Cardiovascular Development
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
The heart is the first functional organ in a vertebrate embryo. There are 5 stages to heart development.
Specification of cardiac precursor cells
| Cardiology Network |
 Discuss Cardiovascular Development further in the WikiDoc Cardiology Network |
| Adult Congenital |
|---|
| Biomarkers |
| Cardiac Rehabilitation |
| Congestive Heart Failure |
| CT Angiography |
| Echocardiography |
| Electrophysiology |
| Cardiology General |
| Genetics |
| Health Economics |
| Hypertension |
| Interventional Cardiology |
| MRI |
| Nuclear Cardiology |
| Peripheral Arterial Disease |
| Prevention |
| Public Policy |
| Pulmonary Embolism |
| Stable Angina |
| Valvular Heart Disease |
| Vascular Medicine |
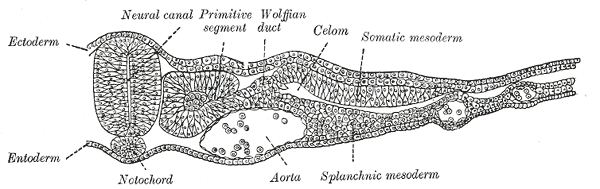
The lateral plate mesoderm delaminates to form two layers: the dorsal somatic (parietal) mesoderm and the ventral splanchnic (visceral) mesoderm. The heart precursor cells come from the two regions of the splanchnic mesoderm called the cardiogenic mesoderm. These cells can differentiate into endocardium which lines the heart chamber and valves and the myocardium which forms the musculature of the ventricles and the atria.
The heart cells are specified in anterior mesoderm by proteins such as Dickkopf-1, Nodal, and Cerberus secreted by the anterior endoderm. Whether Dickkopf-1 and Nodal act directly on the cardiac mesoderm is the subject of research, but it seems that at least they act indirectly by stimulating the production of additional factors from the anterior endoderm. These early signals are essential for heart formation such that removal of the anterior endoderm blocks heart formation. Anterior endoderm is also sufficient to stimulate heart differientation since it can induce non-cardiogenic mesoderm from more posterior positions in the embryo to form heart.
The secretion of Wnt inhibitors (such as Cerberus, Dickkopf and Crescent) by the anterior endoderm also prevents Wnt3a and Wnt8 secreted by the neural tube from inhibiting heart formation. The notochord secretes BMP antagonists (Chordin and Noggin) to prevent formation of cardiac mesoderm in inappropriate places.
Other cardiogenic signals such as BMP and FGF activate the expression of cardiac specific transcription factors such as homeodomain protein Nkx2.5. Nkx2.5 activates a number of downstream transcription factors (such as MEF2 and GATA) which activate the expression of cardiac muscle specific proteins. Mutations in Nkx2.5 result in heart development defects and congenital heart malformations.
Migration of cardiac precursor cells and fusion of the primordia
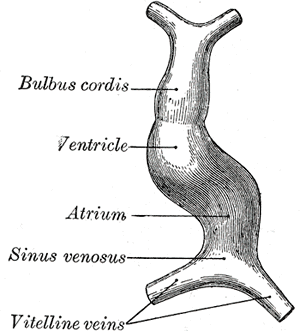
The cardiac precursor cells migrate anteriorly towards the midline and fuse into a single heart tube. Fibronectin in the extracellular matrix directs this migration. If this migration event is blocked, cardia bifida results where the two heart primordia remain separated. During fusion, the heart tube is patterned along the anterior/posterior axis for the various regions and chambers of the heart.
Heart looping
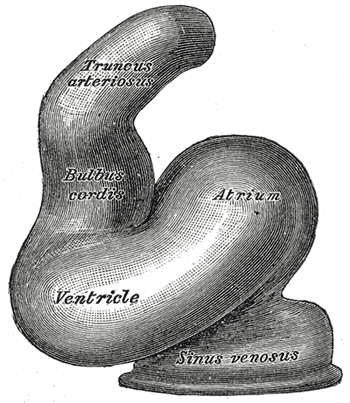
The heart tube undergoes right-ward looping to change from anterior/posterior polarity to left/right polarity. The detailed mechanism is unknown however the looping requires the asymmetrically localized transcription factor Pitx2. It is possible that the asymmetry is caused by the clockwise rotation of cilia in dispersing this transcription factor. Looping also depends on heart specific proteins activated by Nkx2.5 such as Hand1, Hand2, and Xin.
Heart chamber formation

The cell fates of the heart chambers are characterized before heart looping but can not be distinguished until after looping. Hand1 is localized to the left ventricle while Hand2 is localized to the right ventricle.
Septation and valve formation
Proper positioning and function of the valves is critical for chamber formation and proper blood flow. The endocardial cushion serves as a makeshift valve until then.
Embryofetal Heart Rates
The human heart beats more than 3.5 billion times in an average lifetime.
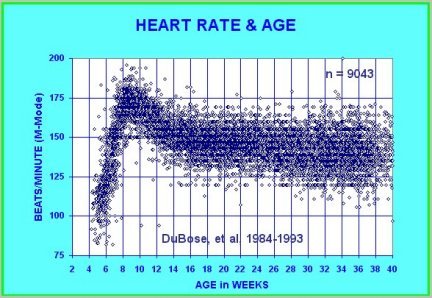
The human embryonic heart begins beating approximately 21 days after conception, or five weeks after the last normal menstrual period (LMP), which is the date normally used to date pregnancy. The human heart begins beating at a rate near the mother’s, about 75-80 beats per minute (BPM). The embryonic heart rate (EHR) then accelerates linearly for the first month of beating, peaking at 165-185 BPM during the early 7th week, (early 9th week after the LMP). This acceleration is approximately 3.3 BPM per day, or about 10 BPM every three days, an increase of 100 BPM in the first month.[1]
After peaking at about 9.2 weeks after the LMP, it decelerates to about 150 BPM (+/-25 BPM) during the 15th week after the LMP. After the 15th week the deceleration slows reaching an average rate of about 145 (+/-25 BPM) BPM at term. The regression formula which describes this acceleration before the embryo reaches 25 mm in crown-rump length or 9.2 LMP weeks is:
Age in days = EHR(0.3)+6
There is no difference in male and female heart rates before birth.[2]
References
- ↑ http://www.obgyn.net/us/us.asp?page=/us/cotm/0001/ehr2000
- ↑ Terry J. DuBose http://www.obgyn.net/english/pubs/features/dubose/ehr-age.htm Sex, Heart Rate and Age]
Template:Developmental biology Template:Development of circulatory system