Amiodarone pulmonary toxicity
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Hardik Patel, M.D.; Sapan Patel M.B.B.S
Synonyms and keywords: Amiodarone lung toxicity; APT
Overview
Amiodarone is an antiarrhythmic drug which is commonly used to treat ventricular and supraventricular arrhythmias, in particular following pulmonary and cardiac surgery, due to a higher incidence of postoperative atrial fibrillation in these patients. It is an iodine-containing compound that tends to accumulate in several organs, including the lungs. It has been associated with a variety of adverse effects. Most individuals administered amiodarone on a chronic basis will experience at least one side effect. The most serious adverse effect is amiodarone-induced pulmonary toxicity. It occurs mostly in patients receiving large doses of the drug over prolonged periods.
Pathophysiology
The pathogenesis of amiodarone-induced pulmonary toxicity is incompletely understood. There are 2 major hypotheses:
- Cytotoxicity- a direct toxic injury to lung cells: Amiodarone may induce the production of toxic O2 radicals, which can directly damage cells. It also appears to promote the accumulation of phospholipids in lung tissue with direct effect on alveolar-capillary membrane.[1]
- Hypersensitivity- an indirect immunologic reaction which is supported by the finding of cytotoxic T cells in bronchoalveolar lavage (BAL) fluid from patients with diagnosed toxicity.[2][1][3]
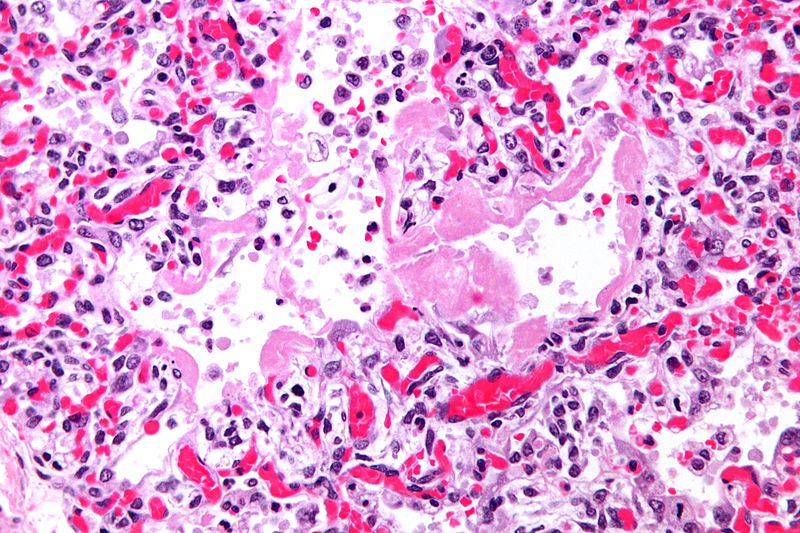
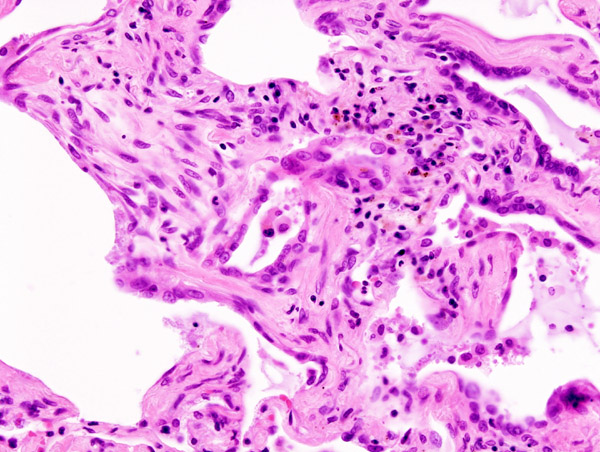
Microscopic Pathology
-
Micrograph of hyaline membranes, as seen in diffuse alveolar damage (DAD), the histologic correlate of adult respiratory distress syndrome (ARDS).
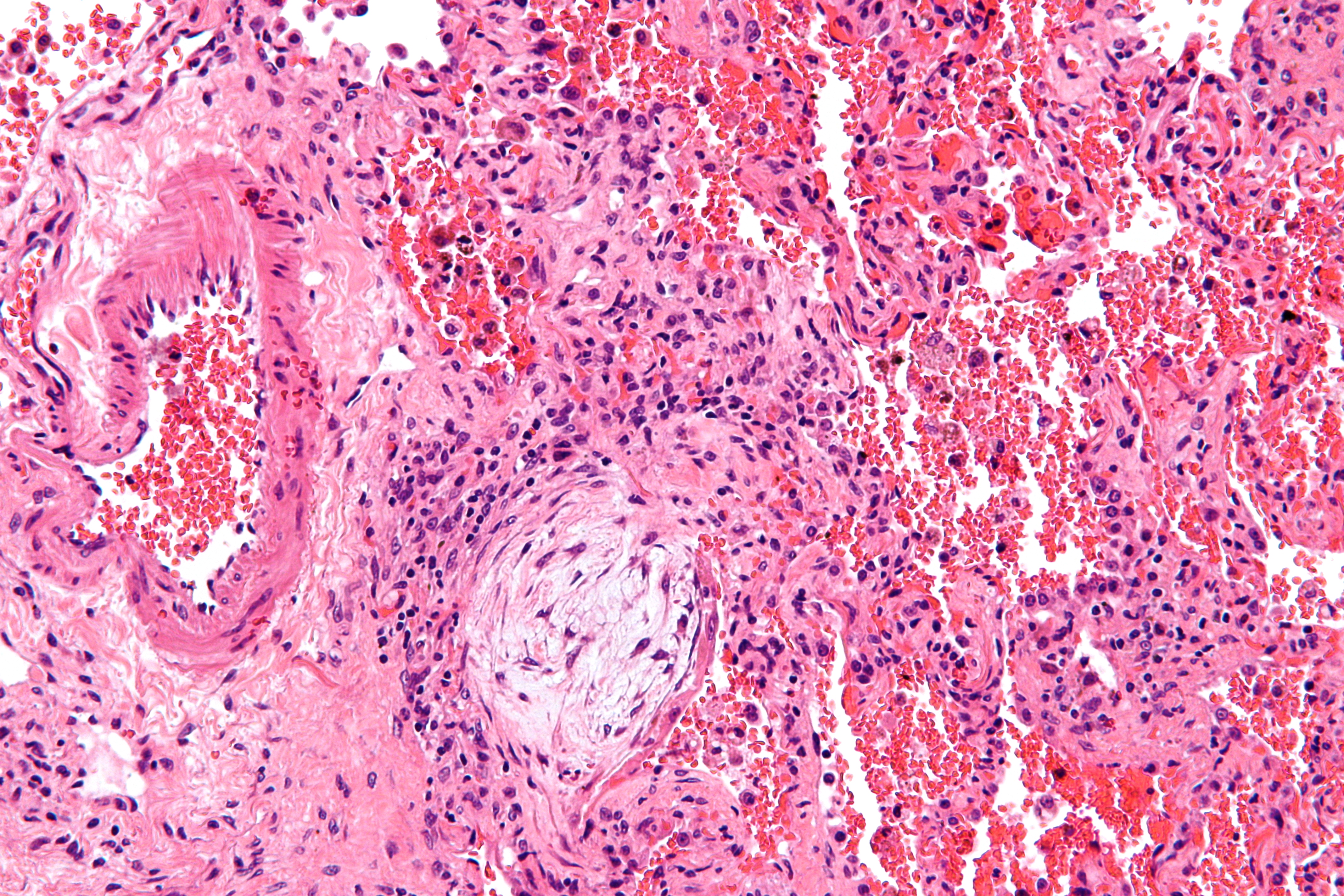
-
Histopathological image of pulmonary fibrosis representing chronic interstitial pneumonitis.
Copyleft image obtained courtesy of http://en.wikipedia.org/wiki/File:Usual_interstitial_pneumonia_%281%29.JPG, http://en.wikipedia.org/wiki/File:Hyaline_membranes_-_very_high_mag.jpg, http://en.wikipedia.org/wiki/File:Masson_body_-_high_mag.jpg.
Differentiating Amiodarone Pulmonary Toxicity from other Diseases
The diagnosis of amiodarone-induced pulmonary toxicity is one of exclusion. Conditions to be ruled out are following:
- Heart failure
- Infectious pneumonia
- Pulmonary embolism
- Malignancy
Epidemiology and Demographics
Pulmonary toxicity is the most serious adverse effect of amiodarone. It may occur in up to 5–10% of treated patients.[2]
Risk Factors
Anyone taking amiodarone is at risk. Patients on higher doses and one who have been taking the drug for a long period of time appear to have a higher risk, and some evidence suggests that individuals with underlying lung disease are also more likely to develop toxicity with amiodarone.
- High cumulative dose (more than 400 mg/d)[2]
- Duration over two months
- Increased age
- Preexisting pulmonary disease
- Major surgical procedures
- Pulmonary angiography
Some individuals were noted to develop pulmonary fibrosis after a week of treatment, while others did not develop it after years of continuous use. Common practice is to avoid the drug if possible in individuals with decreased lung function.
Natural History, Complications, and Prognosis
Amiodarone pulmonary toxicity can manifest itself in several ways:
- Rare but the most dangerous form of pulmonary toxicity
- Sudden, life-threatening, and having diffuse lung involvement
- Most often following major surgical procedures, especially cardiac surgery and pulmonary angiography, but it can be seen at any time and without any obvious precipitating causes.[4][5][6]
- Surgical patients develop symptoms 1-4 days post-extubation and patients who have undergone pulmonary angiography may develop symptoms within 30 minutes of procedure which include severe dyspnea and hypoxia.[7]
- Patients usually must be placed on mechanical ventilators, and their mortality rate even with intensive therapy approaches 50%.[8]
- Chest x ray shows bilateral diffuse pulmonary infiltrates.

Diffuse Alveolar Hemorrhage
- Rare pulmonary toxicity[9][10][11]
- More acute presentation than chronic interstitial pneumonitis which may occur suddenly within few days to months after starting the drug.[12][13]
- Most often in patients with chronic lung disease
- Symptoms include:
- Approximately 25% of cases of amiodarone-induced pulmonary toxicity[14][12]
- More acute presentation than chronic interstitial pneumonitis
- Symptoms mimic infectious pneumonitis and include:
- Fever
- Nonproductive cough
- Pleuritic chest pain
- Dyspnea
- Common physical exam findings are crackles and pleural rubs.
- Chest x ray and CT scan show patchy alveolar opacities

Copyleft image obtained courtesy of http://en.wikipedia.org/wiki/File:CT_BOOP.jpg
Chronic Interstitial Pneumonitis
- Most common presentation of amiodarone-induced pulmonary toxicity
- Insidious and gradual onset over weeks to months in patients on therapy >2 months
- Characterized by the slowly progressing dyspnea, cough, weight loss, and easy fatigue. These symptoms are easy to mistaken for heart disease or the effects of aging, as many patients taking amiodarone have a history of heart disease.
- Chest x ray shows focal or diffuse interstitial opacities
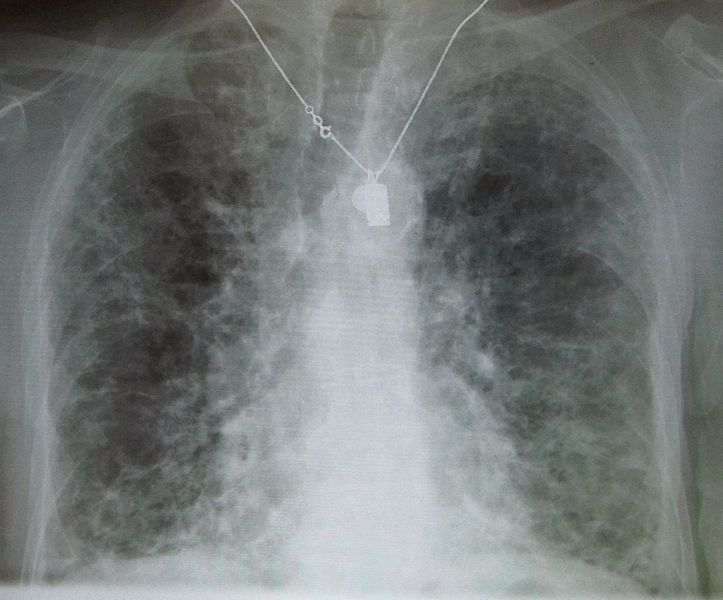
Copyleft image obtained courtesy of http://en.wikipedia.org/wiki/File:IPF_amiodarone.JPG; James Heilman, MD.
Solitary Lung Mass
- This is a rare form of amiodarone-induced pulmonary toxicity which may simulate lung cancer.[15][16][17]
Prognosis
Prognosis is usually favorable in about 75% of the patients after withdrawal of the drug with or without treatment.[18] The mortality rate of amiodarone pulmonary toxicity is 10% of the reported cases but may have higher mortality rate in patients with ARDS.[8]
Diagnosis
Clinical Presentation
Typically, patients who have been on amiodarone for months or even several years develop progressive dyspnea, nonproductive cough, malaise, fever, and occasionally pleuritic chest pain.[19][2][20] However, clinicians evaluating patients with possible amiodarone-induced pulmonary toxicity must obtain a thorough drug exposure history, maintain a high index of suspicion, and use a systematic diagnostic approach to make the correct and firm diagnosis. Symptoms can persist or worsen despite drug withdrawal as elimination half-life averages 40-55 days. Any new pulmonary symptom in patient taking amiodarone should raise the suspicion of toxicity. Physical examination may be unremarkable in milder cases, but in more severely affected individuals, following findings may be noted:
- Bilateral inspiratory crackles
- Pleural rubs
- Hypoxemia
- Respiratory distress
These findings frequently must be distinguished from heart failure. Unexplained pulmonary conditions for which no other likely cause can be identified should be judged as probable amiodarone lung toxicity.
Laboratory Findings
- Chest imaging like chest x-ray and CT scan
- Pulmonary function testing
- DLCO: DLCO is often decreased with toxicity, but it is not useful as a predictive index as most patients with >20% decrease in DLCO while on amiodarone do not develop pulmonary toxicity with continued therapy.[21][22]
- Bronchoalveolar lavage
- Lung biopsy
Diagnostic Criteria
The diagnosis of amiodarone-induced pulmonary toxicity is one of exclusion. Once other diagnoses are excluded, then the clinical diagnosis is suggested when ≥ 3 of the following are present:[2]
- Decrease in TLC of ≥ 15% or in DLCO of ≥ 20%
- New abnormal finding on chest imaging
- New or worsening signs or symptoms
- Phospholipidosis in lung cells
- CD8+ lymphocytosis on BAL
- Lung biopsy with interstitial pneumonitis, BOOP, ARDS, or pulmonary fibrosis.
- Improvement in symptoms with drug discontinuation.
Treatment
- Stop the drug as soon as possible.
- Use the alternative medications and procedures to treat supraventricular arrhythmias and the implantable cardioverter-defibrillator for potentially life-threatening ventricular arrhythmias.
- Pulse systemic glucocorticoid therapy (eg, prednisone 40 to 60 mg/d) with gradual tapering of the dose.[23]
- Polymyxin B-immobilized fiber column direct hemoperfusion.[24]
Prevention and Monitoring
Prevention
Currently there are no proven measures to prevent toxicity. The only strategy that appears to be efficacious is that of using the smallest dose possible for any particular patient.
Monitoring
Patients who are to begin amiodarone therapy should be informed about potential adverse effects and told to report any new respiratory symptoms promptly. They should have an initial chest x-ray and pulmonary function test, including a DLCO for the baseline reference points.
Current guidelines for monitoring are:
- Annual chest x-ray should be obtained as long as patients remain on amiodarone treatment.[25]
- An immediate chest x-ray and pulmonary function testing should be performed if there is clinical suspicion of pulmonary toxicity.[26]
- There are recommendations for monitoring for extrapulmonary toxicity. These include thyroid function testing, monitoring liver enzymes and function, as well as protective measures for skin photosensitivity.[25][26][27][28]
References
- ↑ 1.0 1.1 Jessurun GA, Crijns HJ (1997). "Amiodarone pulmonary toxicity". BMJ. 314 (7081): 619–20. PMC 2126104. PMID 9066469.
- ↑ 2.0 2.1 2.2 2.3 2.4 Martin WJ, Rosenow EC (1988). "Amiodarone pulmonary toxicity. Recognition and pathogenesis (Part I)". Chest. 93 (5): 1067–75. PMID 3282816.
- ↑ Martin WJ, Rosenow EC (1988). "Amiodarone pulmonary toxicity. Recognition and pathogenesis (Part 2)". Chest. 93 (6): 1242–8. PMID 3286141.
- ↑ Greenspon AJ, Kidwell GA, Hurley W, Mannion J (1991). "Amiodarone-related postoperative adult respiratory distress syndrome". Circulation. 84 (5 Suppl): III407–15. PMID 1934438.
- ↑ Kay GN, Epstein AE, Kirklin JK, Diethelm AG, Graybar G, Plumb VJ (1988). "Fatal postoperative amiodarone pulmonary toxicity". Am J Cardiol. 62 (7): 490–2. PMID 3414530.
- ↑ Van Mieghem W, Coolen L, Malysse I, Lacquet LM, Deneffe GJ, Demedts MG (1994). "Amiodarone and the development of ARDS after lung surgery". Chest. 105 (6): 1642–5. PMID 8205854.
- ↑ Wood DL, Osborn MJ, Rooke J, Holmes DR (1985). "Amiodarone pulmonary toxicity: report of two cases associated with rapidly progressive fatal adult respiratory distress syndrome after pulmonary angiography". Mayo Clin Proc. 60 (9): 601–3. PMID 4021550.
- ↑ 8.0 8.1 Myers JL, Kennedy JI, Plumb VJ (1987). "Amiodarone lung: pathologic findings in clinically toxic patients". Hum Pathol. 18 (4): 349–54. PMID 3557438.
- ↑ Iskandar SB, Abi-Saleh B, Keith RL, Byrd RP, Roy TM (2006). "Amiodarone-induced alveolar hemorrhage". South Med J. 99 (4): 383–7. PMID 16634249.
- ↑ Vizioli LD, Cho S (1994). "Amiodarone-associated hemoptysis". Chest. 105 (1): 305–6. PMID 8275760.
- ↑ Borders CW, Bennett S, Mount C, Claassen SL (2012). "A rare case of acute diffuse alveolar hemorrhage following initiation of amiodarone: a case report". Mil Med. 177 (1): 118–20. PMID 22338993.
- ↑ 12.0 12.1 Dean PJ, Groshart KD, Porterfield JG, Iansmith DH, Golden EB (1987). "Amiodarone-associated pulmonary toxicity. A clinical and pathologic study of eleven cases". Am J Clin Pathol. 87 (1): 7–13. PMID 3799544.
- ↑ Tanawuttiwat T, Harindhanavudhi T, Hanif S, Sahloul MZ (2010). "Amiodarone-induced alveolar haemorrhage: a rare complication of a common medication". Heart Lung Circ. 19 (7): 435–7. doi:10.1016/j.hlc.2010.01.008. PMID 20356785.
- ↑ Valle JM, Alvarez D, Antúnez J, Valdés L (1995). "Bronchiolitis obliterans organizing pneumonia secondary to amiodarone: a rare aetiology". Eur Respir J. 8 (3): 470–1. PMID 7789497.
- ↑ Piccione W, Faber LP, Rosenberg MS (1989). "Amiodarone-induced pulmonary mass". Ann Thorac Surg. 47 (6): 918–9. PMID 2757449.
- ↑ Arnon R, Raz I, Chajek-Shaul T, Berkman N, Fields S, Bar-On H (1988). "Amiodarone pulmonary toxicity presenting as a solitary lung mass". Chest. 93 (2): 425–7. PMID 3338315.
- ↑ Azzam I, Tov N, Elias N, Naschitz JE (2006). "Amiodarone toxicity presenting as pulmonary mass and peripheral neuropathy: the continuing diagnostic challenge". Postgrad Med J. 82 (963): 73–5. doi:10.1136/pgmj.2005.040105. PMC 2563731. PMID 16397086.
- ↑ Coudert B, Bailly F, Lombard JN, Andre F, Camus P (1992). "Amiodarone pneumonitis. Bronchoalveolar lavage findings in 15 patients and review of the literature". Chest. 102 (4): 1005–12. PMID 1395734.
- ↑ Camus P, Martin WJ, Rosenow EC (2004). "Amiodarone pulmonary toxicity". Clin Chest Med. 25 (1): 65–75. doi:10.1016/S0272-5231(03)00144-8. PMID 15062598.
- ↑ Ott MC, Khoor A, Leventhal JP, Paterick TE, Burger CD (2003). "Pulmonary toxicity in patients receiving low-dose amiodarone". Chest. 123 (2): 646–51. PMID 12576397.
- ↑ Mason JW (1989). "Prediction of amiodarone-induced pulmonary toxicity". Am J Med. 86 (1): 2–3. PMID 2910091.
- ↑ Gleadhill IC, Wise RA, Schonfeld SA, Scott PP, Guarnieri T, Levine JH; et al. (1989). "Serial lung function testing in patients treated with amiodarone: a prospective study". Am J Med. 86 (1): 4–10. PMID 2910095.
- ↑ Okayasu K, Takeda Y, Kojima J, Yoshizawa A, Kobayashi N, Sugiyama H; et al. (2006). "Amiodarone pulmonary toxicity: a patient with three recurrences of pulmonary toxicity and consideration of the probable risk for relapse". Intern Med. 45 (22): 1303–7. PMID 17170505.
- ↑ Sato N, Kojima K, Horio Y, Goto E, Masunaga A, Ichiyasu H; et al. (2013). "Successful treatment of severe amiodarone pulmonary toxicity with polymyxin B-immobilized fiber column direct hemoperfusion". Chest. 143 (4): 1146–50. doi:10.1378/chest.12-0994. PMID 23546489.
- ↑ 25.0 25.1 Sunderji R, Kanji Z, Gin K (2000). "Pulmonary effects of low dose amiodarone: a review of the risks and recommendations for surveillance". Can J Cardiol. 16 (11): 1435–40. PMID 11109040.
- ↑ 26.0 26.1 Goldschlager N, Epstein AE, Naccarelli GV, Olshansky B, Singh B, Collard HR; et al. (2007). "A practical guide for clinicians who treat patients with amiodarone: 2007". Heart Rhythm. 4 (9): 1250–9. doi:10.1016/j.hrthm.2007.07.020. PMID 17765636.
- ↑ Vassallo P, Trohman RG (2007). "Prescribing amiodarone: an evidence-based review of clinical indications". JAMA. 298 (11): 1312–22. doi:10.1001/jama.298.11.1312. PMID 17878423.
- ↑ Siddoway LA (2003). "Amiodarone: guidelines for use and monitoring". Am Fam Physician. 68 (11): 2189–96. PMID 14677664.