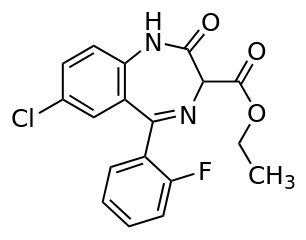
Ethyl loflazepate
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Pregnancy category |
|
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | Hepatic |
| Elimination half-life | 51-103 h |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C18H14ClFN2O3 |
| Molar mass | 360.7 |
| 3D model (JSmol) | |
| |
| |
| | |
|
WikiDoc Resources for Ethyl loflazepate |
|
Articles |
|---|
|
Most recent articles on Ethyl loflazepate Most cited articles on Ethyl loflazepate |
|
Media |
|
Powerpoint slides on Ethyl loflazepate |
|
Evidence Based Medicine |
|
Cochrane Collaboration on Ethyl loflazepate |
|
Clinical Trials |
|
Ongoing Trials on Ethyl loflazepate at Clinical Trials.gov Trial results on Ethyl loflazepate Clinical Trials on Ethyl loflazepate at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Ethyl loflazepate NICE Guidance on Ethyl loflazepate
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Ethyl loflazepate Discussion groups on Ethyl loflazepate Patient Handouts on Ethyl loflazepate Directions to Hospitals Treating Ethyl loflazepate Risk calculators and risk factors for Ethyl loflazepate
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Ethyl loflazepate |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Ethyl loflazepate[1] (marketed under the brand names Meilax, Ronlax and Victan)[2][3][4] is a drug which is a benzodiazepine derivative. It possesses anxiolytic, anticonvulsant, sedative and skeletal muscle relaxant properties.[5] In animal studies it was found to have low toxicity, although in rats evidence of pulmonary phospholipidosis occurred with pulmonary foam cells developing with long-term use of very high doses.[6] Its elimination half-life is 51-103 hours.[7] Its mechanism of action is similar to other benzodiazepines. Ethyl loflazepate also produces an active metabolite which is stronger than the parent compound.[8] Ethyl loflazepate was designed to be a prodrug for descarboxyloflazepate, its active metabolite. It is the active metabolite which is responsible for most of the pharmacological effects rather than ethyl loflazepate.[9] The main metabolites of ethyl loflazepate are descarbethoxyloflazepate, loflazepate and 3-hydroxydescarbethoxyloflazepate.[10] Accumulation of the active metabolites of ethyl loflazepate are not affected by those with renal failure or impairment.[11] The symptoms of an overdose of ethyl loflazepate include sleepiness, agitation and ataxia. Hypotonia may also occur in severe cases. These symptoms occur much more frequently and severely in children.[12] Death from therapeutic maintenance doses of ethyl loflazepate taken for 2 – 3 weeks has been reported in 3 elderly patients. The cause of death was asphyxia due to benzodiazepine toxicity.[13] High doses of the antidepressant fluvoxamine may potentiate the adverse effects of ethyl loflazepate.[14]
Ethyl loflazeplate is commercialized in Mexico, but not prescribed very often, under the trade name Victan. It is officially approved for the following conditions:[15][16]
- Anxiety
- Post-trauma anxiety
- Anxiety associated with severe neuropathic pain.
- Generalized anxiety disorder (GAD)
- Anxiety "crisis"
- Delirium tremens
See also
References
- ↑ DE Patent 2012190
- ↑ Ba, BB; Iliadis, A; Durand, A; Berger, Y; Cano, JP (Oct 1988). "New approach in bioavailability study of two formulations of ethyl loflazepate". Arzneimittel-Forschung. 38 (10): 1486–9. ISSN 0004-4172. PMID 2904268.
- ↑ Ba, BB; Iliadis, A; Cano, JP (1989). "Pharmacokinetic modeling of ethyl loflazepate (Victan) and its main active metabolites". Annals of biomedical engineering. 17 (6): 633–46. doi:10.1007/BF02367467. ISSN 0090-6964. PMID 2574017.
- ↑ "Benzodiazepine Names". non-benzodiazepines.org.uk. Retrieved 2009-04-05.
- ↑ Ueki, S; Watanabe, S; Yamamoto, T; Shibata, S; Shibata, K; Ohta, H; Ikeda, K; Kiyota, Y; Sato, Y (Nov 1983). "Behavioral effects of ethyl loflazepate and its metabolites". Nippon yakurigaku zasshi. Folia pharmacologica Japonica. 82 (5): 395–409. doi:10.1254/fpj.82.395. ISSN 0015-5691. PMID 6142848.
- ↑ Mazue, G; Berthe, J; Newmann, AJ; Brunaud, M (Oct 1981). "A toxicologic evaluation of ethyl fluclozepate (CM 6912)". International journal of clinical pharmacology, therapy, and toxicology. 19 (10): 453–72. ISSN 0174-4879. PMID 6116677.
- ↑ Cautreels, W; Jeanniot, JP (Nov 1980). "Quantitative analysis of CM 6912 (ethyl loflazepate) and its metabolites in plasma and urine by chemical ionization gas chromatography mass spectrometry. Application to pharmacokinetic studies in man". Biomedical mass spectrometry. 7 (11–12): 565–71. doi:10.1002/bms.1200071124. ISSN 0306-042X. PMID 6112027.
- ↑ Sakai, Y; Namima, M (Apr 1985). "Inhibitory effect on 3H-diazepam binding and potentiating action on GABA of ethyl loflazepate, a new minor tranquilizer". Japanese journal of pharmacology. 37 (4): 373–9. doi:10.1254/jjp.37.373. ISSN 0021-5198. PMID 2861304.
- ↑ Chambon, JP; Perio, A; Demarne, H; Hallot, A; Dantzer, R; Roncucci, R; Bizière, K (1985). "Ethyl loflazepate: a prodrug from the benzodiazepine series designed to dissociate anxiolytic and sedative activities". Arzneimittel-Forschung. 35 (10): 1573–7. ISSN 0004-4172. PMID 2866771.
- ↑ Davi, H; Guyonnet, J; Sales, Y; Cautreels, W (1985). "Metabolism of ethyl loflazepate in the rat, the dog, the baboon and in man". Arzneimittel-Forschung. 35 (7): 1061–5. ISSN 0004-4172. PMID 2864933.
- ↑ Ba, BB; Iliadis, A; Durand, A; Berger, Y; Necciari, J; Cano, JP (1989). "Effect of renal failure on the pharmacokinetics of ethyl loflazepate (Victan) in man". Fundamental & clinical pharmacology. 3 (1): 11–7. doi:10.1111/j.1472-8206.1989.tb00025.x. ISSN 0767-3981. PMID 2714728.
- ↑ Pulce C, Mollon P, Pham E, Frantz P, Descotes J (April 1992). "Acute poisonings with ethyle loflazepate, flunitrazepam, prazepam and triazolam in children". Vet Hum Toxicol. 34 (2): 141–3. ISSN 0145-6296. PMID 1354907.
- ↑ Kamijo, Y; Hayashi, I; Nishikawa, T; Yoshimura, K; Soma, K (Mar 2005). "Pharmacokinetics of the active metabolites of ethyl loflazepate in elderly patients who died of asphyxia associated with benzodiazepine-related toxicity". Journal of analytical toxicology. 29 (2): 140–4. doi:10.1093/jat/29.2.140. ISSN 0146-4760. PMID 15902983.
- ↑ Imanishi, T; Onozawa, K; Hayashi, A; Baba, J (Dec 2001). "Effects of fluvoxamine on both the desired anxiolytic effect and the adverse motor incoordination and amnesia induced by benzodiazepines". Nippon yakurigaku zasshi. Folia pharmacologica Japonica. 118 (6): 403–10. doi:10.1254/fpj.118.403. ISSN 0015-5691. PMID 11778459.
- ↑ "Victan". saludzac.gob.mx/. Retrieved 2009-12-25.
- ↑ "VICTAN". Farmacia Guerra. Retrieved 2009-12-25.
- Pages with script errors
- CS1 maint: Multiple names: authors list
- Template:drugs.com link with non-standard subpage
- Articles with changed CASNo identifier
- Articles with changed KEGG identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Benzodiazepines
- Hypnotics
- Organochlorides
- Organofluorides
- Lactams
- Drug