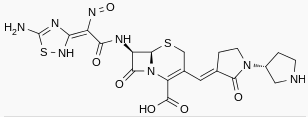
Ceftobiprole medocaril
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C20H22N8O6S2 |
| Molar mass | 534.568 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Ceftobiprole (Zeftera/Zevtera) is a fifth-generation[2] cephalosporin antibiotic with activity against methicillin-resistant Staphylococcus aureus, penicillin-resistant Streptococcus pneumoniae, Pseudomonas aeruginosa, and enterococci.[3][4][5] It was discovered by Basilea Pharmaceutica[6] and was developed by Johnson & Johnson Pharmaceutical Research and Development.[7] It has been shown to be statistically noninferior to the combination of vancomycin and ceftazidime for the treatment of skin and soft tissue infections.[8]
It has been described as a fifth-generation cephalosporin,[9][10] though acceptance for this terminology is not universal.
Pharmacology
Like other cephalosporins, ceftobiprole inhibits bacterial growth by blocking penicillin-binding protein, a key enzyme involved in cell wall synthesis. Ceftobiprole inhibits the 2a penicillin-binding protein (PBP) of methicillin-resistant Staphylococcus aureus and the 2x PBP of Streptococcus pneumoniae,[4] as well as the classic PBP-2 of MSSA. Ceftobiprole is resistant to staphylococcal β-lactamase.[6]
Mode of administration
Ceftobiprole is given intravenously; it cannot be given by mouth.
Regulatory approvals
Ceftobiprole has been approved for use in Canada, Switzerland, and the European Union.[11] It is under review by regulatory authorities in the United States, Australia, Russia, and South Africa.[12] In November 2008, the US FDA declined to approve ceftobiprole, citing data integrity concerns with two of the supporting studies,[13] and prompting Basilea to sue Johnson & Johnson for breach of license agreement on February 2009.[14]
Synonyms
References
- ↑ WHO International Working Group for Drug Statistics Methodology (August 27, 2008). "ATC/DDD Classification (FINAL): New ATC 5th level codes". WHO Collaborating Centre for Drug Statistics Methodology. Retrieved 2008-09-05.
- ↑ Kollef MH (December 2009). "New antimicrobial agents for methicillin-resistant Staphylococcus aureus". Crit Care Resusc. 11 (4): 282–6. PMID 20001879.
- ↑ Yun HC, Ellis MW, Jorgensen JH (2007). "Activity of ceftobiprole against community-associated methicillin-resistant Staphylococcus aureus isolates recently recovered from US military trainees". Diagnostic Microbiology and Infectious Disease. 59 (4): 463–6. doi:10.1016/j.diagmicrobio.2007.06.023. PMID 17911001.
- ↑ 4.0 4.1 Widmer A (2008). "Ceftobiprole: A new option for treatment of skin and soft-tissue infections due to methicillin-resistant Staphylococcus aureus". Clin Infect Dis. 46 (5): 656–8. doi:10.1086/526528. PMID 18225983.
- ↑ Noel GJ, Bush K, Bagchi P, Ianus J, Strauss RS (2008). "A randomized, double-blind trial comparing ceftobiprole medocaril with vancomycin plus ceftazidime plus ceftazidime for the treatment of patients with complicated skin and skin-structure infections". Clin Infect Dis. 46 (5): 647–55. doi:10.1086/526527. PMID 18225981.
- ↑ 6.0 6.1 6.2 Hebeisen P, Heinze-Krauss I, Angehrn P; et al. (2001). "In vitro and in vivo properties of Ro63-9141, a novel broad-spectrum cephalosporin with activity against methicillin-resistant staphylococci". Antimicrob Agents Chemother. 45 (3): 825–36. doi:10.1128/AAC.45.3.825-836.2001. PMC 90381. PMID 11181368.
- ↑ Basilea.com
- ↑ Noel, Gary J.; Karen Bush; Partha Bagchi; Juliana Ianus; Richard S. Strauss (2008). "A Randomized, Double-Blind Trial Comparing Ceftobiprole Medocaril with Vancomycin plus Ceftazidime for the Treatment of Patients with Complicated Skin and Skin-Structure Infections". Clin Infect Dis. 46 (5): 647-655. doi:10.1086/526527. PMID 18225981.
|access-date=requires|url=(help) - ↑ Widmer AF (March 2008). "Ceftobiprole: a new option for treatment of skin and soft-tissue infections due to methicillin-resistant Staphylococcus aureus". Clin. Infect. Dis. 46 (5): 656–8. doi:10.1086/526528. PMID 18225983.
- ↑ Kosinski MA, Joseph WS (July 2007). "Update on the treatment of diabetic foot infections". Clin Podiatr Med Surg. 24 (3): 383–96, vii. doi:10.1016/j.cpm.2007.03.009. PMID 17613382.
- ↑ Basilea antibiotic to treat pneumonia wins European backing. Reuters, ZURICH | Wed Oct 23, 2013.
- ↑ Basilea superbug drug approved in Canada, Reuters News, June 30, 2008
- ↑ http://www.dancewithshadows.com/pillscribe/ceftobiprole-antibiotic-to-fight-tougher-bacterial-infections-fails-to-win-approval-in-us/
- ↑ "Basilea Pharmaceutica Ltd. announces that the U.S. Food and Drug Administration (FDA) issued to the sponsor, Johnson & Johnson Pharmaceutical Research and Development, L.L.C. (Johnson & Johnson PRD), a Complete Response Letter on ceftobiprole for the treatment of complicated skin and skin structure infections (cSSSI" (Press release). Basilea Pharmaceutica. 2009-07-02. Retrieved February 2, 2010.
- ↑ Jones RN, Deshpande LM, Mutnick AH, Biedenbach DJ (2002). "In vitro evaluation of BAL9141, a novel parenteral cephalosporin active against oxacillin-resistant staphylococci". J Antimicrob Chemother. 50 (6): 915–932. doi:10.1093/jac/dkf249. PMID 12461013.
- Pages with script errors
- CS1 maint: Multiple names: authors list
- CS1 maint: Explicit use of et al.
- Pages using citations with accessdate and no URL
- Template:drugs.com link with non-standard subpage
- Drugs with non-standard legal status
- Articles with changed CASNo identifier
- Articles with changed EBI identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Chemical pages without DrugBank identifier
- Drugboxes which contain changes to verified fields
- Drug
- Cephalosporin antibiotics
- Thiadiazoles
- Oximes
- Pyrrolidines
- Pyrrolidones