Benznidazole
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Yashasvi Aryaputra[2], Anmol Pitliya, M.B.B.S. M.D.[3]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Benznidazole is a nitroimidazole antimicrobial that is FDA approved for the treatment of Chagas disease (American trypanosomiasis), caused by Trypanosoma cruzi. Common adverse reactions include abdominal pain, rash, decreased weight, headache, nausea, vomiting, neutropenia, urticaria, pruritis, eosinophilia, decreased appetite.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
There is limited information regarding Benznidazole FDA-Labeled Indications and Dosage (Adult) in the drug label.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding benznidazole Off-Label Guideline-Supported Use and Dosage (Adult) in the drug label.
Non–Guideline-Supported Use
There is limited information regarding benznidazole Off-Label Non-Guideline-Supported Use and Dosage (Adult) in the drug label.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Indications:
- Benznidazole tablets are indicated in pediatric patients 2 to 12 years of age for the treatment of Chagas disease (American trypanosomiasis) caused by Trypanosoma cruzi.
- This indication is approved under accelerated approval based on the number of treated patients who became Immunoglobulin G (IgG) antibody negative against the recombinant antigens of T. cruzi. Continued approval for this indication may be contingent upon verification and description of clinical benefit in confirmatory trials.
Recommended Dosage in Pediatric Patients (2 to 12 Years of Age):
- The total daily dose for pediatric patients 2 to 12 years of age is 5 mg/kg to 8 mg/kg orally administered in two divided doses separated by approximately 12 hours, for a duration of 60 days (see TABLE 1).

Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding benznidazole Off-Label Guideline-Supported Use and Dosage (Pediatric) in the drug label.
Non–Guideline-Supported Use
There is limited information regarding benznidazole Off-Label Non-Guideline-Supported Use and Dosage (Pediatric) in the drug label.
Contraindications
Hypersensitivity
- Benznidazole tablets are contraindicated in patients with a history of hypersensitivity reaction to benznidazole or other nitroimidazole derivatives. Reactions have included severe skin and soft tissue reactions.
Disulfiram
- Benznidazole tablets are contraindicated in patients who have taken disulfiram within the last two weeks. Psychotic reactions may occur in patients who are using benznidazole and disulfiram concurrently.
Alcohol and Products Containing Propylene Glycol
- Consumption of alcoholic beverages or products containing propylene glycol is contraindicated in patients during and for at least 3 days after therapy with Benznidazole tablets. A disulfiram-like reaction (abdominal cramps, nausea, vomiting, headaches, and flushing) may occur due to the interaction between alcohol or propylene glycol and benznidazole.
Warnings
Potential for Genotoxicity and Carcinogenicity
Genotoxicity
- Genotoxicity of benznidazole has been demonstrated in humans, in vitro in several bacterial species and mammalian cell systems, and in vivo in rodents.
- A study evaluating the cytogenetic effect of benznidazole in pediatric patients ranging from 11 months to 11 years of age (the safety and effectiveness of benznidazole tablets in patients less than 2 years old has not been established) with Chagas disease demonstrated a two-fold increase in chromosomal aberrations. In pediatric patients with Chagas disease who were treated with benznidazole, the median incidence of micronucleated interphase lymphocytes in 20 patients increased 2 fold compared to pre-dose values. In the same study, the mean incidence of chromosomal aberrations in 10 patients also increased 2 fold compared to pre-dose values.
Carcinogenicity
- Carcinogenicity has been observed in mice and rats treated chronically with nitroimidazole agents which are structurally similar to benznidazole. Similar data have not been reported for benznidazole. It is not known whether benznidazole is associated with carcinogenicity in humans.
Embryo-Fetal Toxicity
- Based on findings from animal studies, benznidazole tablets can cause fetal harm when administered to a pregnant woman. In animal reproduction studies, benznidazole administered orally to pregnant rats and rabbits during organogenesis was associated with fetal malformations at doses approximately 1-3 times the maximum recommended human dose (MRHD) in rats (anasarca, anophthalmia, and/or microphthalmia) and doses approximately 0.3-1 times the MRHD in rabbits (ventricular septal defect). In rats, reduced maternal weights and smaller litter sizes occurred at a dose approximately 3 times the MRHD. In rabbits, reduced maternal weight gain, and abortions in 2/20 females occurred at a dose approximately equal to the MHRD. Advise pregnant women of the potential risk to a fetus. Pregnancy testing is recommended for females of reproductive potential. Advise females of reproductive potential to use effective contraception during treatment with benznidazole tablets and for 5 days after the last dose.
Hypersensitivity Skin Reactions
- Serious skin and subcutaneous disorders including acute generalized exanthematous pustulosis (AGEP), toxic epidermal necrolysis (TEN), erythema multiforme, and eosinophilic drug reaction have been reported with benznidazole. Discontinue treatment at the first evidence of these serious cutaneous reactions.
- Extensive skin reactions, such as rash (maculopapular, pruritic macules, eczema, pustules, erythematous, generalized, and allergic dermatitis, exfoliative dermatitis) have also been reported. Most cases occurred after approximately 10 days of treatment with benznidazole. Most rashes resolved with treatment discontinuation.
- In case of skin reactions presenting with additional symptoms or signs of systemic involvement such as lymphadenopathy, fever and/or purpura, discontinuation of treatment is recommended.
Central and Peripheral Nervous System Effects
- Treatment with benznidazole tablets can cause paresthesia or symptoms of peripheral neuropathy that may take several months to resolve. Headache and dizziness have been reported. In cases where neurological symptoms occur, immediate discontinuation of treatment is recommended. In most cases, symptoms occur late in the course of treatment.
Hematological Manifestations of Bone Marrow Depression
- There have been reports of hematological manifestations of bone marrow depression, such as neutropenia, thrombocytopenia, anemia and leukopenia, which resolved after treatment discontinuation. Patients with hematological manifestations of bone marrow depression must take benznidazole tablets only under strict medical supervision. Monitor complete blood count. Total and differential leukocyte counts are recommended before, during and after therapy.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- Benznidazole was evaluated in two randomized, double-blind, placebo-controlled trials (Trial 11 and Trial 22) and one uncontrolled trial (Trial 33).
- Trial 1 was conducted in pediatric patients 6 to 12 years of age with chronic indeterminate Chagas disease in Argentina. The chronic indeterminate form includes patients with serologic evidence of T. cruzi infection without symptoms of cardiac or gastrointestinal disease. A total of 106 patients were randomized to receive either benznidazole (5 mg/kg/day twice daily for 60 days; N= 55) or placebo (N=51) and followed for 4 years.
- Trial 2 was conducted in pediatric patients 7 to 12 years of age with chronic indeterminate Chagas disease in Brazil. A total of 129 patients were randomized to receive either benznidazole (7.5 mg/kg/day twice daily for 60 days; N = 64) or placebo (N = 65) and followed for 3 years.
- Trial 3 was an uncontrolled study in pediatric patients 2 to 12 years of age with chronic indeterminate Chagas disease. A total of 37 pediatric patients with Chagas disease were enrolled in this safety and pharmacokinetics study. Patients were treated with benznidazole 5 to 8 mg/kg/day twice daily for 60 days.
Adverse Reactions Leading to Discontinuation
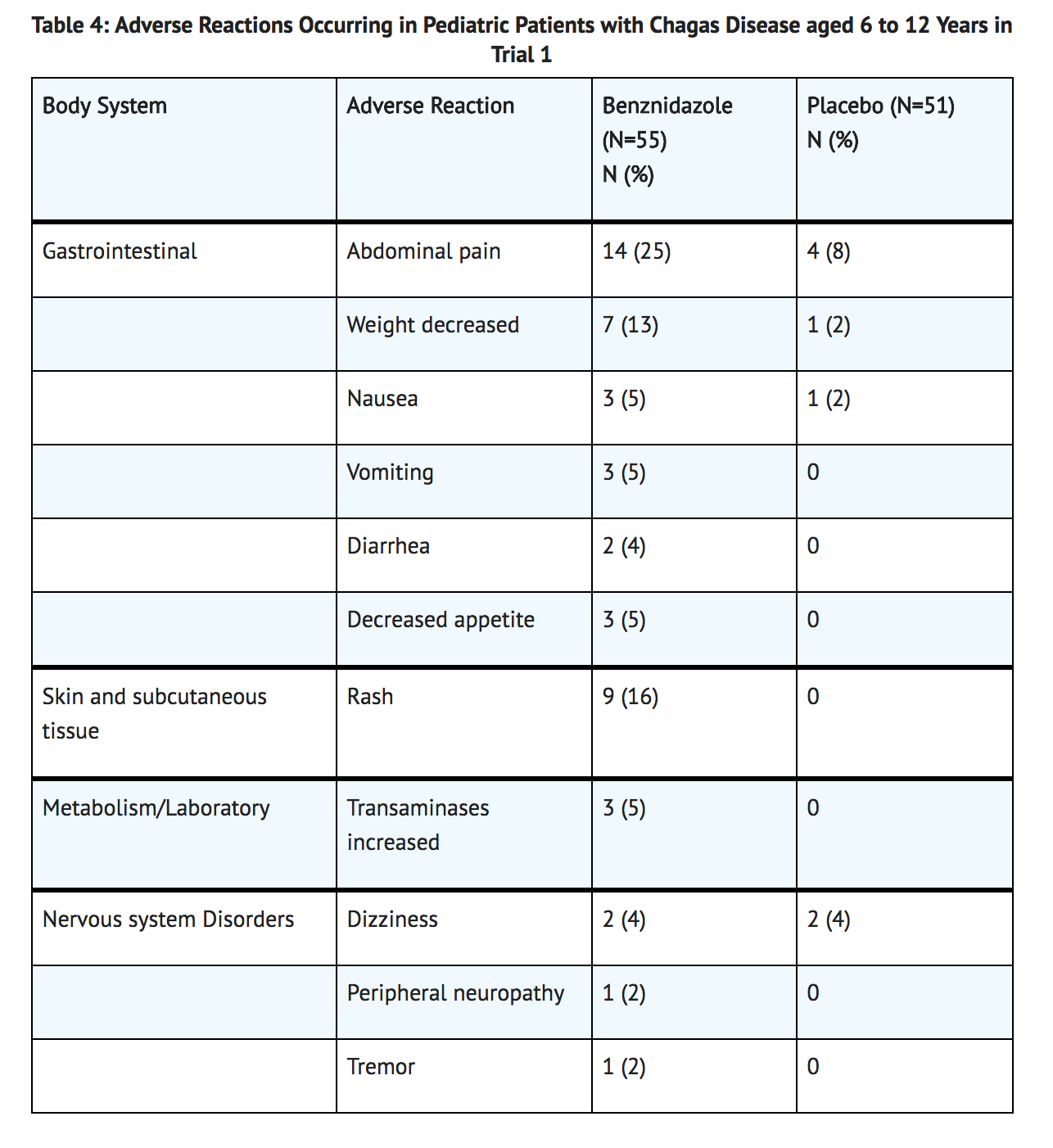
- In Trial 1, benznidazole was discontinued due to an adverse reaction in 5/55 (9%) patients. Some patients had more than one adverse reaction resulting in treatment discontinuation. The adverse reactions included abdominal pain, nausea, vomiting, rash, decreased appetite, headache, and transaminases increased.
Common Adverse Reactions in Pediatric Patients
- The most frequently reported adverse reactions in pediatric patients treated with benznidazole in Trial 1 were abdominal pain (25%), rash (16%), decreased weight (13%), and headache (7%). TABLE 4 lists adverse reactions occurring at a rate of 1% or greater in pediatric patients with Chagas disease aged 6 to 12 years of age in Trial 1.
- In Trial 2, skin lesions were reported in 7 of 64 (11%) pediatric patients treated with benznidazole and in 2 of 65 patients receiving placebo. Adverse reactions reported in fewer than 5% of benznidazole-treated patients included nausea, anorexia, headache, abdominal pain and arthralgia.
- In a subset of 19 pediatric patients 2 to 6 years of age treated with benznidazole in Trial 3, 6 patients (32%) had the following adverse reactions: rash, leukopenia, urticaria, eosinophilia, decreased appetite, and neutropenia. These adverse reactions were similar to those observed in the overall population of 37 patients.
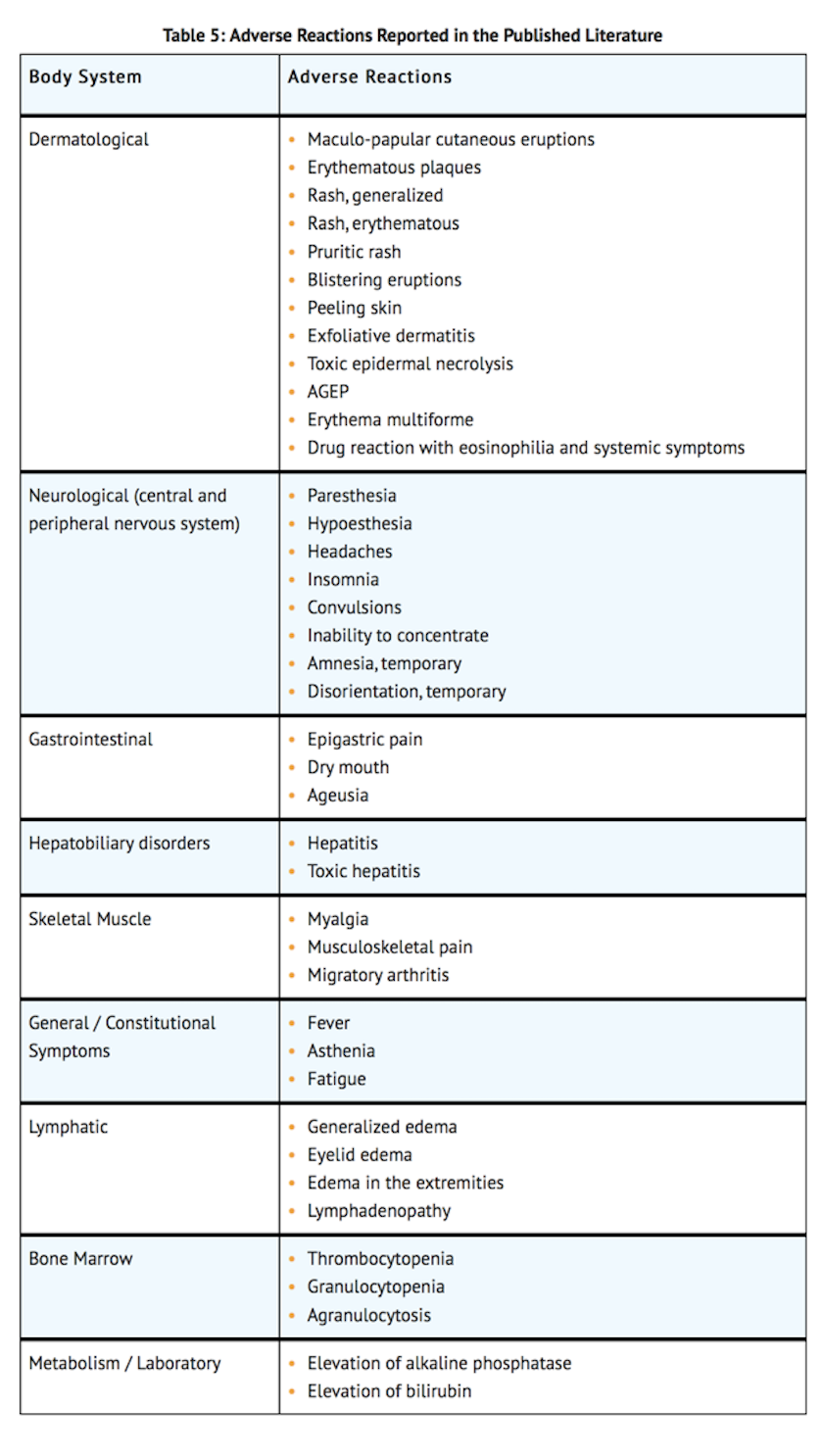
Postmarketing Experience
- The following adverse reactions have been identified during the use of other formulations of benznidazole outside of the United States. Because these reactions are reported from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Drug Interactions
- Disulfiram
- Alcohol and Products Containing Propylene Glycol
Disulfiram
- Psychotic reactions have been reported in patients who are concurrently taking disulfiram and nitroimidazole agents (structurally related to benznidazole, but not with benznidazole). Benznidazole tablets should not be given to patients who have taken disulfiram within the last two weeks.
Alcohol and Products Containing Propylene Glycol
- Abdominal cramps, nausea, vomiting, headaches, and flushing may occur if alcoholic beverages or products containing propylene glycol are consumed during or following therapy with nitroimidazole agents which are structurally related to benznidazole. Although no similar reactions have been reported with benznidazole, discontinue alcoholic beverage or products containing propylene glycol during and for at least 3 days after therapy with benznidazole tablets.
Use in Specific Populations
Pregnancy
Risk Summary
- Based on findings from animal studies, benznidazole tablets may cause fetal harm when administered to a pregnant woman. Published postmarketing reports on benznidazole use during pregnancy are insufficient to inform a drug-associated risk of adverse pregnancy-related outcomes. There are risks to the fetus associated with Chagas Disease. In animal reproduction studies, benznidazole administered orally to pregnant rats and rabbits during organogenesis was associated with fetal malformations at doses approximately 1-3 times the MRHD in rats (anasarca, anophthalmia, and/or microphthalmia) and doses approximately 0.3-1.0 times the MRHD in rabbits (ventricular septal defect). Advise pregnant women of the potential risk to a fetus.
- The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Clinical Considerations
- Disease-associated Maternal and/or Embryo/Fetal Risk
- Published data from case-control and observational studies on chronic Chagas disease during pregnancy are inconsistent in their findings. Some studies showed an increased risk of pregnancy loss, prematurity and neonatal mortality in pregnant women who have chronic Chagas disease while other studies did not demonstrate these findings. Chronic Chagas disease is usually not life-threatening. Since pregnancy findings are inconsistent, treatment of chronic Chagas disease during pregnancy is not recommended due to risk of embryo-fetal toxicity from benznidazole tablets.
- Acute symptomatic Chagas disease is rare in pregnant women; however, symptoms may be serious or life-threatening. There have been reports of pregnant women with life-threatening symptoms associated with acute Chagas disease who were treated with benznidazole. If a pregnant women presents with acute symptomatic Chagas disease, the risks versus benefits of treatment with benznidazole tablets to the mother and the fetus should be evaluated on a case-by-case basis.
Data (Animal Data)
- In an embryo-fetal toxicity study in pregnant rats, an oral dose of benznidazole of 150 mg/kg/day during organogenesis (days 6-17 of gestation) was associated with maternal weight loss, reduced fetal weights, and smaller litter sizes. Benznidazole was also associated with a low incidence of fetal malformations including anasarca in one fetus at a dose of 50 mg/kg/day and anasarca and eye abnormalities (anophthalmia and microphthalmia) in 5 fetuses in 5 litters at a high dose of 150 mg/kg/day (approximately equivalent to 1 and 3 times, respectively, the MRHD based on whole body surface area comparisons). The No Observed Adverse Effect Level (NOAEL) dose for maternal toxicity in this study, 50 mg/kg/day, is approximately equal to the MRHD based on body surface area comparisons. The NOAEL dose for fetal toxicity was 15 mg/kg/day which is approximately equivalent to 0.3 times the MRHD based on whole body surface area comparisons.
- In an embryo-fetal study in pregnant rabbits, oral (gavage) administration of benznidazole during organogenesis (days 6 to 19 of gestation) at a high dose of 25 mg/kg/day was associated with maternal toxicity including reduced weight gain and food consumption and abortions in 2/20 females. Benznidazole was also associated with a low incidence of fetal abnormalities including ventricular septal defect in 2 fetuses in 2 litters at a dose of 7.5 mg/kg/day and in 1 fetus at a dose of 25 mg/kg/day (approximately equivalent to 0.3 and 1 times respectively the MRHD based on whole body surface area comparisons). The NOAEL values for maternal and fetal toxicity in this study were 7.5 and 2.5 mg/kg/day respectively, which are respectively equivalent to approximately 0.3 and 0.1 times the MRHD based on body surface area comparisons.
- In a pre- postnatal study in rats, first generation (F1) pups born to dams administered 15, 50, and 75 mg/kg/day benznidazole demonstrated normal pre-weaning behavior, physical and functional development, neurological findings, and reproductive parameters. However, cesarean section data for the pregnant first generation (F1) females in the high-dose group included significantly higher pre-implantation loss and significantly lower mean values for corpora lutea counts, number of implantations, and number of live embryos. Also small testes and/or epididymides were observed in 1/20 and 2/20 first generation males in the mid- and high-dose groups respectively, and two of the affected animals failed to mate or induce pregnancy. However, the mean values for mating performance, fertility index, testes weight, testes and epididymides sperm counts, and epididymal sperm motility and progression were not altered in any of the F1 males in benznidazole treatment groups. The number of live second generation (F2) fetuses born to F1 dams was reduced in the high-dose group. The NOAEL value was considered to be 50 mg/kg/day which is approximately equal to the MRHD based on body surface area comparisons.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Benznidazole in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Benznidazole during labor and delivery.
Nursing Mothers
Limited published literature based on breast milk sampling reports that benznidazole is present in human milk at infant doses of 5.5 to 17% of the maternal weight-adjusted dosage and a milk/plasma ratio ranging between 0.3-2.79. There are no reports of adverse effects on the breastfed infant and no information on the effects of benznidazole on milk production. Because of the potential for serious adverse reactions, and transmission of Chagas disease, advise patients that breastfeeding is not recommended during treatment with benznidazole tablets.
Pediatric Use
The safety and effectiveness of benznidazole tablets have been established in pediatric patients 2 to 12 years of age for the treatment of Chagas disease. Use in pediatric patients 2 to 12 years of age was established in two adequate and well-controlled trials in pediatric patients 6 to 12 years old with additional safety and pharmacokinetic data from pediatric patients 2 to 6 years of age.
Safety and effectiveness in pediatric patients below the age of 2 years and above the age of 12 years have not been established.
Geriatic Use
There is no FDA guidance on the use of Benznidazole in geriatric settings.
Gender
There is no FDA guidance on the use of Benznidazole with respect to specific gender populations.
Race
There is no FDA guidance on the use of Benznidazole with respect to specific racial populations.
Renal Impairment
Use of benznidazole tablets has not been evaluated in patients with renal impairment.
Hepatic Impairment
Use of benznidazole tablets has not been evaluated in patients with hepatic impairment.
Females of Reproductive Potential and Males
Pregnancy Testing
- Pregnancy testing is recommended for females of reproductive potential.
Contraception (Females)
- Benznidazole tablets can cause fetal harm when administered to a pregnant woman. Advise females of reproductive potential to use effective contraception during treatment with benznidazole tablets and for 5 days after the final dose.
Infertility (Males)
- Based on findings in rodents, benznidazole tablets may impair fertility in males of reproductive potential. It is not known whether effects on fertility are reversible.
Immunocompromised Patients
There is no FDA guidance one the use of Benznidazole in patients who are immunocompromised.
Administration and Monitoring
Administration
Assessment Prior to Initiating Benznidazole Tablets
- Obtain a pregnancy test in females of reproductive potential prior to therapy with Benznidzole tablets.
Preparation of Slurry as an Alternative Method of Administration
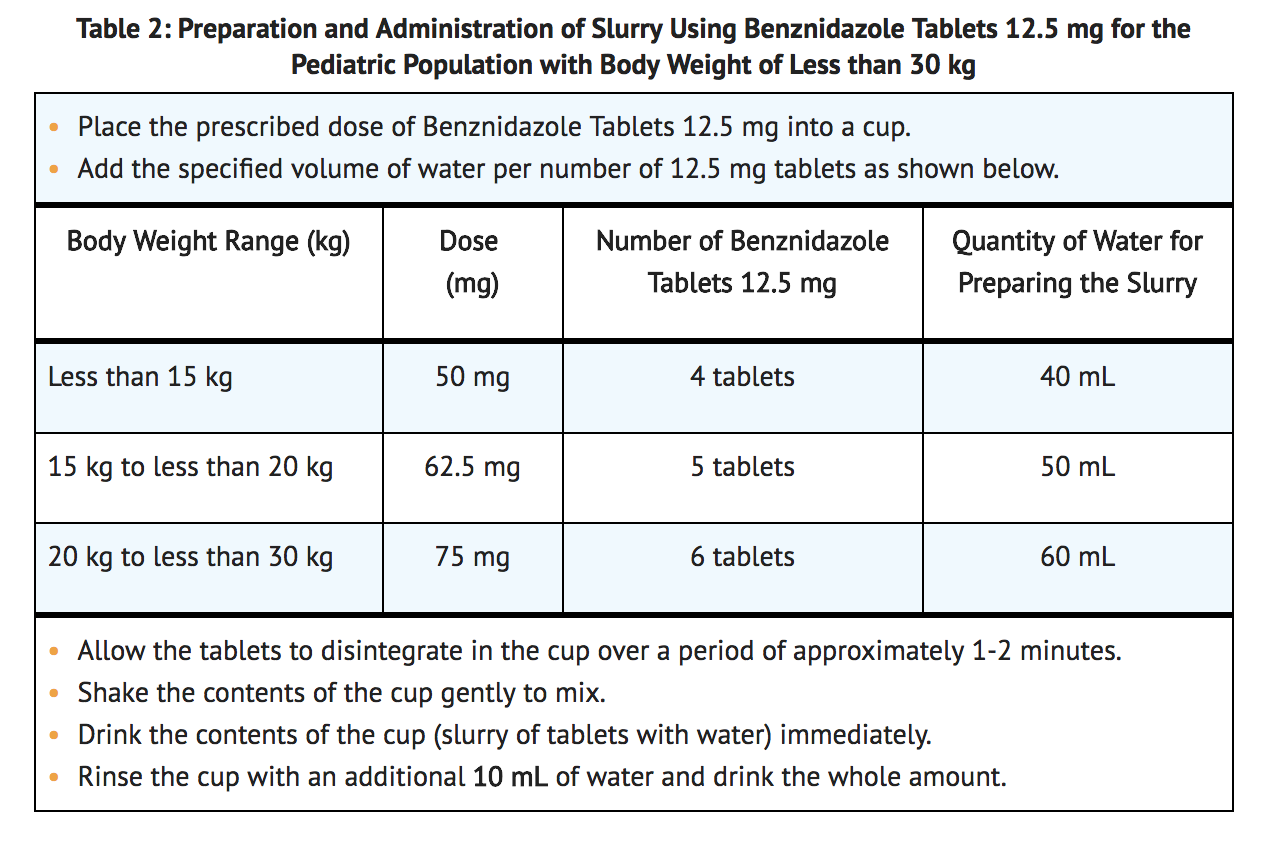
- Preparation of Slurry Using benznidazole tablets 12.5 mg for the Pediatric Population with Body Weight Less Than 30 kg.
- Benznidazole tablets 12.5 mg may be made into slurry in a specified volume of water for the pediatric population with body weight less than 30 kg (see TABLE 2). The 12.5 mg tablet slurry is prepared by the following method:
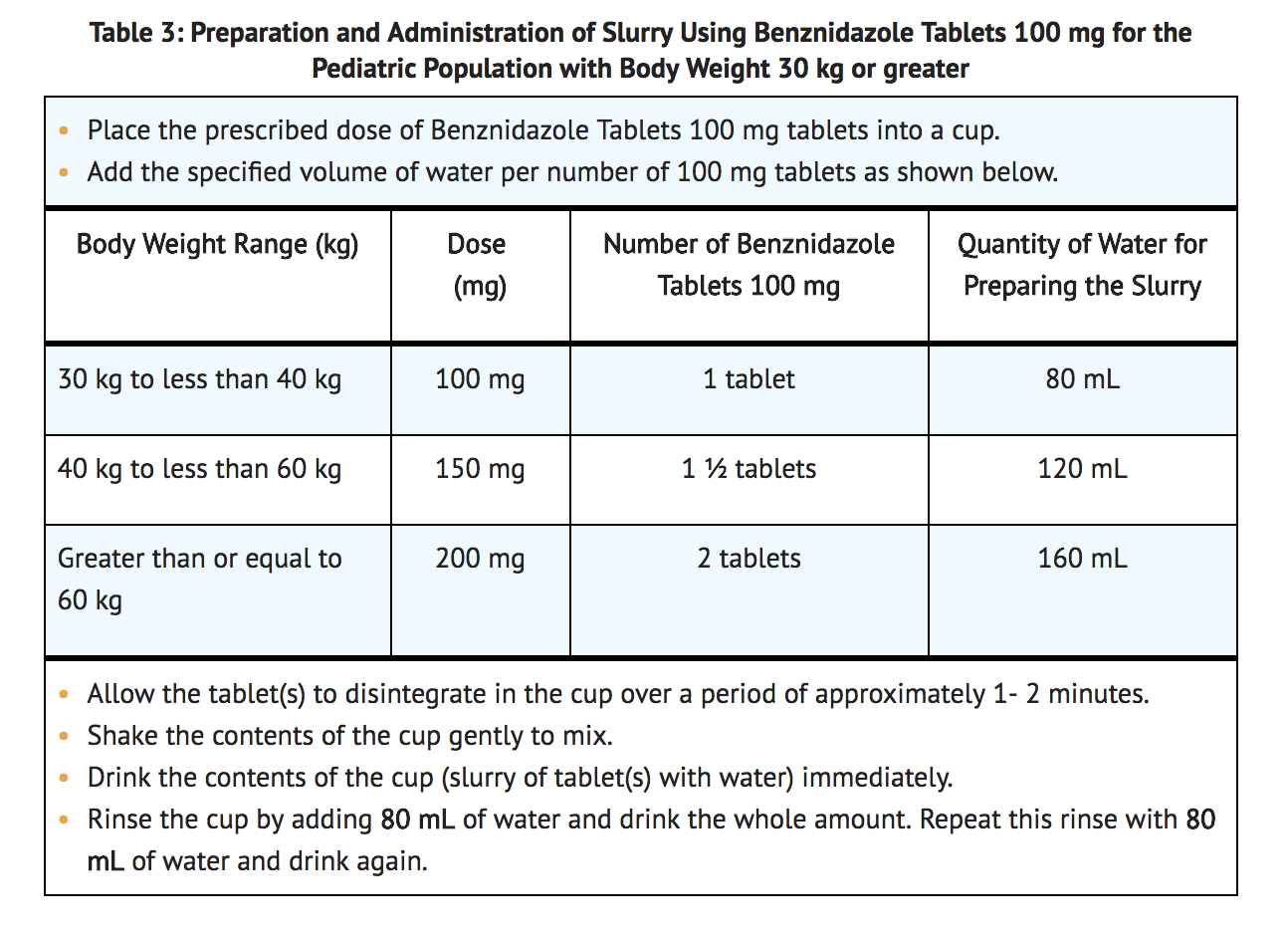
- Preparation of Slurry Using benznidazole tablets 100 mg for the Pediatric Population with Body Weight (30 kg or greater).
- Benznidazole tablets 100 mg may be made into a slurry in a specified volume of water for the pediatric population with body weight of 30 kg or greater (see TABLE 3). The 100 mg tablet slurry is prepared as follows:
Monitoring
- Improvement of symptoms of Chagas disease, caused by Trypanosoma cruzi, may be indicative of efficacy.
- Complete blood count with differential: Before, during, and after therapy.
- Pregnancy Test: Prior to therapy initiation, in females of reproductive potential.
IV Compatibility
There is limited information regarding the compatibility of Benznidazole and IV administrations.
Overdosage
There is limited information regarding Benznidazole overdosage. If you suspect drug poisoning or overdose, please contact the National Poison Help hotline (1-800-222-1222) immediately.
Pharmacology
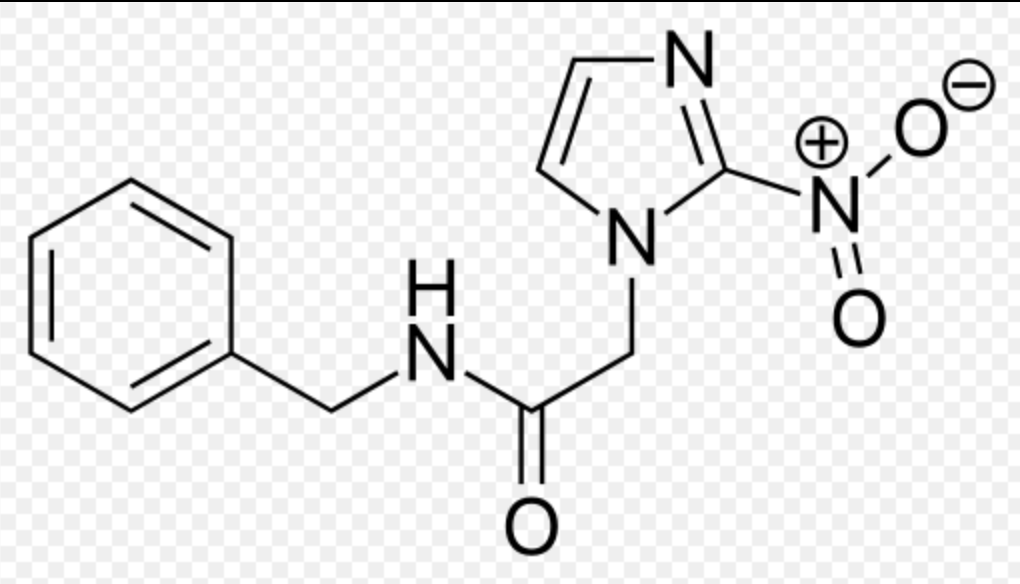
| |
Benznidazole
| |
| Systematic (IUPAC) name | |
| N-benzyl-2-(2-nitro-1H-imidazol-1-yl)acetamide | |
| Identifiers | |
| CAS number | |
| ATC code | P01 |
| PubChem | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 260.249 g/mol |
| SMILES | & |
| Physical data | |
| Melt. point | 188.5–190 °C (371–374 °F) |
| Pharmacokinetic data | |
| Bioavailability | High |
| Metabolism | Liver |
| Half life | 12 hours |
| Excretion | Kidney and fecal |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | by mouth |
Mechanism of Action
- Benznidazole is a nitroimidazole antimicrobial drug.
Structure
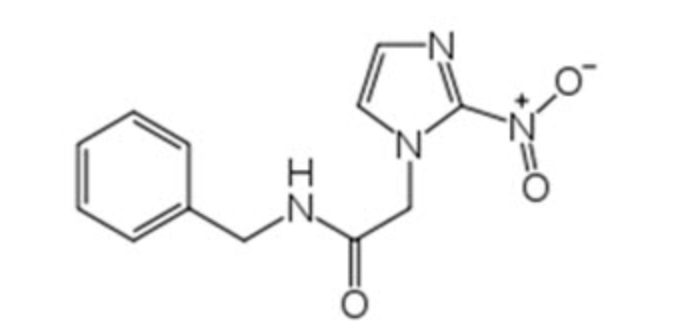
Pharmacodynamics
- The pharmacodynamics of benznidazole is unknown.
Pharmacokinetics
Absorption
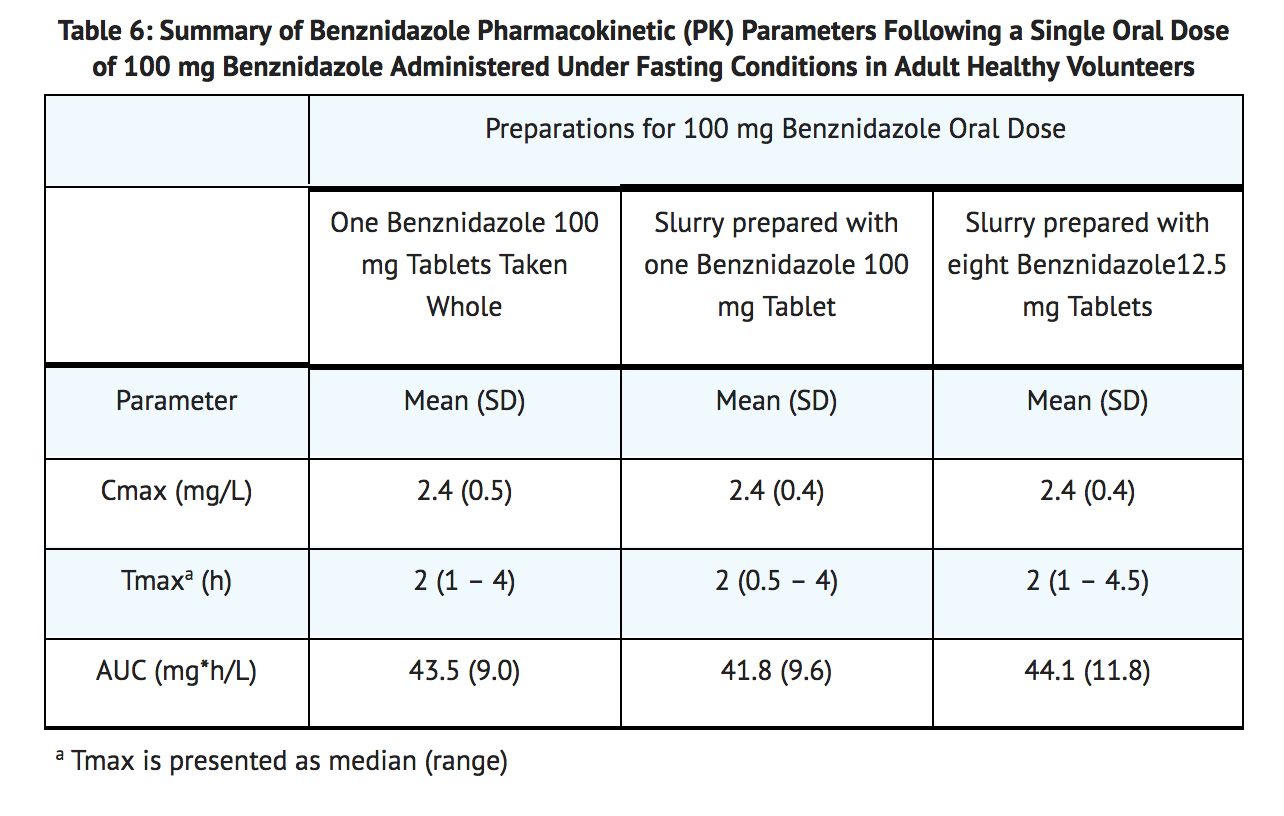
- The absorption of benznidazole from three different 100 mg benznidazole preparations was comparable when administered as a single dose under fasting conditions in adult healthy volunteers (TABLE 6).
Effect of Food
- Benznidazole Cmax and AUC were not affected by the administration of benznidazole 100 mg tablet with a high-fat, high-caloric meal (approximately 1034 total kcal, 67 kcal from fat, 42 kcal from carbohydrates, 59 kcal from protein) compared with fasted conditions in adult healthy volunteers. Serum concentrations of benznidazole reached peak levels at 3.2 hours (1-10 hours) after administration of benznidazole tablets 100 mg tablet after a high-fat, high-caloric meal, and at 2.0 hours (0.5-4 hours) in fasted conditions.
Distribution
- Protein binding is reported to be approximately 44 to 60 %.
Elimination
- The elimination half-life on benznidazole is approximately 13 hours in healthy volunteers following single dose.
Metabolism
- Benznidazole metabolism pathway is unknown.
Excretion
- Benznidazole and unknown metabolites are reported to be excreted in the urine and feces.
Specific Populations
- The effect of sex, race, renal impairment, or hepatic impairment on the pharmacokinetics of benznidazole is unknown.
Drug Interaction Studies
- In vitro studies showed that benznidazole is a P-gp substrate and does not notably induce Cytochrome P450 enzymes 1A2, 2B6, and 3A4 at concentrations up to 100 uM.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenicity
- Long-term carcinogenicity studies for benznidazole have not been performed.
- Nitroimidazoles, which have similar chemical structures to benznidazole have been reported to be carcinogenic in mice and rats.
Genetic Toxicity
- Genotoxicity of benznidazole has been demonstrated in vitro in several bacterial species and mammalian cell systems and in vivo in mammals.
- Benznidazole was mutagenic in several strains of S. typhimurium (TA 100, 102 1535, 1537, 1538, 97, 98 99 53 and UTH8414), E.coli, and K. pneumoniae.
- Benznidazole was genotoxic in several in vitro mammalian cell assays including a chromosome aberration assay in human lymphocytes and in sister chromatid exchange assays in human lymphocytes and in Human Hep G2 cells.
- In vivo, benznidazole was shown to be positive for genotoxicity in a mouse bone marrow micronucleus assay, in mouse and human red blood cell micronucleus assays, in a mouse abnormal sperm head assay and in a human peripheral blood lymphocyte assay. However in other micronucleus studies in mice and rats, oral doses of benznidazole did not cause a significant increase in the frequency of chromosomal aberrations in bone marrow cells or micronuclei in peripheral blood cells.
Impairment of Fertility
- In a 6-month, chronic repeated-dosing study with Wistar rats, benznidazole was shown to produce dose-dependent testicular and epididymal atrophy at a dose of 30 mg/kg/day (approximately equivalent to 0.6 times the MRHD based on whole body surface area comparisons). Aspermia was also evident in affected rats, but fertility was not assessed in this study. The NOAEL value in this study was considered to be 10 mg/kg/day (5 mg/kg twice daily) in males which is approximately 0.2-times the MRHD based on body surface area comparison. In other literature reports, benznidazole has been shown to cause testicular atrophy and inhibit spermatogenesis in pubertal and adult rats and mice5-7.
- In a female fertility study, oral (gavage) administration of benznidazole to female Wistar rats twice daily for a 2-week pre-mating period, during mating, and through day 7 of gestation was associated with transient lower body weight gain and food consumption. There was no benznidazole-related effect on mating performance or fertility and no adverse macroscopic or reproductive organ weight changes. However, benznidazole reproductive performance was associated with a higher post-implantation loss with lower live litter size at a dose of 150 mg/kg/day (equivalent to approximately 3 times the MRHD based on whole body surface area comparisons). The NOAEL value for this study was consider to be 50 mg/kg/day which is approximately equivalent to the MRHD based on whole body surface area comparison.
Animal Toxicology and/or Pharmacology
- Single oral dose toxicity studies in rats have established that benznidazole causes ultrastructural changes in the adrenal cortex, colon, esophagus, ovaries, and testis 5, 8-11. In these tissues, these changes were associated with the presences of nitro reductase activity, the production of reactive metabolites, and or covalent binding of metabolites.
- Neurotoxicity including brain axonal degeneration and Purkinje cell degeneration was observed with repeated-oral dosing in dogs without adverse changes in peripheral nerves12-14. Neurological signs included: apathy, hypertonia, hyperreflexia, ataxia, loss of balance, oscillatory movements of the trunk and head, strong contractions of the back and leg muscles, opisthotonus and nystagmus. Neurotoxicity was not observed in other test species, including mouse, rat, guinea pig, and rabbit.
Clinical Studies
- The safety and effectiveness of benznidazole for the treatment of Chagas disease in patients 6 to 12 years of age was established in two adequate and well-controlled trials (Trial 1 and Trial 2) as described below.
- Trial 1 was a randomized, double-blind, placebo-controlled trial in children 6 to 12 years of age with chronic indeterminate Chagas disease conducted in Argentina. The chronic indeterminate form of Chagas disease includes patients with serologic evidence of T. cruzi infection without symptoms of cardiac or gastrointestinal disease. A total of 106 patients were randomized to receive either benznidazole (5 mg/kg/day for 60 days) or placebo and followed for 4 years. Patients with at least two positive conventional serologic tests for antibodies to T. cruzi were included in the study. The conventional serologic tests used include indirect hemagglutination assay (IHA), immunofluorescence antibody assay (IFA), and/or enzyme linked immunosorbent assay (ELISA) and were based on the detection of antibodies against T. cruzi parasites.
- Trial 2 was a randomized, double-blind, placebo-controlled trial in pediatric patients 7 to 12 years of age with chronic indeterminate Chagas disease conducted in Brazil. A total of 129 patients were randomized to receive either benznidazole (7.5 mg/kg/day for 60 days) or placebo and followed for 3 years. Patients with three positive conventional serologic tests for antibodies to T. cruzi were included in the study. The conventional serologic tests include IHA, IFA, and/or ELISA and were based on the detection of antibodies against T. cruzi parasites.
- Both trials measured antibodies by conventional and nonconventional assays. The nonconventional assays include F29-ELISA and AT- chemiluminescence-ELISA that are based on detection of anti-T. cruzi IgG antibodies against the recombinant antigens, F29 and AT from the flagella of T. cruzi parasites. Benznidazole treatment resulted in a significantly higher percentage of seronegative patients by a nonconventional assay. Results at the end of follow-up are reported in the following table.

- In Trial 1 using conventional ELISA, 4 of 53 (7.5%) benznidazole subjects and 2 of 50 (4.0%) placebo subjects seroconverted to negative by the end of follow-up (difference 3.5, 95% CI (-7.0, 14.9)). In Trial 2 using conventional ELISA, 4 of 64 (6.3%) of benznidazole subjects and 0 of 65 placebo subjects seroconverted to negative by the end of follow-up (difference 6.3, 95% CI (0.3, 15.2)).
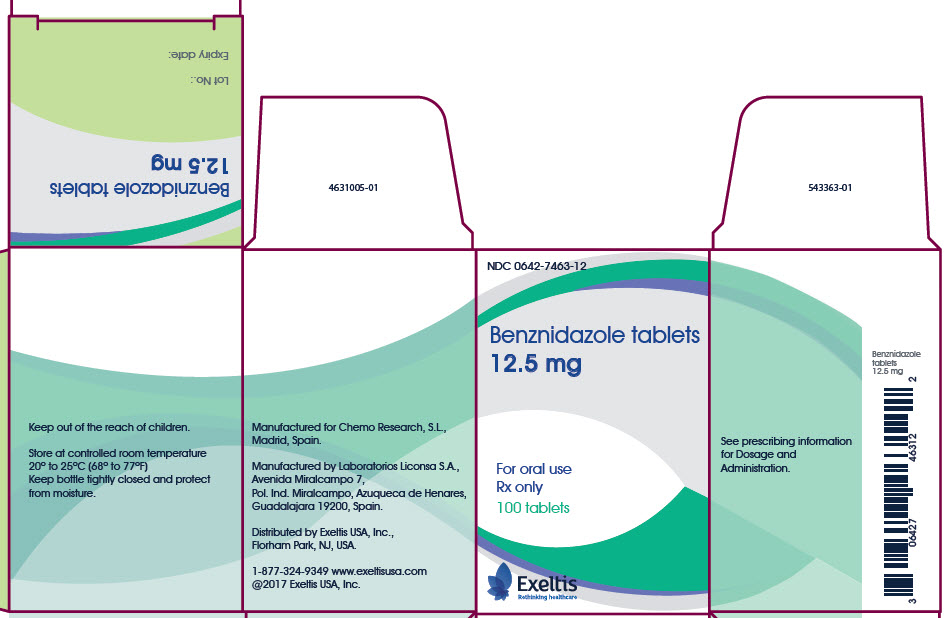
How Supplied
- Benznidazole tablets (12.5 mg or 100 mg) are supplied as follows:
- 100 mg white tablets, round and functionally scored twice as a cross on both sides. Each tablet is about 10 mm in diameter debossed with “E” on one side of each quarter portion.
- 12.5 mg white tablets, round and unscored. Each tablet is about 5 mm in diameter debossed with “E” on one side.
- Benznidazole tablets 100 mg are available in bottles of 100 tablets (NDC 0642-7464-10).
- Benznidazole tablets 12.5 mg are available in bottles of 100 tablets (NDC 0642-7463-12).
Storage
- Store at controlled room temperature 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F). Keep bottle tightly closed and protect from moisture.
Images
Drug Images
{{#ask: Page Name::Benznidazole |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Benznidazole |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Embryo-Fetal Toxicity
- Advise pregnant women and females of reproductive potential that exposure to benznidazole tablets during pregnancy can result in fetal harm.
- Advise females to inform their healthcare provider of a known or suspected pregnancy.
- Advise females of reproductive potential to use effective contraception while taking benznidazole tablets and for 5 days after the last dose.
Lactation
- Advise women not to breastfeed during treatment with benznidazole tablets.
Infertility
- Advise males of reproductive potential that benznidazole tablets may impair fertility.
Important Administration Instructions
- Advise patients and parents/caregivers of pediatric patients taking Benznidazole tablets that:
- Benznidazole tablets 100 mg are functionally scored tablets which can be split into one-half (50 mg) or one-quarter (25 mg) at the scored lines to provide doses less than 100 mg.
- Benznidazole tablets 12.5 mg and 100 mg (whole or split) can be made into a slurry in a specified volume of water for the pediatric population.
Hypersensitivity Skin Reactions
- Advise patients that serious skin reactions can occur with benznidazole tablets. In case of skin reactions, presenting with additional symptoms of systemic involvement such as lymphadenopathy, fever and/or purpura, discontinuation of treatment is necessary.
Central and Peripheral Nervous System Effects
- Advise patients that treatment can potentially cause paresthesia or symptoms of peripheral neuropathy. In cases where neurological symptoms occur, immediate discontinuation of treatment is recommended.
Hematological Manifestations of Bone Marrow Depression
- Advise patients that there have been hematological manifestations of bone marrow depression, such as anemia and leukopenia, which are reversible, and normalized after treatment discontinuation.
Interaction with Alcohol
- Advise patients to discontinue consumption of alcoholic beverages or products containing propylene glycol while taking benznidazole tablets and for at least three days afterward because abdominal cramps, nausea, vomiting, headaches, and flushing may occur.
Precautions with Alcohol
Alcohol-Benznidazole interaction has not been established. Talk to your doctor regarding the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Benznidazole Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Benznidazole Look-Alike Drug Names in the drug label.
Drug Shortage
Price
References
The contents of this FDA label are provided by the National Library of Medicine.