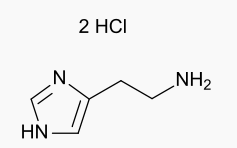
Histamine dihydrochloride
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Rabin Bista, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
NOTE: Most over the counter (OTC) are not reviewed and approved by the FDA. However, they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Overview
Histamine dihydrochloride is a OTC granule that is FDA approved for the treatment of symptoms associated with allergic reactions and Helps support healthy respiratory system. Common adverse reactions include breathing difficulties, chills, cough, diarrhoea, flushing, headaches.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications
- Helps support healthy respiratory system and alleviates symptoms associated with allergic reactions
Dosage
- Adults: Take five granules three times daily or as recommended by your healthcare practitioner.
- Children: Take three granules and follow adult directions.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Histamine dihydrochloride in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Histamine dihydrochloride in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Dosage
Children: Take three granules and follow adult directions.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Histamine dihydrochloride in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Histamine dihydrochloride in pediatric patients.
Contraindications
- Condition1
Warnings
- If pregnant or breastfeeding, ask a health professional before use.
- Keep out of reach of children.
- In case of overdose, get medical help or contact a Poison Control Center right away.
Adverse Reactions
Clinical Trials Experience
breathing difficulties, chills, cough, diarrhoea, flushing, headaches
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Histamine dihydrochloride in the drug label.
Drug Interactions
There is limited information regarding Histamine dihydrochloride Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- If pregnant or breastfeeding, ask a health professional before use.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Histamine dihydrochloride in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Histamine dihydrochloride during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Histamine dihydrochloride with respect to nursing mothers.
Pediatric Use
There is no FDA guidance on the use of Histamine dihydrochloride with respect to pediatric patients.
Geriatic Use
There is no FDA guidance on the use of Histamine dihydrochloride with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Histamine dihydrochloride with respect to specific gender populations.
Race
There is no FDA guidance on the use of Histamine dihydrochloride with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Histamine dihydrochloride in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Histamine dihydrochloride in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Histamine dihydrochloride in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Histamine dihydrochloride in patients who are immunocompromised.
Administration and Monitoring
Administration
- Oral
Monitoring
There is limited information regarding Monitoring of Histamine dihydrochloride in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Histamine dihydrochloride in the drug label.
Overdosage
- In case of overdose, get medical help or contact a Poison Control Center right away.
Pharmacology
| |
Histamine dihydrochloride
| |
| Systematic (IUPAC) name | |
| 2-(1H-imidazol-5-yl)ethanamine dihydrochloride | |
| Identifiers | |
| CAS number | |
| ATC code | L03 |
| PubChem | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 184.067 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status |
Approved for use in the EU (2008) and Israel (2011) |
| Routes | Subcutaneous injection |
Mechanism of Action
Structure
Active Ingredient
- HISTAMINUM HYDROCHLORICUM (Histamine hydrochloride) 15CH GR
- HISTAMINUM HYDROCHLORICUM (Histamine hydrochloride) 200K GR
- HISTAMINUM HYDROCHLORICUM (Histamine hydrochloride) 30K GR
- HISTAMINUM HYDROCHLORICUM (Histamine hydrochloride) 5CH GR
- HISTAMINUM HYDROCHLORICUM (Histamine hydrochloride) 6CH GR
- HISTAMINUM HYDROCHLORICUM (Histamine hydrochloride) 9CH GR
Inactive Ingredients
- Lactose,
- sucrose.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Histamine dihydrochloride in the drug label.
Pharmacokinetics
There is limited information regarding Pharmacokinetics of Histamine dihydrochloride in the drug label.
Nonclinical Toxicology
There is limited information regarding Nonclinical Toxicology of Histamine dihydrochloride in the drug label.
Clinical Studies
There is limited information regarding Clinical Studies of Histamine dihydrochloride in the drug label.
How Supplied
Storage
There is limited information regarding Histamine dihydrochloride Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Histamine dihydrochloride |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
PRINCIPAL DISPLAY PANEL
UNDA
Do not use if label perforation is broken
Homeopathic Remedy
NDC : 62106-5986-4
HISTAMINUM HYDROCHLORICUM
Histamine hydrochloride
140 granules
Indications
Helps support healthy respiratory system and alleviates symptoms associated with allergic reactions
Ingrediendts and Appearance
{{#ask: Label Page::Histamine dihydrochloride |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
QUESTIONS? 1-888-737-6925.
Precautions with Alcohol
- Alcohol-Histamine dihydrochloride interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- HISTAMINUM HYDROCHLORICUM®[1]
Look-Alike Drug Names
There is limited information regarding Histamine dihydrochloride Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.