Pentylenetetrazol
 | |
| Clinical data | |
|---|---|
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
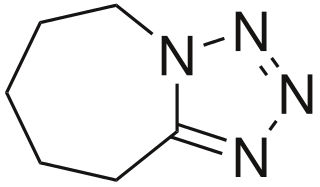
| Formula | C6H10N4 |
| Molar mass | 138.171 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
|
WikiDoc Resources for Pentylenetetrazol |
|
Articles |
|---|
|
Most recent articles on Pentylenetetrazol Most cited articles on Pentylenetetrazol |
|
Media |
|
Powerpoint slides on Pentylenetetrazol |
|
Evidence Based Medicine |
|
Cochrane Collaboration on Pentylenetetrazol |
|
Clinical Trials |
|
Ongoing Trials on Pentylenetetrazol at Clinical Trials.gov Trial results on Pentylenetetrazol Clinical Trials on Pentylenetetrazol at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Pentylenetetrazol NICE Guidance on Pentylenetetrazol
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Pentylenetetrazol Discussion groups on Pentylenetetrazol Patient Handouts on Pentylenetetrazol Directions to Hospitals Treating Pentylenetetrazol Risk calculators and risk factors for Pentylenetetrazol
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Pentylenetetrazol |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Pentylenetetrazol, also known as pentylenetetrazole, metrazol, pentetrazol (INN), pentamethylenetetrazol, Cardiazol or PTZ, was a drug used as a circulatory and respiratory stimulant. High doses cause convulsions, as discovered by the Hungarian-American neurologist and psychiatrist Ladislas J. Meduna in 1934. It has been used in convulsive therapy, but was never considered to be effective, and side-effects such as seizures were difficult to avoid. Its approval by the FDA was revoked in 1982.[1]
Mechanism
The mechanism of pentylenetetrazol is not well understood, and it may have multiple mechanisms of action. In 1984, Squires et al. published a report analyzing pentylenetetrazol and several structurally related convulsant drugs. They found that in vivo convulsant potency was strongly correlated to in vitro affinity to the picrotoxin binding site on the GABA-A receptor complex. Many GABA-A ligands are effective anticonvulsants, such as the sedatives diazepam and phenobarbital, but presumably pentylenetetrazol has the opposite effect when it binds to the GABA-A receptor.[2]
Several studies have focused on the way pentylenetetrazol influences neuronal ion channels. A 1987 study found that pentylenetetrazol increases calcium influx and sodium influx, both of which depolarize the neuron. Because these effects were antagonized by calcium channel blockers, it was concluded that pentylenetetrazol acts at calcium channels, and it causes calcium channels to lose selectivity and conduct sodium ions as well.[3]
Uses
Pentylenetetrazol has been used experimentally to study seizure phenomena and to identify pharmaceuticals that may control seizure susceptibility. Pentylenetetrazol is also a prototypical anxiogenic drug and, has been extensively utilized in animal models of anxiety. Pentylenetetrazol produces a reliable discriminative stimulus which is largely mediated by the GABAA receptor. Several classes of compounds can modulate the pentylenetetrazol discriminative stimulus including 5-HT1A, 5-HT3, NMDA, glycine, and L-type calcium channel ligands.[4]
Recently, Stanford University researchers have renewed interest in PTZ as a candidate for pharmacological treatment of Down syndrome. Published in the April 2007 issue of Nature Neuroscience, their brief communication outlined an experiment designed to test the underlying theory proposed to explain the purported efficacy of GABAA antagonists in restoring the declarative memory deficits associated with the mouse model of human Down Syndrome. Ts65Dn mice injected with a 2-week regimen of either of two compounds picrotoxin or bilobalide (both GABA antagonists) showed marked improvements in both exploration and recognition of novel objects over controls injected with only saline. These results were duplicated in a second experiment with mice fed either plain milk or a combination of milk and a non-epileptogenic dose of PTZ daily for 17 days. PTZ-fed mice achieved novel object task scores comparable to wild-type (normal) mice. These improvements persisted at least 1 to 2 months after the treatment regimen. Not surprisingly these compounds' efficacies were accompanied by the normalization of Long-term potentiation in the dentate gyrus one month after the end of treatment, further suggesting persistent drug-mediated improvements in learning and memory.[5]
The finding of pentylenetetrazol's effectiveness in treating a mouse model of Down syndrome has led to it being explored as a means of correcting other learning deficiencies. Specifically, hamsters denied their natural circadian rhythm (though not denied sleep) had their memory restored to near-normal levels when treated with pentylenetetrazol.[6]
Alternatives
In 1939, pentylenetetrazol was replaced by electroconvulsive therapy as the preferred method for inducing seizures in England's mental hospitals. However, it wasn't disapproved of in the United States until 1982.
References
- ↑ Minkel JR (February 25, 2007). "Drug May Counteract Down Syndrome". Scientific American. Retrieved 2007-03-20.
- ↑ Squires RF, Saederup E, Crawley JN, Skolnick P, Paul SM (1984). "Convulsant potencies of tetrazoles are highly correlated with actions on GABA / benzodiazepine / picrotoxin receptor complexes in brain". Life Sci. 35 (14): 1439–44. doi:10.1016/0024-3205(84)90159-0. PMID 6090836.
- ↑ Papp A, Fehér O, Erdélyi L (1987). "The ionic mechanism of the pentylenetetrazol convulsions". Acta Biol. Hung. 38 (3–4): 349–61. PMID 3503442.
- ↑ Jung ME, Lal H, Gatch MB (2002). "The discriminative stimulus effects of pentylenetetrazol as a model of anxiety: recent developments". Neurosci. Biobehav. Rev. 26 (4): 429–39. doi:10.1016/S0149-7634(02)00010-6. PMID 12204190.
- ↑ Fernandez F, Morishita W, Zuniga E, Nguyen J, Blank M, Malenka RC, Garner CC (2007). "Pharmacotherapy for cognitive impairment in a mouse model of Down syndrome" (PDF). Nat. Neurosci. 10 (4): 411–3. doi:10.1038/nn1860. PMID 17322876.
- ↑ Ruby NF, Hwang CE, Wessells C, Fernandez F, Zhang P, Sapolsky R, Heller HC (2008). "Hippocampal-dependent learning requires a functional circadian system". PNAS USA. 105 (40): 15593–8. doi:10.1073/pnas.0808259105. PMC 2563080. PMID 18832172.
- Pages with script errors
- CS1 maint: Multiple names: authors list
- Articles with changed ChemSpider identifier
- Articles with changed KEGG identifier
- Articles with changed EBI identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Articles with changed InChI identifier
- Chemical articles with unknown parameter in Infobox drug
- Chemical pages without DrugBank identifier
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Drugboxes which contain changes to watched fields
- Stimulants
- Withdrawn drugs
- Respiratory agents
- Drug