Penicillin binding proteins
Penicillin-binding proteins (PBPs) are a group of proteins that are characterized by their affinity for and binding of penicillin. They are a normal constitutent of many bacteria; the name just reflects the way by which the protein was discovered. All beta-lactam antibiotics bind to PBP to have their effect of preventing cell wall construction by the bacterium.
Diversity
There are a large number of PBPs, usually several in each organism, and they are found as both membrane-bound and cytoplasmic proteins. For example, Spratt (1977) reports that six different PBPs are routinely detected in all strains of E. coli ranging in molecular weight from 40000 to 91000. The different PBPs occur in different numbers per cell and have varied affinities for penicillin (see appendix). The PBPs are usually broadly classified into high-molecular-weight (HMW) and low-molecular-weight (LMW) categories (Basu and Chattopadhyay et al, 1992).
Function
PBPs are all involved in the final stages of the synthesis of peptidoglycan, which is the major component of bacterial cell walls. Bacterial cell wall synthesis is essential to growth, cell division (thus reproduction) and maintaining the cellular structure in bacteria. Inhibition of PBPs leads to irregularities in cell wall structure such as elongation, lesions, loss of selective permeability, and eventual cell death and lysis.
PBPs have been shown to catalyse a number of reactions involved in the process of synthesising cross-linked peptidoglycan from lipid intermediates and mediating the removal of D-alanine from the precursor of peptidoglycan. Purified enzymes have been shown to catalyse the following reactions: D-alanine carboxypeptidase, peptidoglycan transpeptidase, and peptidoglycan endopeptidase. In all bacteria that have been studied, enzymes have been shown to catalyse more than one of the above reactions (Spratt, 1977). The enzyme has a penicillin-insensitive transglycosylase N-terminal domain (involved in formation of linear glycan strands) and a penicillin-sensitive transpeptidase C-terminal domain (involved in cross-linking of the peptide subunits) and the serine at the active site is conserved in all members of the PBP family (Basu and Chattopadhyay et al, 1992).
Illustrations
Figure 1. The carboxypeptidase reaction catalysed by PBPs, where E is the free enzyme, S is the substrate, ES is the enzyme + substrate complex (non-covalent), ES* is the enzyme + substrate complex (covalently-bound), EP is the enzyme + substrate complex with the hydrolysed peptide (non-covalent) and P is the free peptide product (Disteche and Bouille et al, 1982).
Figure 2. The peptidoglycan layer in the bacterial cell wall is a crystal lattice structure formed from linear chains of two alternating sugars N-acetyl glucosamine and N-acetyl muramic acid. PBPs (the transpeptideases) cross link amino acids in different sugar chains to form this three-dimensional structure, which is very strong and rigid. The specific amino acid sequence and molecular structure vary between species. There are up to 40 layers of this polymer in gram positive bacteria.
Antibiotics
PBPs bind β-lactam antibiotics because they are similar in chemical structure to the modular pieces that form the peptidoglycan (Disteche and Bouille et al, 1982). When they bind to penicillin, the β-lactam amide bond is ruptured to form a covalent bond with the serine residue at the PBPs active site. This is an irreversible reaction and inactivates the enzyme.
There has been a great deal of research into PBPs because of their role in antibiotics and resistance. Bacterial cell wall synthesis and the role of PBPs in its synthesis is a very good target for drugs of selective toxicity because the metabolic pathways and enzymes are unique to bacteria (Chanbers, 1999). Resistance to antibiotics has come about through overproduction of PBPs and formation of PBPs that have low affinity for penicillins (among other mechanisms such as lactamase production). Research on PBPs has led to the discovery our new semi-synthetic β-lactams, wherein altering the side-chains on the original penicillin molecule has increased the affinity of PBPs for penicillin, and, thus, increased effectiveness in bacteria with developing resistance.
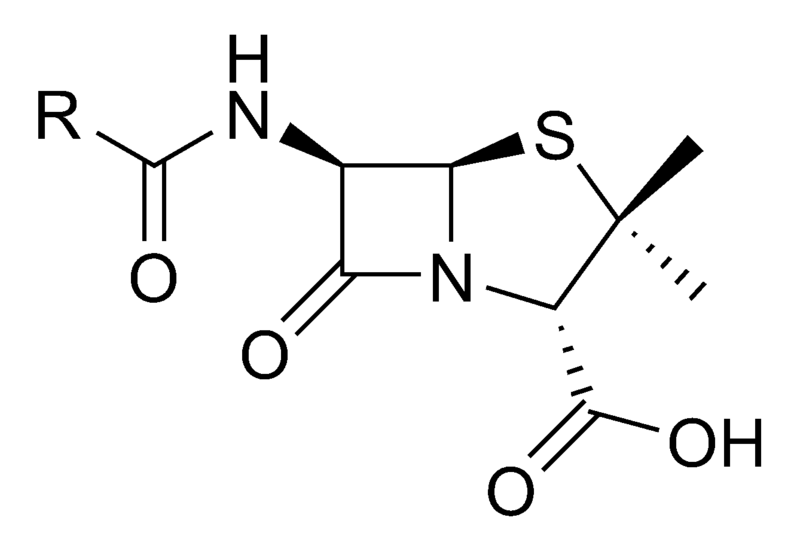
The β-lactam ring is an analogous structure to all β-lactam antibiotics. Shown here is the core structure of penicillin, the square in the middle with an oxygen double-bonded to it is the β-lactam ring (Walsh, 2003)
References
- Basu J., Chattopadhyay R., Kundu M., and Chakrabarti P. (1992) Purification and partial characterization of a penicillin binding protein from Mycobacterium smegmatis. American Society for Microbiology – Journal of Bacteriology. Vol 174, No 14 pp 4829–4832.
- Chanbers H. F. (1999) Penicillin-binding protein-mediated resistance in pneumococci and staphylococci. The Journal of Infectious Diseases. Chicago. Vol.179 Pp 353– 360
- Spratt B. G. (1977) Properties of the penicillin binding proteins of Escherichia coli K12. Journal of Biochem. No. 14, 342-352.
- Walsh C. (2003) Antibiotics actions, origins and resistance. American society for microbiology, Washington DC.
Appendix
(This table may be found in the original article by clicking the 'Spratt 1977' link above)
| Protein | Apparent molecular weight | Binding of penicillin( % total ) | Molecules/cell |
|---|---|---|---|
| 1 | 91000 | 8.1 | 230 |
| 2 | 66000 | 0.7 | 20 |
| 3 | 60000 | 1.9 | 50 |
| 4 | 49000 | 4.0 | 110 |
| 5 | 42000 | 64.7 | 1800 |
| 6 | 40000 | 20.6 | 570 |