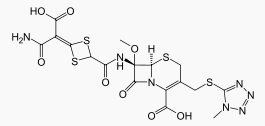
Cefotetan disodium
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Gloria Picoy [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Cefotetan disodium is a 2nd generation cephalosporin that is FDA approved for the treatment of urinary tract infection, lower respiratory tract infection, skin and skin structure infections, gynecologic infection, intra-abdominal infection, and bone and joint infection; and for prophylaxis of postoperative infection. Common adverse reactions include diarrhea and nausea.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Treatment
Cefotetan for Injection is indicated for the therapeutic treatment of the following infections when caused by susceptible strains of the designated organisms:
- Urinary Tract Infections caused by E. coli, Klebsiella spp (including K. pneumoniae), Proteus mirabilis and Proteus spp (which may include the organisms now called Proteus vulgaris, Providencia rettgeri, and Morganella morganii).
- Dosage: 500 mg every 12 hours IV; or 1 or 2 g every 24 hours IV; or 1 or 2 g every 12 hours IV
- Lower Respiratory Tract Infections caused by Streptococcus pneumoniae, Staphylococcus aureus (penicillinase- and nonpenicillinase-producing strains), Haemophilus influenzae (including ampicillin-resistant strains), Klebsiella species (including K. pneumoniae), E. coli, Proteus mirabilis, and Serratia marcescens.
- Dosage: 1 or 2 g every 12 hours IV for 5 to 10 days
- Dosage in severe cases: 2 g every 12 hours IV for 5 to 10 days
- Dosage in life-threatening 3 g every 12 hours IV for 5 to 10 days
- Skin and Skin Structure Infections due to Staphylococcus aureus (penicillinase- and nonpenicillinase-producing strains), Staphylococcus epidermidis, Streptococcus pyogenes, Streptococcus species (excluding enterococci), Escherichia coli, Klebsiella pneumoniae, Peptococcus niger, Peptostreptococcus species.
- Dosage for Mild - Moderate cases: 2 g every 24 hours IV or 1 g every 12 hours IV for 5 to 10 days
- Dosage for severe cases: 2 g every 12 hours IV for 5 to 10 days
- Gynecologic Infections caused by Staphylococcus aureus (including penicillinase- and nonpenicillinase-producing strains), Staphylococcus epidermidis, Streptococcus species (excluding enterococci), Streptococcus agalactiae, E. coli, Proteus mirabilis, Neisseria gonorrhoeae, Bacteroides species (excluding B. distasonis, B. ovatus, B. thetaiotaomicron), Fusobacterium species*, and gram-positive anaerobic cocci (including Peptococcus niger and Peptostreptococcus species).
- Dosage: 1 or 2 g every 12 hours IV for 5 to 10 days
- Dosage in severe cases: 2 g every 12 hours IV for 5 to 10 days
- Dosage in life-threatening 3 g every 12 hours IV for 5 to 10 days
Cefotetan, like other cephalosporins, has no activity against Chlamydia trachomatis. Therefore, when cephalosporins are used in the treatment of pelvic inflammatory disease, and C. trachomatis is one of the suspected pathogens, appropriate antichlamydial coverage should be added.
- Intra-abdominal lnfections caused by E. coli, Klebsiella species (including K. pneumoniae), Streptococcus species (excluding enterococci), Bacteroides species (excluding B. distasonis, B. ovatus, B. thetaiotaomicron) and Clostridium species.
- Dosage: 1 or 2 g every 12 hours IV for 5 to 10 days
- Dosage in severe cases: 2 g every 12 hours IV for 5 to 10 days
- Dosage in life-threatening 3 g every 12 hours IV for 5 to 10 days
- Bone and Joint Infections caused by Staphylococcus
- Dosage: 1 or 2 g every 12 hours IV for 5 to 10 days
- Dosage in severe cases: 2 g every 12 hours IV for 5 to 10 days
- Dosage in life-threatening 3 g every 12 hours IV for 5 to 10 days
Prophylaxis
- The preoperative administration of cefotetan may reduce the incidence of certain postoperative infections in patients undergoing surgical procedures that are classified as clean contaminated or potentially contaminated (e.g., cesarean section, abdominal or vaginal hysterectomy, transurethral surgery, biliary tract surgery, and gastrointestinal surgery).
- Dosage: 1 or 2 g once, intravenously, 30 to 60 minutes prior to surgery
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Cefotetan disodium in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Cefotetan disodium in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Safety and effectiveness in pediatric patients have not been established.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Cefotetan disodium in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Cefotetan disodium in pediatric patients.
Contraindications
- Patients with a known allergy to the cephalosporin group of antibiotics
- Patients who have experienced a cephalosporin associated hemolytic anemia
Warnings
BEFORE THERAPY WITH CEFOTETAN IS INSTITUTED, CAREFUL INQUIRY SHOULD BE MADE TO DETERMINE WHETHER THE PATIENT HAS HAD PREVIOUS HYPERSENSITIVITY REACTIONS TO CEFOTETAN, CEPHALOSPORINS, PENICILLINS, OR OTHER DRUGS. IF THIS PRODUCT IS TO BE GIVEN TO PENICILLIN-SENSITIVE PATIENTS, CAUTION SHOULD BE EXERCISED BECAUSE CROSS-HYPERSENSITIVITY AMONG BETA-LACTAM ANTIBIOTICS HAS BEEN CLEARLY DOCUMENTED AND MAY OCCUR IN UP TO 10% OF PATIENTS WITH A HISTORY OF PENICILLIN ALLERGY. IF AN ALLERGIC REACTION TO CEFOTETAN OCCURS, DISCONTINUE THE DRUG. SERIOUS ACUTE HYPERSENSITIVITY REACTIONS MAY REQUIRE TREATMENT WITH EPINEPHRINE AND OTHER EMERGENCY MEASURES, INCLUDING OXYGEN, INTRAVENOUS FLUIDS, INTRAVENOUS ANTIHISTAMINES, CORTICOSTEROIDS, PRESSOR AMINES, AND AIRWAY MANAGEMENT, AS CLINICALLY INDICATED.
AN IMMUNE MEDIATED HEMOLYTIC ANEMIA HAS BEEN OBSERVED IN PATIENTS RECEIVING CEPHALOSPORIN CLASS ANTIBIOTICS. SEVERE CASES OF HEMOLYTIC ANEMIA, INCLUDING FATALITIES, HAVE BEEN REPORTED IN ASSOCIATION WITH THE ADMINISTRATION OF CEFOTETAN. SUCH REPORTS ARE UNCOMMON. THERE APPEARS TO BE AN INCREASED RISK OF DEVELOPING HEMOLYTIC ANEMIA ON CEFOTETAN RELATIVE TO OTHER CEPHALOSPORINS OF AT LEAST 3 FOLD. IF A PATIENT DEVELOPS ANEMIA ANYTIME WITHIN 2 TO 3 WEEKS SUBSEQUENT TO THE ADMINISTRATION OF CEFOTETAN, THE DIAGNOSIS OF A CEPHALOSPORIN ASSOCIATED ANEMIA SHOULD BE CONSIDERED AND THE DRUG STOPPED UNTIL THE ETIOLOGY IS DETERMINED WITH CERTAINTY. BLOOD TRANSFUSIONS MAY BE CONSIDERED AS NEEDED.
PATIENTS WHO RECEIVE COURSES OF CEFOTETAN FOR TREATMENT OR PROPHYLAXIS OF INFECTIONS SHOULD HAVE PERIODIC MONITORING FOR SIGNS AND SYMPTOMS OF HEMOLYTIC ANEMIA INCLUDING A MEASUREMENT OF HEMATOLOGICAL PARAMETERS WHERE APPROPRIATE.
Clostridium difficile associated diarrhea (CDAD) has been reported with use of nearly all antibacterial agents, including cefotetan, and may range in severity from mild diarrhea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon leading to overgrowth of C. difficile.
C. difficile produces toxins A and B which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over two months after the administration of antibacterial agents.
If CDAD is suspected or confirmed, ongoing antibiotic use not directed against C. difficile may need to be discontinued. Appropriate fluid and electrolyte management, protein supplementation, antibiotic treatment of C. difficile, and surgical evaluation should be instituted as clinically indicated.
In common with many other broad-spectrum antibiotics, cefotetan may be associated with a fall in prothrombin activity and, possibly, subsequent bleeding. Those at increased risk include patients with renal or hepatobiliary impairment or poor nutritional state, the elderly, and patients with cancer. Prothrombin time should be monitored and exogenous vitamin K administered as indicated.
Adverse Reactions
Clinical Trials Experience
In clinical studies, the following adverse effects were considered related to cefotetan therapy. Those appearing in italics have been reported during postmarketing experience.
- Gastrointestinal: symptoms occurred in 1.5% of patients, the most frequent were diarrhea (1 in 80) and nausea (1 in 700); pseudomembranous colitis. Onset of pseudomembranous colitis symptoms may occur during or after antibiotic treatment or surgical prophylaxis.
- Hematologic: laboratory abnormalities occurred in 1.4% of patients and included eosinophilia (1 in 200), positive direct Coombs test (1 in 250), and thrombocytosis (1 in 300); agranulocytosis, hemolytic anemia, leukopenia, thrombocytopenia, and prolonged prothrombin time with or without bleeding.
- Hepatic: enzyme elevations occurred in 1.2% of patients and included a rise in ALT (SGPT) (1 in 150), AST (SGOT) (1 in 300), alkaline phosphatase (1 in 700), and LDH (1 in 700).
- Hypersensitivity: reactions were reported in 1.2% of patients and included rash (1 in 150) and itching (1 in 700); anaphylactic reactions and urticaria.
- Local : effects were reported in less than 1% of patients and included phlebitis at the site of injection (1 in 300), and discomfort (1 in 500).
- Renal : Elevations in BUN and serum creatinine have been reported.
- Urogenital : Nephrotoxicity has rarely been reported.
- Miscellaneous : Fever
In addition to the adverse reactions listed above which have been observed in patients treated with cefotetan, the following adverse reactions and altered laboratory tests have been reported for cephalosporin-class antibiotics: pruritus, Stevens-Johnson syndrome, erythema multiforme, toxic epidermal necrolysis, vomiting, abdominal pain, colitis, superinfection, vaginitis including vaginal candidiasis, renal dysfunction, toxic nephropathy, hepatic dysfunction including cholestasis, aplastic anemia, hemorrhage, elevated bilirubin, pancytopenia, and neutropenia.
Several cephalosporins have been implicated in triggering seizures, particularly in patients with renal impairment, when the dosage was not reduced. If seizures associated with drug therapy occur, the drug should be discontinued. Anticonvulsant therapy can be given if clinically indicated.
Postmarketing Experience
There is limited information regarding Cefotetan disodium Postmarketing Experience in the drug label.
Drug Interactions
Increases in serum creatinine have occurred when cefotetan was given alone. If cefotetan and an aminoglycoside are used concomitantly, renal function should be carefully monitored, because nephrotoxicity may be potentiated.
Drug/Laboratory Test Interactions
The administration of cefotetan may result in a false positive reaction for glucose in the urine using Clinitest®‡, Benedict's solution, or Fehling's solution. It is recommended that glucose tests based on enzymatic glucose oxidase be used.
As with other cephalosporins, high concentrations of cefotetan may interfere with measurement of serum and urine creatinine levels by Jaffé reaction and produce false increases in the levels of creatinine reported.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): B
Reproduction studies have been performed in rats and monkeys at doses up to 20 times the human dose and have revealed no evidence of impaired fertility or harm to the fetus due to cefotetan. There are, however, no adequate and well-controlled studies in pregnant women. Because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if clearly needed.
Pregnancy Category (AUS): B1
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Cefotetan disodium in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Cefotetan disodium during labor and delivery.
Nursing Mothers
Cefotetan is excreted in human milk in very low concentrations. Caution should be exercised when cefotetan is administered to a nursing woman.
Pediatric Use
Safety and effectiveness in pediatric patients have not been established.
Geriatic Use
Of the 925 subjects who received cefotetan in clinical studies, 492 (53%) were 60 years and older, while 76 (8%) were 80 years and older. No overall differences in safety or effectiveness were observed between these subjects and younger subjects, and the other reported clinical experience has not identified differences in responses between elderly and younger patients, but greater sensitivity of some older individuals cannot be ruled out.
This drug is known to be substantially excreted by the kidney, and the risk of toxic reactions to this drug may be greater in patients with impaired renal function. Because elderly patients are more likely to have decreased renal function, care should be taken in dose selection, and it may be useful to monitor renal function.
Gender
There is no FDA guidance on the use of Cefotetan disodium with respect to specific gender populations.
Race
There is no FDA guidance on the use of Cefotetan disodium with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Cefotetan disodium in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Cefotetan disodium in patients with hepatic impairment.
Females of Reproductive Potential and Males
Cefotetan has adverse affects on the testes of prepubertal rats. Subcutaneous administration of 500 mg/kg/day (approximately 8 to 16 times the usual adult human dose) on days 6 to 35 of life (thought to be developmentally analogous to late childhood and prepuberty in humans) resulted in reduced testicular weight and seminiferous tubule degeneration in 10 of 10 animals. Affected cells included spermatogonia and spermatocytes; Sertoli and Leydig cells were unaffected. Incidence and severity of lesions were dose-dependant; at 120 mg/kg/day (approximately 2 to 4 times the usual human dose) only 1 of 10 treated animals was affected, and the degree of degeneration was mild.
Similar lesions have been observed in experiments of comparable design with other methylthiotetrazole-containing antibiotics and impaired fertility has been reported, particularly at high dose levels. No testicular effects were observed in 7-week-old rats treated with up to 1000 mg/kg/day SC for 5 weeks, or in infant dogs (3 weeks old) that received up to 300 mg/kg/day IV for 5 weeks. The relevance of these findings to humans is unknown.
Immunocompromised Patients
There is no FDA guidance one the use of Cefotetan disodium in patients who are immunocompromised.
Administration and Monitoring
Administration
Intravenous
Monitoring
There is limited information regarding Cefotetan disodium Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Cefotetan disodium and IV administrations.
Overdosage
Information on overdosage with cefotetan in humans is not available. If overdosage should occur, it should be treated symptomatically and hemodialysis considered, particularly if renal function is compromised.
Pharmacology
Mechanism of Action
The bactericidal action of cefotetan results from inhibition of cell wall synthesis. Cefotetan has in vitro activity against a wide range of aerobic and anaerobic gram-positive and gram-negative organisms. The methoxy group in the 7-alpha position provides cefotetan with a high degree of stability in the presence of beta-lactamases including both penicillinases and cephalosporinase of gram-negative bacteria.
Cefotetan has been shown to be active against most strains of the following organisms both in vitro and in clinical infections.
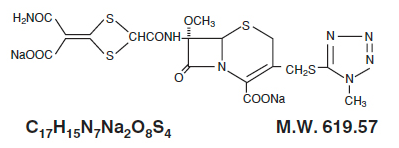
Structure
Structural formula:
Pharmacodynamics
There is limited information regarding Cefotetan disodium Pharmacodynamics in the drug label.
Pharmacokinetics
High plasma levels of cefotetan are attained after intravenous administration of single doses to normal volunteers.
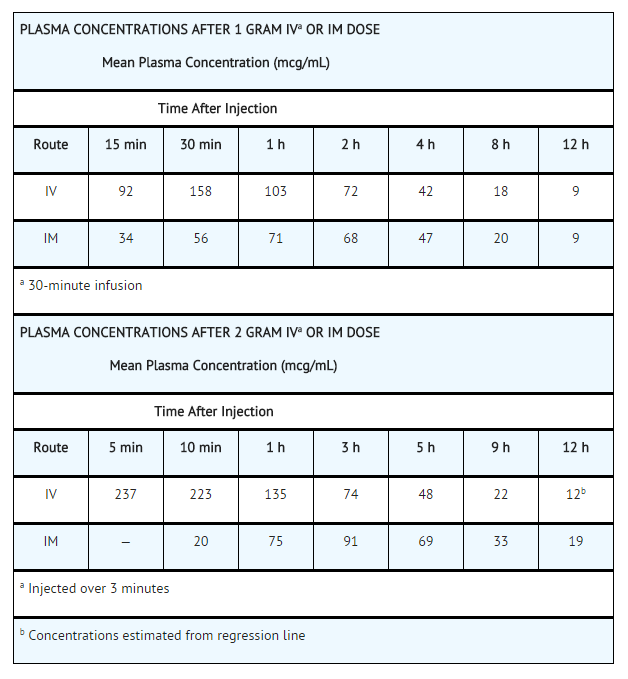
The plasma elimination half-life of cefotetan is 3 to 4.6 hours after intravenous administration.
Repeated administration of cefotetan does not result in accumulation of the drug in normal subjects.
Cefotetan is 88% plasma protein bound.
No active metabolites of cefotetan have been detected; however, small amounts (less than 7%) of cefotetan in plasma and urine may be converted to its tautomer, which has antimicrobial activity similar to the parent drug.
In normal patients, from 51% to 81% of an administered dose of cefotetan is excreted unchanged by the kidneys over a 24 hour period, which results in high and prolonged urinary concentrations. Following intravenous doses of 1 gram and 2 grams, urinary concentrations are highest during the first hour and reach concentrations of approximately 1700 and 3500 mcg/mL, respectively.
In volunteers with reduced renal function, the plasma half-life of cefotetan is prolonged. The mean terminal half-life increases with declining renal function, from approximately 4 hours in volunteers with normal renal function to about 10 hours in those with moderate renal impairment. There is a linear correlation between the systemic clearance of cefotetan and creatinine clearance. When renal function is impaired, a reduced dosing schedule based on creatinine clearance must be used.
In pharmacokinetic studies of eight elderly patients (greater than 65 years) with normal renal function and six healthy volunteers (aged 25 to 28 years), mean (± 1 sd) Total Body Clearance (1.8 (0.1) L/h vs. 1.8 (0.3) L/h) and mean Volume of Distribution (10.4 (1.2) L vs. 10.3 (1.6) L) were similar following administration of a one gram intravenous bolus dose.
Therapeutic levels of cefotetan are achieved in many body tissues and fluids including:
- skin
- ureter
- muscle
- bladder
- fat
- maxillary sinus mucosa
- myometrium
- tonsil
- endometrium
- bile
- cervix
- peritoneal fluid
- ovary
- umbilical cord serum
- kidney
- amniotic fluid
Nonclinical Toxicology
Carcinogenesis and Mutagenesis
Although long-term studies in animals have not been performed to evaluate carcinogenic potential, no mutagenic potential of cefotetan was found in standard laboratory tests.
Clinical Studies
There is limited information regarding Cefotetan disodium Clinical Studies in the drug label.
How Supplied
Cefotetan sodium 10 grams for injection
Storage
Store at 20° to 25°C (68° to 77°F)
Images
Drug Images
{{#ask: Page Name::Cefotetan disodium |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Cefotetan disodium |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Diarrhea is a common problem caused by antibiotics which usually ends when the antibiotic is discontinued. Sometimes after starting treatment with antibiotics, patients can develop watery and bloody stools (with or without stomach cramps and fever) even as late as two or more months after having taken the last dose of the antibiotic. If this occurs, patients should contact their physician as soon as possible.
Patients should be counseled that antibacterial drugs, including cefotetan, should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When cefotetan is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by cefotetan or other antibacterial drugs in the future.
As with some other cephalosporins, a disulfiram-like reaction characterized by flushing, sweating, headache, and tachycardia may occur when alcohol (beer, wine, etc.) is ingested within 72 hours after cefotetan administration. Patients should be cautioned about the ingestion of alcoholic beverages following the administration of cefotetan.
Precautions with Alcohol
Alcohol-Cefotetan disodium interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- Cefotan [1]
Look-Alike Drug Names
There is limited information regarding Cefotetan disodium Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Label Page=Cefotetan disodium |Label Name=Cefotetan disodium 10 mg.png
}}