Tropicamide
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Gloria Picoy [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Tropicamide is an antimuscarinic that is FDA approved for the {{{indicationType}}} of For mydriasis and cycloplegia for diagnostic procedures. Common adverse reactions include blurred vision, burning sensation in eye.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
There is limited information regarding Tropicamide FDA-Labeled Indications and Dosage (Adult) in the drug label.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Tropicamide in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Tropicamide in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Tropicamide FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Tropicamide in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Tropicamide in pediatric patients.
Contraindications
There is limited information regarding Tropicamide Contraindications in the drug label.
Warnings
There is limited information regarding Tropicamide Warnings' in the drug label.
Adverse Reactions
Clinical Trials Experience
There is limited information regarding Tropicamide Clinical Trials Experience in the drug label.
Postmarketing Experience
There is limited information regarding Tropicamide Postmarketing Experience in the drug label.
Drug Interactions
There is limited information regarding Tropicamide Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
There is no FDA guidance on usage of Tropicamide in women who are pregnant.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Tropicamide in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Tropicamide during labor and delivery.
Nursing Mothers
There is no FDA guidance on the use of Tropicamide in women who are nursing.
Pediatric Use
There is no FDA guidance on the use of Tropicamide in pediatric settings.
Geriatic Use
There is no FDA guidance on the use of Tropicamide in geriatric settings.
Gender
There is no FDA guidance on the use of Tropicamide with respect to specific gender populations.
Race
There is no FDA guidance on the use of Tropicamide with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Tropicamide in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Tropicamide in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Tropicamide in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Tropicamide in patients who are immunocompromised.
Administration and Monitoring
Administration
There is limited information regarding Tropicamide Administration in the drug label.
Monitoring
There is limited information regarding Tropicamide Monitoring in the drug label.
IV Compatibility
There is limited information regarding the compatibility of Tropicamide and IV administrations.
Overdosage
There is limited information regarding Tropicamide overdosage. If you suspect drug poisoning or overdose, please contact the National Poison Help hotline (1-800-222-1222) immediately.
Pharmacology
There is limited information regarding Tropicamide Pharmacology in the drug label.
Mechanism of Action
There is limited information regarding Tropicamide Mechanism of Action in the drug label.
Structure
There is limited information regarding Tropicamide Structure in the drug label.
Pharmacodynamics
There is limited information regarding Tropicamide Pharmacodynamics in the drug label.
Pharmacokinetics
There is limited information regarding Tropicamide Pharmacokinetics in the drug label.
Nonclinical Toxicology
There is limited information regarding Tropicamide Nonclinical Toxicology in the drug label.
Clinical Studies
There is limited information regarding Tropicamide Clinical Studies in the drug label.
How Supplied
There is limited information regarding Tropicamide How Supplied in the drug label.
Storage
There is limited information regarding Tropicamide Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Tropicamide |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Tropicamide |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
There is limited information regarding Tropicamide Patient Counseling Information in the drug label.
Precautions with Alcohol
Alcohol-Tropicamide interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Tropicamide Brand Names in the drug label.
Look-Alike Drug Names
There is limited information regarding Tropicamide Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
| File:Tropicamide.svg | |
| Clinical data | |
|---|---|
| ATC code | |
| Pharmacokinetic data | |
| Protein binding | 45% |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C17H20N2O2 |
| Molar mass | 284.353 g/mol |
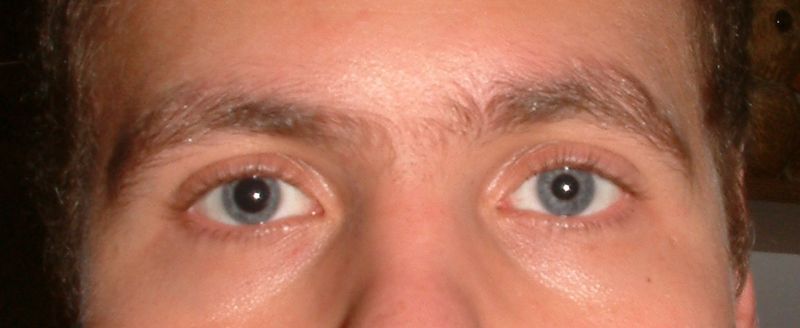
Tropicamide (troe-PIK-a-mide) is a muscarinic acetylcholine receptor blocker. It is used to dilate (enlarge) the pupil of the eye in order to allow better examination of the lens, vitreous humor, and retina. Due to its relatively short duration of effect (4 - 8 hours), it is typically used during eye examinations, but may also be used before or after eye surgery.
Application of tropicamide produces short acting mydriasis (dilation) and cycloplegia. Systemic side effects are very rare, although loss of consciousness has been reported in a ten-year-old child after administration of tropicamide eyedrops.[citation needed]
Tropicamide, a parasympatholytic, is occasionally administered in eye drops in combination with p-hydroxyamphetamine (brand name Paremyd), which is a sympathomimetic. The use of drugs stimulating multiple divisions of the autonomic nervous system ensures the dilation the pupil without opposing muscle group firing.
Other brand names include Mydriacyl.
See also
- Pages with script errors
- Pages with broken file links
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs with no legal status
- Articles containing unverified chemical infoboxes
- All articles with unsourced statements
- Articles with unsourced statements from September 2007
- Articles with invalid date parameter in template