Ciclopirox
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Vignesh Ponnusamy, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Ciclopirox is an antifungal agent that is FDA approved for the {{{indicationType}}} of tinea pedis, tinea cruris, tinea corporis, candidiasis and tinea versicolor. Common adverse reactions include erythema, pruritus, and sensation of burning of skin.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Dermal Infections
- Ciclopirox Cream is indicated for the topical treatment of the following dermal infections: tinea pedis, tinea cruris, and tinea corporis due to Trichophyton rubrum, Trichophyton mentagrophytes, Epidermophyton floccosum, and Microsporum canis; candidiasis (moniliasis) due to Candida albicans; and tinea (pityriasis) versicolor due to Malassezia furfur.
- Gently massage Ciclopirox Cream 0.77% into the affected and surrounding skin areas twice daily, in the morning and evening. Clinical improvement with relief of pruritus and other symptoms usually occurs within the first week of treatment. If a patient shows no clinical improvement after four weeks of treatment with Ciclopirox Cream the diagnosis should be redetermined. Patients with tinea versicolor usually exhibit clinical and mycological clearing after two weeks of treatment.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ciclopirox in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Ciclopirox in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Dermal Infections
- Safety and effectiveness in pediatric patients below the age of 10 years have not been established.
- Ciclopirox Cream is indicated for the topical treatment of the following dermal infections: tinea pedis, tinea cruris, and tinea corporis due to Trichophyton rubrum, Trichophyton mentagrophytes, Epidermophyton floccosum, and Microsporum canis; candidiasis (moniliasis) due to Candida albicans; and tinea (pityriasis) versicolor due to Malassezia furfur.
- Gently massage Ciclopirox Cream 0.77% into the affected and surrounding skin areas twice daily, in the morning and evening. Clinical improvement with relief of pruritus and other symptoms usually occurs within the first week of treatment. If a patient shows no clinical improvement after four weeks of treatment with Ciclopirox Cream the diagnosis should be redetermined. Patients with tinea versicolor usually exhibit clinical and mycological clearing after two weeks of treatment.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ciclopirox in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Ciclopirox in pediatric patients.
Contraindications
- Ciclopirox Cream is contraindicated in individuals who have shown hypersensitivity to any of its components.
Warnings
- Ciclopirox Cream is not for ophthalmic use. Keep out of reach of children.
Precautions
- If a reaction suggesting sensitivity or chemical irritation should occur with the use of Ciclopirox Cream, treatment should be discontinued and appropriate therapy instituted.
Adverse Reactions
Clinical Trials Experience
- In all controlled clinical studies with 514 patients using Ciclopirox Cream and in 296 patients using the vehicle cream, the incidence of adverse reactions was low. This included pruritus at the site of application in one patient and worsening of the clinical signs and symptoms in another patient using ciclopirox cream and burning in one patient and worsening of the clinical signs and symptoms in another patient using the vehicle cream.
Postmarketing Experience
There is limited information regarding Postmarketing Experience of Ciclopirox in the drug label.
Drug Interactions
There is limited information regarding Ciclopirox Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- Pregnancy Category D
- Reproduction studies have been performed in the mouse, rat, rabbit, and monkey (via various routes of administration) at doses 10 times or more the topical human dose and have revealed no significant evidence of impaired fertility or harm to the fetus due to ciclopirox. There are, however, no adequate or well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Ciclopirox in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Ciclopirox during labor and delivery.
Nursing Mothers
- It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Ciclopirox Cream 0.77% is administered to a nursing woman.
Pediatric Use
- Safety and effectiveness in pediatric patients below the age of 10 years have not been established.
Geriatic Use
There is no FDA guidance on the use of Ciclopirox with respect to geriatric patients.
Gender
There is no FDA guidance on the use of Ciclopirox with respect to specific gender populations.
Race
There is no FDA guidance on the use of Ciclopirox with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Ciclopirox in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Ciclopirox in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Ciclopirox in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Ciclopirox in patients who are immunocompromised.
Administration and Monitoring
Administration
- Topical
Monitoring
There is limited information regarding Monitoring of Ciclopirox in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Ciclopirox in the drug label.
Overdosage
Chronic Overdose
There is limited information regarding Chronic Overdose of Ciclopirox in the drug label.
Pharmacology
| |
Ciclopirox
| |
| Systematic (IUPAC) name | |
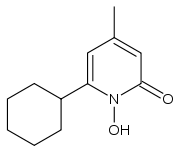
| 6-cyclohexyl-1-hydroxy-4-methylpyridin-2(1H)-one | |
| Identifiers | |
| CAS number | |
| ATC code | D01 G01AX12 (WHO) |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 207.269 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | <5% with prolonged use |
| Protein binding | 94 to 97% |
| Metabolism | ? |
| Half life | 1.7 hours |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
B |
| Legal status |
[[Prescription drug|Template:Unicode-only]](US) Rx-only (CA) |
| Routes | Topical (applied as a nail lacquer or shampoo) |
Mechanism of Action
- Ciclopirox is a broad-spectrum, antifungal agent that inhibits the growth of pathogenic dermatophytes, yeasts, and Malassezia furfur. Ciclopirox exhibits fungicidal activity in vitro against isolates of Trichophyton rubrum, Trichophyton mentagrophytes, Epidermophyton floccosum, Microsporum canis, and Candida albicans.
Structure
- Ciclopirox Cream 0.77% is for topical use.
- Each gram of Ciclopirox cream 0.77% contains 7.70 mg of Ciclopirox (as Ciclopirox Olamine) in a water miscible vanishing cream base consisting of purified water USP, cetyl alcohol NF, mineral oil USP, octyldodecanol NF, stearyl alcohol NF, cocamide DEA, polysorbate 60 NF, myristyl alcohol, sorbitan monostearate NF, lactic acid USP, and benzyl alcohol NF (1%) as preservative.
- Ciclopirox Cream contains a synthetic, broad-spectrum, antifungal agent ciclopirox (as ciclopirox olamine).
- The chemical name is 6-cyclohexyl-1-hydroxy-4-methyl-2(1H)-pyridone, 2-aminoethanol salt.
- The CAS Registry Number is 41621-49-2.
- The chemical structure is:
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Ciclopirox in the drug label.
Pharmacokinetics
- Pharmacokinetic studies in men with tagged ciclopirox solution in polyethylene glycol 400 showed an average of 1.3% absorption of the dose when it was applied topically to 750 cm2 on the back followed by occlusion for 6 hours. The biological half-life was 1.7 hours and excretion occurred via the kidney. Two days after application only 0.01% of the dose applied could be found in the urine. Fecal excretion was negligible.
- Penetration studies in human cadaverous skin from the back, with ciclopirox cream 0.77% with tagged ciclopirox showed the presence of 0.8 to 1.6% of the dose in the stratum corneum 1.5 to 6 hours after application. The levels in the dermis were still 10 to 15 times above the minimum inhibitory concentrations.
- Autoradiographic studies with human cadaverous skin showed that ciclopirox penetrates into the hair and through the epidermis and hair follicles into the sebaceous glands and dermis, while a portion of the drug remains in the stratum corneum.
- Draize Human Sensitization Assay, 21-Day Cumulative Irritancy study, Phototoxicity study, and Phot-Draize study conducted in a total of 142 healthy male subjects showed no contact sensitization of the delayed hypersensitivity type, no irritation, no phototoxicity, and no photo-contact sensitization due to Ciclopirox Cream 0.77%.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- A carcinogenicity study in female mice dosed cutaneously twice per week for 50 weeks followed by a 6-month drug-free observation period prior to necropsy revealed no evidence of tumors at the application site. The following in vitro and in vivo genotoxicity tests have been conducted with ciclopirox olamine: studies to evaluate gene mutation in the Ames SalmonellaIMammalian Microsome Assay (negative) and Yeast Saccharomyces Cerevisiae Assay (negative) and studies to evaluate chromosome aberrations in vivo in the Mouse Dominant Lethal Assay and in the Mouse Micronucleus Assay at 500 mg/kg (negative). The following battery of in vitro genotoxicity tests were conducted with ciclopirox: a chromosome aberration assay in V79 Chinese Hamster Cells, with and without metabolic activation (positive); a gene mutation assay in the HGPRT - test with V79 Chinese Hamster Cells (negative); and a primary DNA damage assay (i.e., unscheduled DNA Synthesis Assay in A549 Human Cells (negative)). An in vitro Cell Transformation Assay in BALB/C3T3 Cells was negative for cell transformation. In an in vivo Chinese Hamster Bone Marrow Cytogenetic Assay, ciclopirox was negative for chromosome aberrations at 5000 mg/kg.
Clinical Studies
There is limited information regarding Clinical Studies of Ciclopirox in the drug label.
How Supplied
- Ciclopirox Cream 0.77% (Ciclopirox Olamine Cream USP) is supplied in:
- NDC 0168-0313-15 15 gram tube
- NDC 0168-0313-30 30 gram tube
- NDC 0168-0313-90 90 gram tube
- Store at 20°-25°C (68°-77°F).
Storage
There is limited information regarding Ciclopirox Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Ciclopirox |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Ciclopirox |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- The patient should be told to:
- Use the medication for the full treatment time even though symptoms may have improved and notify the physician if there is no improvement after four weeks.
- Inform the physician if the area of application shows signs of increased irritation (redness, itching, burning, blistering, swelling, or oozing) indicative of possible sensitization.
- Avoid the use of occlusive wrappings or dressings.
Precautions with Alcohol
- Alcohol-Ciclopirox interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- CICLOPIROX OLAMINE ®[1]
Look-Alike Drug Names
There is limited information regarding Ciclopirox Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Ciclopirox |Pill Name=No image.jpg |Drug Name= |Pill Ingred=|+sep=; |Pill Imprint= |Pill Dosage= |Pill Color=|+sep=; |Pill Shape= |Pill Size (mm)= |Pill Scoring= |Pill Image= |Drug Author= |NDC=
}}
{{#subobject:
|Label Page=Ciclopirox |Label Name=Ciclopirox02.png
}}
{{#subobject:
|Label Page=Ciclopirox |Label Name=Ciclopirox03.png
}}
{{#subobject:
|Label Page=Ciclopirox |Label Name=Ciclopirox04.png
}}
