNA (esophageal endoscopic ultrasound guided fine needle aspiration)
Diagnostic Sequence in Lung Cancer
Initial Imaging Evaluation
- In patients with chest symptoms concerning for lung cancer or an abnormal chest radiograph, contrast-enhanced chest CT is recommended to evaluate the primary lesion and assess nodal involvement or distant metastases.[7]
- Brain imaging, preferably with MRI, should be performed in all patients diagnosed with lung cancer.[8]
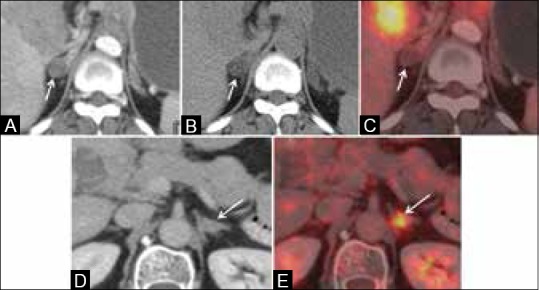
- Fluorodeoxyglucose positron emission tomography-CT (FDG PET-CT) is recommended for patients being considered for potentially curative local therapy (surgery or radiotherapy) to evaluate mediastinal lymph nodes and exclude metastatic disease.[9]
- Indeterminate pulmonary nodules should be managed according to Fleischner Society guidelines; more than 95% are benign, and the probability of malignancy is <1% for nodules <6 mm.[10]
 |
 |
 |
 |
Tissue Diagnosis and Staging
- Definitive diagnosis is established by biopsy of tumor tissue from the lung and/or lymph nodes using percutaneous or bronchoscopic techniques, often guided by endobronchial ultrasound, which also aids in staging.[7]
Molecular Profiling
- Comprehensive molecular testing should be performed for all nonsmoking individuals with lung cancer to guide targeted therapy and immunotherapy decisions.[12]
- Next-generation sequencing using DNA and RNA panels is recommended to identify actionable variants and gene rearrangements, including EGFR, ALK, ROS1, and RET.[12]
Liquid Biopsy and Disease Monitoring
- Liquid biopsy using circulating tumor DNA (ctDNA) is an emerging diagnostic approach, particularly when tumor tissue is limited.[13]
- Preoperative ctDNA status has prognostic significance, with improved 5-year overall survival reported in ctDNA-negative compared with ctDNA-high patients.
- At disease progression or treatment resistance, repeat tissue biopsy or liquid biopsy may identify resistance mechanisms, such as alterations in the PI3K/AKT/mTOR pathway or RAS, to inform subsequent therapeutic strategies.[14]
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 Purandare, NilenduC; Rangarajan, Venkatesh (2015). "Imaging of lung cancer: Implications on staging and management". Indian Journal of Radiology and Imaging. 25 (2): 109. doi:10.4103/0971-3026.155831. ISSN 0971-3026.
- ↑ 2.0 2.1 2.2 Ma, Shu-Hua; Le, Hong-Bo; Jia, Bao-hui; Wang, Zhao-Xin; Xiao, Zhuang-Wei; Cheng, Xiao-Ling; Mei, Wei; Wu, Min; Hu, Zhi-Guo; Li, Yu-Guang (2008). "Peripheral pulmonary nodules: Relationship between multi-slice spiral CT perfusion imaging and tumor angiogenesis and VEGF expression". BMC Cancer. 8 (1). doi:10.1186/1471-2407-8-186. ISSN 1471-2407.
- ↑ Gerard A. Silvestri, Lynn T. Tanoue, Mitchell L. Margolis, John Barker, Frank Detterbeck.11/30/11.The Noninvasive Staging of Non Small-cell Lung Cancer. Chestpubs. http://chestjournal.chestpubs.org/content/123/1_suppl/147S.full/
- ↑ Mountain, CF (2003). A Handbook for Staging, Imaging, and Lymph Node Classification. Charles P Young Company. Retrieved 2007-09-01. Unknown parameter
|coauthors=ignored (help) - ↑ Collins, LG (Jan 2007). "Lung cancer: diagnosis and management". American Family Physician. American Academy of Family Physicians. 75 (1): 56–63. PMID 17225705. Retrieved 2007-08-10. Unknown parameter
|coauthors=ignored (help) - ↑ Harms, A.; Kriegsmann, M.; Fink, L.; Länger, F.; Warth, A. (2017). "Die neue TNM-Klassifikation für Lungentumoren". Der Pathologe. 38 (1): 11–20. doi:10.1007/s00292-017-0268-y. ISSN 0172-8113.
- ↑ 7.0 7.1 Murphy C, Pandya T, Swanton C, Solomon BJ (November 2025). "Lung Cancer in Nonsmoking Individuals: A Review". JAMA. 334 (20): 1836–1845. doi:10.1001/jama.2025.17695. PMC 7618360 Check
|pmc=value (help). PMID 41114991 Check|pmid=value (help). - ↑ Rangachari D, Yamaguchi N, VanderLaan PA, Folch E, Mahadevan A, Floyd SR, Uhlmann EJ, Wong ET, Dahlberg SE, Huberman MS, Costa DB (April 2015). "Brain metastases in patients with EGFR-mutated or ALK-rearranged non-small-cell lung cancers". Lung Cancer. 88 (1): 108–11. doi:10.1016/j.lungcan.2015.01.020. PMC 4355240. PMID 25682925.
- ↑ Riba MB, Donovan KA, Ahmed K, Andersen B, Braun I, Breitbart WS, Brewer BW, Corbett C, Fann J, Fleishman S, Garcia S, Greenberg DB, Handzo GF, Hoofring LH, Huang CH, Hutchinson S, Johns S, Keller J, Kumar P, Lahijani S, Martin S, Niazi SK, Pailler M, Parnes F, Rao V, Salman J, Scher E, Schuster J, Teply M, Usher A, Valentine AD, Vanderlan J, Lyons MS, McMillian NR, Darlow SD (May 2023). "NCCN Guidelines® Insights: Distress Management, Version 2.2023". J Natl Compr Canc Netw. 21 (5): 450–457. doi:10.6004/jnccn.2023.0026. PMID 37156476 Check
|pmid=value (help). - ↑ [+https://jamanetwork.com/journals/jama/article-abstract/2788136 "Validate User"] Check
|url=value (help). - ↑ 11.0 11.1 11.2 11.3 Farsad M (2020). "FDG PET/CT in the Staging of Lung Cancer". Curr Radiopharm. 13 (3): 195–203. doi:10.2174/1874471013666191223153755. PMC 8206197 Check
|pmc=value (help). PMID 31868151. - ↑ 12.0 12.1 Riely GJ, Wood DE, Ettinger DS, Aisner DL, Akerley W, Bauman JR, Bharat A, Bruno DS, Chang JY, Chirieac LR, DeCamp M, Desai AP, Dilling TJ, Dowell J, Durm GA, Gettinger S, Grotz TE, Gubens MA, Juloori A, Lackner RP, Lanuti M, Lin J, Loo BW, Lovly CM, Maldonado F, Massarelli E, Morgensztern D, Mullikin TC, Ng T, Owen D, Owen DH, Patel SP, Patil T, Polanco PM, Riess J, Shapiro TA, Singh AP, Stevenson J, Tam A, Tanvetyanon T, Yanagawa J, Yang SC, Yau E, Gregory KM, Hang L (May 2024). "Non-Small Cell Lung Cancer, Version 4.2024, NCCN Clinical Practice Guidelines in Oncology". J Natl Compr Canc Netw. 22 (4): 249–274. doi:10.6004/jnccn.2204.0023. PMID 38754467 Check
|pmid=value (help). - ↑ Black JR, Bartha G, Abbott CW, Boyle SM, Karasaki T, Li B, Chen R, Harris J, Veeriah S, Colopi M, Bakir MA, Liu WK, Lyle J, Navarro FC, Northcott J, Pyke RM, Hill MS, Thol K, Huebner A, Bailey C, Colliver EC, Martínez-Ruiz C, Grigoriadis K, Pawlik P, Moore DA, Marinelli D, Shutkever OG, Murphy C, Sivakumar M, Shaw JA, Hackshaw A, McGranahan N, Jamal-Hanjani M, Frankell AM, Chen RO, Swanton C (January 2025). "Ultrasensitive ctDNA detection for preoperative disease stratification in early-stage lung adenocarcinoma". Nat Med. 31 (1): 70–76. doi:10.1038/s41591-024-03216-y. PMC 11750713 Check
|pmc=value (help). PMID 39806071 Check|pmid=value (help). - ↑ Jee J, Lebow ES, Yeh R, Das JP, Namakydoust A, Paik PK, Chaft JE, Jayakumaran G, Rose Brannon A, Benayed R, Zehir A, Donoghue M, Schultz N, Chakravarty D, Kundra R, Madupuri R, Murciano-Goroff YR, Tu HY, Xu CR, Martinez A, Wilhelm C, Galle J, Daly B, Yu HA, Offin M, Hellmann MD, Lito P, Arbour KC, Zauderer MG, Kris MG, Ng KK, Eng J, Preeshagul I, Victoria Lai W, Fiore JJ, Iqbal A, Molena D, Rocco G, Park BJ, Lim LP, Li M, Tong-Li C, De Silva M, Chan DL, Diakos CI, Itchins M, Clarke S, Pavlakis N, Lee A, Rekhtman N, Chang J, Travis WD, Riely GJ, Solit DB, Gonen M, Rusch VW, Rimner A, Gomez D, Drilon A, Scher HI, Shah SP, Berger MF, Arcila ME, Ladanyi M, Levine RL, Shen R, Razavi P, Reis-Filho JS, Jones DR, Rudin CM, Isbell JM, Li BT (November 2022). "Overall survival with circulating tumor DNA-guided therapy in advanced non-small-cell lung cancer". Nat Med. 28 (11): 2353–2363. doi:10.1038/s41591-022-02047-z. PMC 10338177 Check
|pmc=value (help). PMID 36357680 Check|pmid=value (help).