Loteprednol
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Vignesh Ponnusamy, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Loteprednol is a corticosteroid that is FDA approved for the {{{indicationType}}} of post-operative inflammation and pain following ocular surgery. Common adverse reactions include anterior chamber inflammation, eye pain, and foreign body sensation.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Post-operative Inflammation and Pain following Ocular Surgery
- Dosing Information
- Invert closed bottle and shake once to fill tip before instilling drops.
- Apply one to two drops of LOTEMAX into the conjunctival sac of the affected eye four times daily beginning the day after surgery and continuing throughout the first 2 weeks of the post-operative period.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Loteprednol in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Loteprednol in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Loteprednol in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Loteprednol in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Loteprednol in pediatric patients.
Contraindications
- LOTEMAX, as with other ophthalmic corticosteroids, is contraindicated in most viral diseases of the cornea and conjunctiva including epithelial herpes simplex keratitis (dendritic keratitis), vaccinia, and varicella, and also in mycobacterial infection of the eye and fungal diseases of ocular structures
Warnings
Precautions
- Intraocular Pressure (IOP) Increase
- Prolonged use of corticosteroids may result in glaucoma with damage to the optic nerve, defects in visual acuity and fields of vision. Steroids should be used with caution in the presence of glaucoma. If this product is used for 10 days or longer, intraocular pressure should be monitored.
- Cataracts
- Use of corticosteroids may result in posterior subcapsular cataract formation.
- Delayed Healing
- The use of steroids after cataract surgery may delay healing and increase the incidence of bleb formation. In those diseases causing thinning of the cornea or sclera, perforations have been known to occur with the use of topical steroids. The initial prescription and renewal of the medication order should be made by a physician only after examination of the patient with the aid of magnification such as slit lamp biomicroscopy and, where appropriate, fluorescein staining.
- Bacterial Infections
- Prolonged use of corticosteroids may suppress the host response and thus increase the hazard of secondary ocular infections. In acute purulent conditions of the eye, steroids may mask infection or enhance existing infection.
- Viral Infections
- Employment of a corticosteroid medication in the treatment of patients with a history of herpes simplex requires great caution. Use of ocular steroids may prolong the course and may exacerbate the severity of many viral infections of the eye (including herpes simplex).
- Fungal Infections
- Fungal infections of the cornea are particularly prone to develop coincidentally with long-term local steroid application. Fungus invasion must be considered in any persistent corneal ulceration where a steroid has been used or is in use. Fungal cultures should be taken when appropriate.
- Contact Lens Wear
- Patients should not wear contact lenses during their course of therapy with LOTEMAX
Adverse Reactions
Clinical Trials Experience
Adverse reactions associated with ophthalmic steroids include elevated intraocular pressure, which may be associated with infrequent optic nerve damage, visual acuity and field defects, posterior subcapsular cataract formation, delayed wound healing and secondary ocular infection from pathogens including herpes simplex, and perforation of the globe where there is thinning of the cornea or sclera.
The most common adverse drug reactions reported were anterior chamber inflammation (5%), eye pain (2%), and foreign body sensation (2%).
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Loteprednol in the drug label.
Drug Interactions
There is limited information regarding Loteprednol Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- Pregnancy Category C
- Loteprednol etabonate has been shown to be embryotoxic (delayed ossification) and teratogenic (increased incidence of meningocele, abnormal left common carotid artery, and limb flexures) when administered orally to rabbits during organogenesis at a dose of 3 mg/kg/day (35 times the maximum daily clinical dose), a dose which caused no maternal toxicity. The no-observed-effect-level (NOEL) for these effects was 0.5 mg/kg/day (6 times the maximum daily clinical dose). Oral treatment of rats during organogenesis resulted in teratogenicity (absent innominate artery at ≥5 mg/kg/day doses, and cleft palate and umbilical hernia at ≥50 mg/kg/day) and embryotoxicity (increased post-implantation losses at 100 mg/kg/day and decreased fetal body weight and skeletal ossification with ≥50 mg/kg/day). Treatment of rats with 0.5 mg/kg/day (6 times the maximum clinical dose) during organogenesis did not result in any reproductive toxicity. Loteprednol etabonate was maternally toxic (significantly reduced body weight gain during treatment) when administered to pregnant rats during organogenesis at doses of ≥5 mg/kg/day.
- Oral exposure of female rats to 50 mg/kg/day of loteprednol etabonate from the start of the fetal period through the end of lactation, a maternally toxic treatment regimen (significantly decreased body weight gain), gave rise to decreased growth and survival, and retarded development in the offspring during lactation; the NOEL for these effects was 5 mg/kg/day. Loteprednol etabonate had no effect on the duration of gestation or parturition when administered orally to pregnant rats at doses up to 50 mg/kg/day during the fetal period.
- There are no adequate and well controlled studies in pregnant women. LOTEMAX® should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Loteprednol in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Loteprednol during labor and delivery.
Nursing Mothers
- It is not known whether topical ophthalmic administration of corticosteroids could result in sufficient systemic absorption to produce detectable quantities in human milk. Systemic steroids appear in human milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other untoward effects. Caution should be exercised when LOTEMAX is administered to a nursing woman.
Pediatric Use
- Safety and effectiveness in pediatric patients have not been established.
Geriatic Use
There is no FDA guidance on the use of Loteprednol with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Loteprednol with respect to specific gender populations.
Race
There is no FDA guidance on the use of Loteprednol with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Loteprednol in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Loteprednol in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Loteprednol in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Loteprednol in patients who are immunocompromised.
Administration and Monitoring
Administration
- Topical
Monitoring
There is limited information regarding Monitoring of Loteprednol in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Loteprednol in the drug label.
Overdosage
Acute Overdose
Chronic Overdose
There is limited information regarding Chronic Overdose of Loteprednol in the drug label.
Pharmacology
There is limited information regarding Loteprednol Pharmacology in the drug label.
Mechanism of Action
- Corticosteroids inhibit the inflammatory response to a variety of inciting agents and probably delay or slow healing. They inhibit the edema, fibrin deposition, capillary dilation, leukocyte migration, capillary proliferation, fibroblast proliferation, deposition of collagen, and scar formation associated with inflammation. While glucocorticoids are known to bind to and activate the glucocorticoid receptor, the molecular mechanisms involved in glucocorticoid/glucocorticoid receptor-dependent modulation of inflammation are not clearly established. However, corticosteroids are thought to inhibit prostaglandin production through several independent mechanisms.
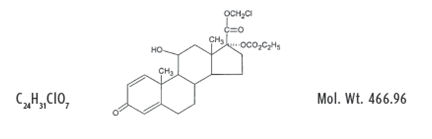
Structure
- LOTEMAX (loteprednol etabonate ophthalmic gel) 0.5% contains a sterile, topical corticosteroid for ophthalmic use. Loteprednol etabonate is a white to off-white powder.
- Loteprednol etabonate is represented by the following structural formula:
- Chemical Name:
- chloromethyl 17α-[(ethoxycarbonyl)oxy]-11β-hydroxy-3-oxoandrosta-1,4-diene-17β-carboxylate
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Loteprednol in the drug label.
Pharmacokinetics
- Loteprednol is lipid soluble and can penetrate into cells. Loteprednol etabonate is synthesized through structural modifications of prednisolone-related compounds so that it will undergo a predictable transformation to an inactive metabolite. Based upon in vivo and in vitro preclinical metabolism studies, loteprednol etabonate undergoes extensive metabolism to the inactive carboxylic acid metabolites, PJ-91 and PJ-90. The systemic exposure to loteprednol etabonate following ocular administration of LOTEMAX has not been studied in humans.
Nonclinical Toxicology
- Long-term animal studies have not been conducted to evaluate the carcinogenic potential of loteprednol etabonate. Loteprednol etabonate was not genotoxic in vitro in the Ames test, the mouse lymphoma tk assay, or in a chromosome aberration test in human lymphocytes, or in vivo in the single dose mouse micronucleus assay. Treatment of male and female rats with up to 50 mg/kg/day and 25 mg/kg/day of loteprednol etabonate, respectively, (600 and 300 times the maximum clinical dose, respectively) prior to and during mating did not impair fertility in either gender.
Clinical Studies
- In two randomized, multicenter, double-masked, parallel-group, vehicle-controlled studies in 813 subjects with, post-operative inflammation, LOTEMAX was more effective compared to its vehicle in resolving anterior chamber inflammation and pain following cataract surgery. Primary endpoints were complete resolution of anterior chamber cells (cell count of 0) and no pain at post-operative day 8.
- In these studies, LOTEMAX had a statistically significant higher incidence of subjects with complete clearing of anterior chamber cells (31% vs. 14-16%) and were pain free at post-operative day 8 (73-76% vs. 42-46%).
How Supplied
- LOTEMAX® (loteprednol etabonate ophthalmic gel) 0.5% is a sterile ophthalmic gel supplied in a white low density polyethylene plastic bottle with a white controlled drop tip and a pink polypropylene cap in the following size:
- 5 g in a 10 mL bottle (NDC 24208-503-07)
- Use only if imprinted neckband is intact.
- Storage: Store upright at 15º-25º C (59º-77º F).
Storage
There is limited information regarding Loteprednol Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Loteprednol |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Loteprednol |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Administration
- Invert closed bottle and shake once to fill tip before instilling drops.
- Risk of Contamination
- Patients should be advised not to allow the dropper tip to touch any surface, as this may contaminate the gel.
- Contact Lens Wear
- Patients should be advised not to wear contact lenses when using LOTEMAX.
- Risk of Secondary Infection
- If pain develops, redness, itching or inflammation becomes aggravated, the patient should be advised to consult a physician.
Precautions with Alcohol
- Alcohol-Loteprednol interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- LOTEMAX®[1]
Look-Alike Drug Names
There is limited information regarding Loteprednol Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Loteprednol |Pill Name=No image.jpg |Drug Name= |Pill Ingred=|+sep=; |Pill Imprint= |Pill Dosage= |Pill Color=|+sep=; |Pill Shape= |Pill Size (mm)= |Pill Scoring= |Pill Image= |Drug Author= |NDC=
}}
{{#subobject:
|Label Page=Loteprednol |Label Name=Loteprednol02.png
}}
{{#subobject:
|Label Page=Loteprednol |Label Name=Loteprednol03.png
}}