Azithromycin detailed information
 | |
| Clinical data | |
|---|---|
| Pregnancy category | |
| Routes of administration | oral (capsule) |
| ATC code | |
| Pharmacokinetic data | |
| Bioavailability | 38% for 250 mg capsules |
| Metabolism | hepatic |
| Elimination half-life | 68 hours |
| Excretion | biliary, renal (4.5%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
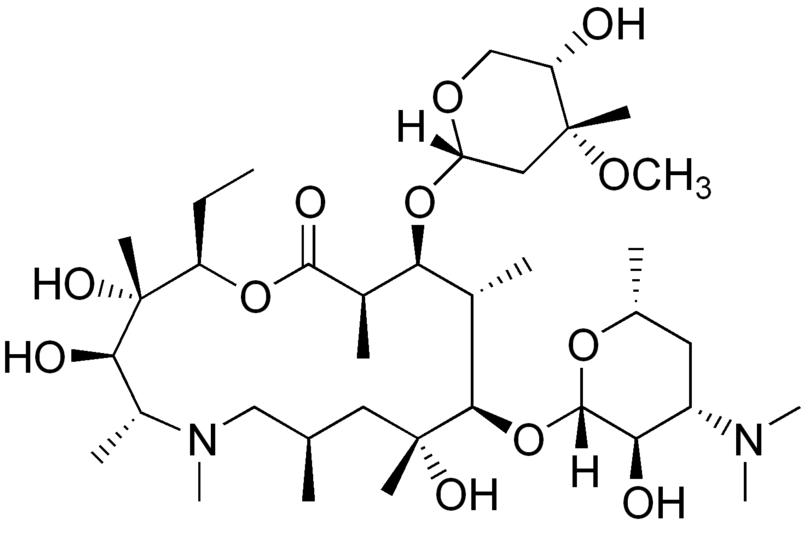
| Formula | C38H72N2O12 |
| Molar mass | 748.88 |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Azithromycin is an azalide, a subclass of macrolide antibiotics. Azithromycin (brand names Zithromax® in Italy, United States and Portugal; Vinzam® / Zitromax® in Spain; Zmax®; Sumamed®; Aztrin®, Zitrocin®, Azibiot®, Azifine®) is one of the world's best-selling antibiotics, and is derived from erythromycin; however, it differs chemically from erythromycin in that a methyl-substituted nitrogen atom is incorporated into the lactone ring, thus making the lactone ring 15-membered. Azithromycin is used to treat certain bacterial infections, most often bacteria causing middle ear infections, tonsillitis, throat infections, laryngitis, bronchitis, pneumonia and sinusitis. It is also effective against certain sexually transmitted infectious diseases, such as non-gonococcal urethritis and cervicitis. Recent studies have also indicated it to be effective against late-onset asthma, but these findings are controversial and not widely accepted as of yet.
Etymology
Azithromycin's name is derived from the azane-substituent and erythromycin. Its accurate chemical name is
(2R,3S,4R,5R,8R,10R,11R,12S,13S,14R)-13- [(2,6-dideoxy-3-C-methyl-3-O-methyl -α-L-ribo-hexopyranosyl)oxy]-2-ethyl- 3,4,10-trihydroxy-3,5,6,8,10,12,14-heptamethyl -11-[[3,4,6-trideoxy-3-(dimethylamino) -β-D-xylo-hexopyranosyl]]oxy]-1-oxa- 6-azacyclopentadecan-15-one.
History
A team of Pliva's researchers, Gabrijela Kobrehel, Gorjana Radobolja-Lazarevski and Zrinka Tamburašev led by Dr. Slobodan Đokić, discovered azithromycin in 1980. It was patented in 1981, and was later found by Pfizer's scientists while going through patent documents. In 1986 Pliva and Pfizer signed a licensing agreement which gave Pfizer exclusive rights for the sale of azithromycin in Western Europe and the United States. Pliva brought their azithromycin on the market in Central and Eastern Europe under the brand name of Sumamed in 1988, Pfizer Zithromax in 1991, and Zentiva Azitrox.
Available forms
Azithromycin is commonly administered in tablet or oral suspension (a one-dose version was made available in 2005). It is also available for intravenous injection. Tablets come in 250 mg and 500 mg doses. Oral suspension comes in 100 mg/teaspoon and 200 mg/teaspoon strengths. The 250 mg tablets are often dispensed in packages of six and commonly referred to as a "Z-Pak," whereas the 500 mg tablets are commonly available commercially in a pack of three tablets, or "Tri-Pak," intended as a three-day treatment. A common dose of oral azithromycin therapy consists of a "double dose" of medication on the first day of treatment and subsequent treatment for four or five additional days. With the "Z-Pak," this means two 250 mg tablets (a total of 500 mg) on the first day and one 250 mg tablet once daily for the next four days.
Pfizer brand-name (e.g., Zithromax®) azithromycin tablets are mottled pink, unscored, film-coated, modified-oval-shaped tablets containing azithromycin monohydrate equivalent to 250 mg or 500 mg azithromycin and the following inactive ingredients: butylated hydroxytoluene, calcium phosphate, carmine, colloidal silicon dioxide, FD&C red # 40 lake, FD&C yellow # 6 lake, hypromellose (2910, 15cP), lactose monohydrate, magnesium stearate, pregelatinized starch, sodium lauryl sulfate, talc, titanium dioxide and triacetin. The 250 mg tablets have the following appearance: They are mottled pink, of a modified-oval-shape, and have "Pfizer" engraved into one side with "306" engraved into the other. The 500 mg tablet is similar in appearance with the exception of its relative size and "ZTM500" engraved onto one side with "Pfizer" on the reverse. As with all medications, generic azithromycin tablets produced by companies other than Pfizer without the Zithromax brand name may have different appearances.
Brand name Zithromax in Greece and Cyprus is sold in boxes containing a blister of six white 250mg capsules. On the capsules there is printed the "Pfizer" logo and the symbols "ZTM 250".
Azithromycin is now available with microcencapsulated in betacyclodextrin, in India with the brand name of VRENTROX-DS and VENTROX-250.
Microbiology
Azithromycin prevents bacteria from growing by interfering with their protein synthesis. Azithromycin binds to the 50S subunit of the bacterial ribosome, and thus inhibits translation of mRNA. Nucleic acid synthesis is not affected. Azithromycin has similar antimicrobial spectrum as erythromycin, but is more effective against certain gram-negative bacteria, particularly Haemophilus influenzae.
Azithromycin has been proven to be most effective against the most isolates of the following microorganisms:
- Staphylococcus aureus
- Streptococcus agalactiae
- Streptococcus pneumoniae
- Streptococcus pyogenes
- Haemophilus ducreyi
- Haemophilus influenzae
- Moraxella catarrhalis
- Neisseria gonorrhoeae
- Chlamydia pneumoniae
- Chlamydia trachomatis
- Mycoplasma pneumoniae
Azithromycin has been shown to be effective against malaria when used in combination with artesunate or quinine; the optimal dose for this is not yet known.[1]
Pharmacokinetics
Unlike erythromycin, azithromycin is acid-stable and can therefore be taken orally with no need of protection from gastric acids. It is readily absorbed, and diffused into most tissues and phagocytes. Due to the high concentration in phagocytes, azithromycin is actively transported to the site of infection. During active phagocytosis, large concentrations of azithromycin are released. The concentration of azithromycin in the tissues can be over 50 times higher than in plasma. This is due to ion trapping and the high lipid solubility.
Metabolism
Following a single 500 mg dose, plasma concentrations of azithromycin declined in a polyphasic pattern with a mean apparent plasma clearance of 630 mL/min and a terminal elimination half-life of 68 hours. The prolonged terminal half-life is thought to be due to extensive uptake and subsequent release of drug from tissues.
Biliary excretion of azithromycin, predominantly unchanged, is a major route of elimination. Over the course of a week, approximately 6% of the administered dose appears as unchanged drug in urine.
Side effects
Most common side effects are gastrointestinal; diarrhea (4-5%), nausea (3%), abdominal pain (2-3%) and vomiting. Fewer than 1% of patients stop taking the drug due to side effects. Serious allergic reactions, dermatologic reactions, and fatalities have been reported but are extremely rare. As with all antimicrobial agents, pseudomembranous colitis can occur during and up to several weeks after azithromycin therapy. This drug may interfere with the effectiveness of birth control pills; other forms of contraception may be required during the treatment period. Although mild diarrhea is normal, if it is moderate to severe, or if it continues or is bothersome, check with your doctor. If you experience vomiting, or hearing loss or ringing in the ears, contact your doctor as soon as possible.
Allergic reaction
Allergic reaction to this medicine is unlikely, but seek immediate medical attention if it occurs. Patients who suffer from an allergic reaction to Azithromycin can experience blood in the stool 4-10 days after ingestion, although cases of this have been recorded as early as after the first day of ingestion. These allergies are usually non-severe if the treatment is immediately stopped. A severe reaction includes a severe rash, hives, breathing difficulties, or dizziness. If experiencing difficulty breathing; tightness of chest; swelling of eyelids, face, legs, hands, eyes, throat, tongue or lips; or you develop a rash or hives, tell your doctor immediately. If you notice other effects not listed above, contact your doctor, nurse, or pharmacist.
Reference links
- ↑ Noedl H, Krudsood S, Chalermratana K; et al. (2006). "Azithromycin combination therapy with artesunate or quinine for the treatment of uncomplicated Plasmodium falciparum malaria in adults: a randomised, phase 2 clinical trial in Thailand". 43 (10): 1264&ndash, 71. PMID 17051490. Unknown parameter
|journel=ignored (help)
Additional Resources
de:Azithromycin hu:Azitromicin nl:Azithromycine fi:Atsitromysiini th:อะซิโทรมัยซิน Template:Jb1
- Pages with script errors
- Pages with citations using unsupported parameters
- CS1 maint: Explicit use of et al.
- CS1 maint: Multiple names: authors list
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs with no legal status
- Articles containing unverified chemical infoboxes
- Macrolide antibiotics
- Antimalarial agents