Torsades de pointes medical therapy: Difference between revisions
No edit summary |
|||
| Line 25: | Line 25: | ||
|- | |- | ||
| bgcolor="LightGreen"|<nowiki>"</nowiki>'''2.''' Acute and long-term pacing is recommended for patients presenting with torsades de pointes due to [[heart block]] and symptomatic [[bradycardia]]. ''([[ACC AHA Guidelines Classification Scheme#Level of Evidence|Level of Evidence: A]])''<nowiki>"</nowiki> | | bgcolor="LightGreen"|<nowiki>"</nowiki>'''2.''' Acute and long-term pacing is recommended for patients presenting with torsades de pointes due to [[heart block]] and symptomatic [[bradycardia]]. ''([[ACC AHA Guidelines Classification Scheme#Level of Evidence|Level of Evidence: A]])''<nowiki>"</nowiki> | ||
|} | |||
{|class="wikitable" | |||
|- | |||
| colspan="1" style="text-align:center; background:LemonChiffon"|[[ACC AHA Guidelines Classification Scheme#Classification of Recommendations|Class IIa]] | |||
|- | |||
|bgcolor="LemonChiffon"|<nowiki>"</nowiki>'''1.''' Management with intravenous [[magnesium sulfate]] is reasonable for patients who present with [[LQTS]] and few episodes of torsades de pointes. [[Magnesium]] is not likely to be effective in patients with a normal [[QT interval]]. ''([[ACC AHA Guidelines Classification Scheme#Level of Evidence|Level of Evidence: B]])''<nowiki>"</nowiki> | |||
|- | |||
|bgcolor="LemonChiffon"|<nowiki>"</nowiki>'''2.''' Acute and long-term pacing is reasonable for patients who present with recurrent pause-dependent torsades de pointes. ''([[ACC AHA Guidelines Classification Scheme#Level of Evidence|Level of Evidence: B]])''<nowiki>"</nowiki> | |||
|- | |||
|bgcolor="LemonChiffon"|<nowiki>"</nowiki>'''3.''' [[Beta blockade]] combined with pacing is reasonable acute therapy for patients who present with torsades de pointes and [[sinus bradycardia]]. ''([[ACC AHA Guidelines Classification Scheme#Level of Evidence|Level of Evidence: C]])''<nowiki>"</nowiki> | |||
|- | |||
|bgcolor="LemonChiffon"|<nowiki>"</nowiki>'''4.''' [[Isoproterenol]] is reasonable as temporary treatment in acute patients who present with recurrent pause-dependent torsades de pointes who do not have congenital [[LQTS]]. ''([[ACC AHA Guidelines Classification Scheme#Level of Evidence|Level of Evidence: B]])''<nowiki>"</nowiki> | |||
|} | |} | ||
Revision as of 19:44, 26 October 2012
|
Torsades de pointes Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Torsades de pointes medical therapy On the Web |
|
American Roentgen Ray Society Images of Torsades de pointes medical therapy |
|
Risk calculators and risk factors for Torsades de pointes medical therapy |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
An understanding of the pathophysiology has led to development of treatment modalities like pacing, isoproterenol and drugs like magnesium and beta blockers.If the episode of does not terminate on its own and degenerates into ventricular fibrillation, cardioversion is required.
Medical Therapy
Acute Treatment
If the episode of does not terminate on its own and degenerates into ventricular fibrillation, cardioversion is required.
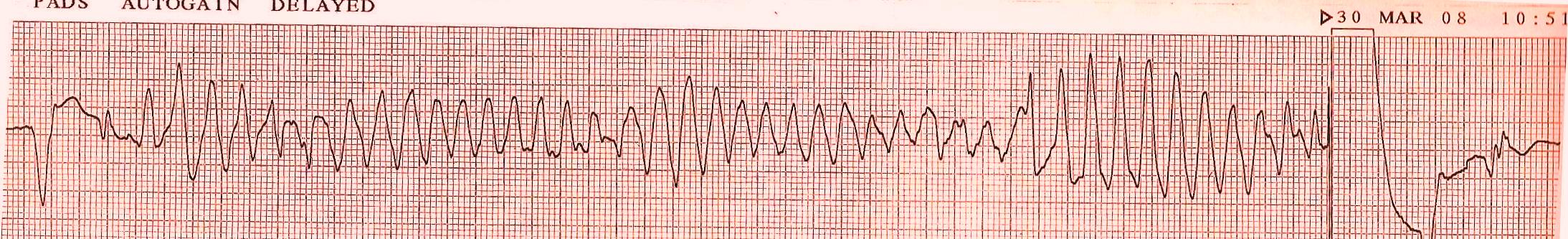
Once the patient is back in normal sinus rhythm, a vigorous search for and correction of conditions that predispose to torsades de pointes which include hypokalemia, hypomagnesemia, and bradycardia should be made. Magnesium sulfate (1-2 g IV over 30-60 seconds) reduces the influx of calcium thereby lowering the amplitude of early after depolarizations and should also be infused even if the magnesium is normal. [1][2] Administration of lidocaine is generally not effective, but mexiletene may be helpful in suppressing the recurrence of torsade de pointe.
ACC/AHA/ESC 2006 Guidelines for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death (DO NOT EDIT) [3]
| Class I |
| "1.Withdrawal of any offending drugs and correction of electrolyte abnormalities are recommended in patients presenting with torsades de pointes. (Level of Evidence: A) " |
| "2. Acute and long-term pacing is recommended for patients presenting with torsades de pointes due to heart block and symptomatic bradycardia. (Level of Evidence: A)" |
| Class IIa |
| "1. Management with intravenous magnesium sulfate is reasonable for patients who present with LQTS and few episodes of torsades de pointes. Magnesium is not likely to be effective in patients with a normal QT interval. (Level of Evidence: B)" |
| "2. Acute and long-term pacing is reasonable for patients who present with recurrent pause-dependent torsades de pointes. (Level of Evidence: B)" |
| "3. Beta blockade combined with pacing is reasonable acute therapy for patients who present with torsades de pointes and sinus bradycardia. (Level of Evidence: C)" |
| "4. Isoproterenol is reasonable as temporary treatment in acute patients who present with recurrent pause-dependent torsades de pointes who do not have congenital LQTS. (Level of Evidence: B)" |
References
- ↑ Hoshino K, Ogawa K, Hishitani T, Isobe T, Eto Y (2004). "Optimal administration dosage of magnesium sulfate for torsades de pointes in children with long QT syndrome". J Am Coll Nutr. 23 (5): 497S–500S. PMID 15466950. Unknown parameter
|month=ignored (help) - ↑ Hoshino K, Ogawa K, Hishitani T, Isobe T, Etoh Y (2006). "Successful uses of magnesium sulfate for torsades de pointes in children with long QT syndrome". Pediatr Int. 48 (2): 112–7. doi:10.1111/j.1442-200X.2006.02177.x. PMID 16635167. Unknown parameter
|month=ignored (help) - ↑ Zipes DP, Camm AJ, Borggrefe M, Buxton AE, Chaitman B, Fromer M; et al. (2006). "ACC/AHA/ESC 2006 Guidelines for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death: a report of the American College of Cardiology/American Heart Association Task Force and the European Society of Cardiology Committee for Practice Guidelines (writing committee to develop Guidelines for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death): developed in collaboration with the European Heart Rhythm Association and the Heart Rhythm Society". Circulation. 114 (10): e385–484. doi:10.1161/CIRCULATIONAHA.106.178233. PMID 16935995.