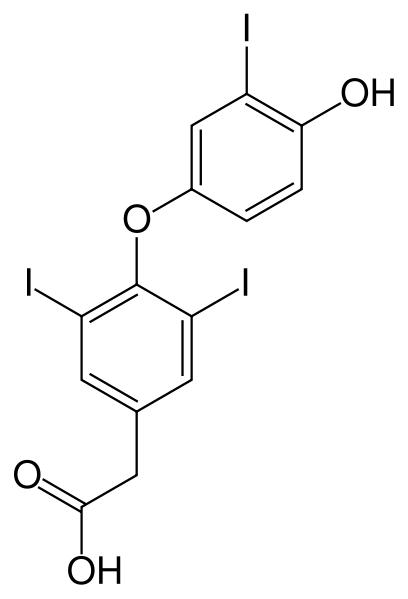
Tiratricol
 | |
 | |
| Clinical data | |
|---|---|
| Synonyms | 3,3',5-triiodothyroacetic acid TRIAC |
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | Presumed high |
| Metabolism | Hepatic glucuronidation |
| Excretion | Biliary |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C14H9I3O4 |
| Molar mass | 621.932 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
|
WikiDoc Resources for Tiratricol |
|
Articles |
|---|
|
Most recent articles on Tiratricol |
|
Media |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Tiratricol at Clinical Trials.gov Clinical Trials on Tiratricol at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Tiratricol
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Tiratricol Discussion groups on Tiratricol Patient Handouts on Tiratricol Directions to Hospitals Treating Tiratricol Risk calculators and risk factors for Tiratricol
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Tiratricol |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Tiratricol (also known as TRIAC or triiodothyroacetic acid) is a thyroid hormone analogue.
Uses
It is indicated in the management of thyroid hormone resistance syndrome[1] and is used, in combination with levothyroxine, to suppress thyroid-stimulating hormone production in patients with thyroid cancer.
It has been investigated for use in reducing goiter.[2]
It has also shown some effectiveness in reducing the atrophy caused when using corticosteroids.[3]
Tiratricol has also been widely marketed, under various trade names, as a weight loss aid. In 1999 and 2000, the United States Food and Drug Administration and Health Canada both issued warnings to the public regarding the use of dietary supplements containing tiratricol.[4][5]
Legal status
Tiratricol is not approved for sale in Canada or the United States. It was once an approved drug in Brazil, but its marketing authorization was suspended in 2003, effectively prohibiting its sale.[6] Tiratricol is still available in France for therapy of thyroid hormone resistance and adjuvant therapy of thyroid cancer.[7] It is available as an orphan drug to be prescribed by registered specialists in Europe.
References
- ↑ Carvalho GA, Ramos HE (2004). "[Thyroid hormone resistance syndrome]". Arq Bras Endocrinol Metabol (in Portuguese). 48 (1): 83–92. doi:10.1590/S0004-27302004000100010. PMID 15611821. Template:PDFlink
- ↑ Brenta G, Schnitman M, Fretes O; et al. (November 2003). "Comparative efficacy and side effects of the treatment of euthyroid goiter with levo-thyroxine or triiodothyroacetic acid". J. Clin. Endocrinol. Metab. 88 (11): 5287–92. doi:10.1210/jc.2003-030095. PMID 14602763.
- ↑ Yazdanparast P, Carlsson B, Oikarinen A, Risteli J, Lavin T, Faergemann J (November 2006). "Action of topical thyroid hormone analogue, triiodothyroacetic acid in reversing glucocorticoid-induced skin atrophy in humans". Thyroid. 16 (11): 1157–62. doi:10.1089/thy.2006.16.1157. PMID 17123343.
- ↑ "FDA Warns Against Consuming Dietary Supplements Containing Tiratricol" (Press release). U.S. Food and Drug Administration. November 21, 2000. Retrieved 2007-08-08.
- ↑ "Health Canada issues warning on products containing Tiratricol (TRIAC)" (Press release). Health Canada. December 2, 1999. Retrieved 2007-08-08.
- ↑ "Anvisa suspende Tiratricol" (in Portuguese). Brazilian Society of Endocrinology and Metabolism. Archived from the original on 2007-10-09. Retrieved 2007-08-08.
- ↑ Laboratoires DB PHARMA: Teatrois information
- Pages with script errors
- CS1 maint: Unrecognized language
- CS1 maint: Explicit use of et al.
- CS1 maint: Multiple names: authors list
- Template:drugs.com link with non-standard subpage
- Articles with changed CASNo identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Orphan drugs
- Organoiodides
- Phenols
- Phenol ethers
- Drug