Ranibizumab
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Sheng Shi, M.D. [2]; Aparna Vuppala, M.B.B.S. [3]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Ranibizumab is a Monoclonal antibodies that is FDA approved for the treatment of neovascular (wet) age-related macular degeneration (AMD), macular edema following retinal vein occlusion (RVO), diabetic macular edema (DME). Common adverse reactions include cataract, conjunctival hemorrhage, eye irritation, foreign body sensation, in the eyes, inflammatory disorder of the eye, pain in eye, raised intraocular pressure, visual disturbance, vitreous detachment, vitreous floaters.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Neovascular (Wet) Age-Related Macular Degeneration (AMD)
- Dosing information
- Recommended dosage: 0.5 mg (0.05 mL of 10 mg/mL Ranibizumab solution) IV once a month (approximately 28 days).
- Although not as effective, patients may be treated with 3 monthly doses followed by less frequent dosing with regular assessment. In the nine months after 3 initial monthly doses, less frequent dosing with 4-5 doses on average is expected to maintain visual acuity while monthly dosing may be expected to result in an additional average 1-2 letter gain. Patients should be assessed regularly
- Although not as effective, patients may also be treated with one dose every 3 months after 4 monthly doses. Compared with continued monthly dosing, dosing every 3 months over the next 9 months will lead to an approximate 5-letter (1-line) loss of visual acuity benefit, on average. Patients should be assessed regularly
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ranibizumab in adult patients.
Non–Guideline-Supported Use
Choroidal retinal neovascularization
- Dosnig information
- ’‘’1.25 mg/0.05 mL or 0.5 mg/0.05 mL ‘’‘ [1]
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
The safety and effectiveness of Ranibizumab in pediatric patients have not been established.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Ranibizumab in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Ranibizumab in pediatric patients.
Contraindications
Ocular or Periocular Infections
Ranibizumab is contraindicated in patients with ocular or periocular infections.
Hypersensitivity
Ranibizumab is contraindicated in patients with known hypersensitivity to ranibizumab or any of the excipients in Ranibizumab. Hypersensitivity reactions may manifest as severe intraocular inflammation.
Warnings
Endophthalmitis and Retinal Detachments
Intravitreal injections, including those with Ranibizumab, have been associated with endophthalmitis and retinal detachments. Proper aseptic injection technique should always be used when administering Ranibizumab. In addition, patients should be monitored following the injection to permit early treatment should an infection occur.
Increases in Intraocular Pressure
Increases in intraocular pressure have been noted both pre-injection and post-injection (at 60 minutes) while being treated with Ranibizumab. Monitor intraocular pressure prior to and following intravitreal injection with Ranibizumab and manage appropriately.
Thromboembolic Events
Although there was a low rate of arterial thromboembolic events (ATEs) observed in the Ranibizumab clinical trials, there is a potential risk of ATEs following intravitreal use of VEGF inhibitors. ATEs are defined as nonfatal stroke, nonfatal myocardial infarction, or vascular death (including deaths of unknown cause).
Neovascular (Wet) Age-Related Macular Degeneration
The ATE rate in the three controlled neovascular AMD studies (AMD-1, AMD-2, AMD-3) during the first year was 1.9% (17 of 874) in the combined group of patients treated with 0.3 mg or 0.5 mg Ranibizumab compared with 1.1% (5 of 441) in patients from the control arms . In the second year of Studies AMD-1 and AMD-2, the ATE rate was 2.6% (19 of 721) in the combined group of Ranibizumab-treated patients compared with 2.9% (10 of 344) in patients from the control arms. In Study AMD-4, the ATE rates observed in the 0.5 mg arms during the first and second year were similar to rates observed in Studies AMD-1, AMD-2, and AMD-3. In a pooled analysis of 2-year controlled studies (AMD-1, AMD-2, and a study of Ranibizumab used adjunctively with verteporfin photodynamic therapy), the stroke rate (including both ischemic and hemorrhagic stroke) was 2.7% (13 of 484) in patients treated with 0.5 mg Ranibizumab compared to 1.1% (5 of 435) in patients in the control arms (odds ratio 2.2 (95% confidence interval (0.8-7.1))).
Macular Edema Following Retinal Vein Occlusion
The ATE rate in the two controlled RVO studies during the first 6 months was 0.8% in both the Ranibizumab and control arms of the studies (4 of 525 in the combined group of patients treated with 0.3 mg or 0.5 mg Ranibizumab and 2 of 260 in the control arms). The stroke rate was 0.2% (1 of 525) in the combined group of Ranibizumab-treated patients compared to 0.4% (1 of 260) in the control arms.
Diabetic Macular Edema
In a pooled analysis of Studies DME-1 and DME-2 , the ATE rate at 2 years was 7.2% (18 of 250) with 0.5 mg Ranibizumab, 5.6% (14 of 250) with 0.3 mg Ranibizumab, and 5.2% (13 of 250) with control. The stroke rate at 2 years was 3.2% (8 of 250) with 0.5 mg Ranibizumab, 1.2% (3 of 250) with 0.3 mg Ranibizumab, and 1.6% (4 of 250) with control. At 3 years, the ATE rate was 10.4% (26 of 249) with 0.5 mg Ranibizumab and 10.8% (27 of 250) with 0.3 mg Ranibizumab; the stroke rate was 4.8% (12 of 249) with 0.5 mg Ranibizumab and 2.0% (5 of 250) with 0.3 mg Ranibizumab.
Fatal Events in DME Patients
A pooled analysis of Studies DME-1 and DME-2 showed that fatalities in the first 2 years occurred in 4.4% (11 of 250) of patients treated with 0.5 mg Ranibizumab, in 2.8% (7 of 250) of patients treated with 0.3 mg Ranibizumab, and in 1.2% (3 of 250) of control patients. Over 3 years, fatalities occurred in 6.4% (16 of 249) of patients treated with 0.5 mg Ranibizumab and in 4.4% (11 of 250) of patients treated with 0.3 mg Ranibizumab. Although the rate of fatal events was low and included causes of death typical of patients with advanced diabetic complications, a potential relationship between these events and intravitreal use of VEGF inhibitors cannot be excluded.
Adverse Reactions
Clinical Trials Experience
Serious adverse reactions related to the injection procedure have occurred in < 0.1% of intravitreal injections, including endophthalmitis, rhegmatogenous retinal detachment, and iatrogenic traumatic cataract.
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in one clinical trial of a drug cannot be directly compared with rates in the clinical trials of the same or another drug and may not reflect the rates observed in practice. The data below reflect exposure to 0.5 mg Ranibizumab in 440 patients with neovascular AMD in Studies AMD-1, AMD-2, and AMD-3, and 259 patients with macular edema following RVO. The data also reflect exposure to 0.3 mg Ranibizumab in 250 patients with DME. Safety data observed in Study AMD-4 were consistent with these results. On average, the rates and types of adverse reactions in patients were not significantly affected by dosing regimen.
Ocular Reactions
Table 1 shows frequently reported ocular adverse reactions in Ranibizumab-treated patients compared with the control group.
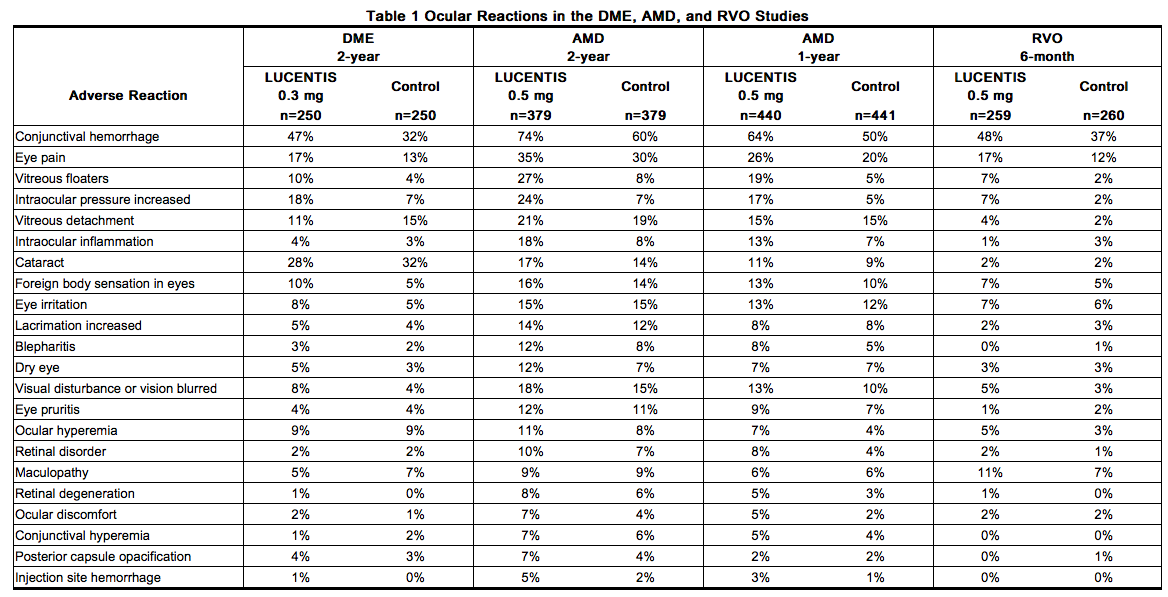
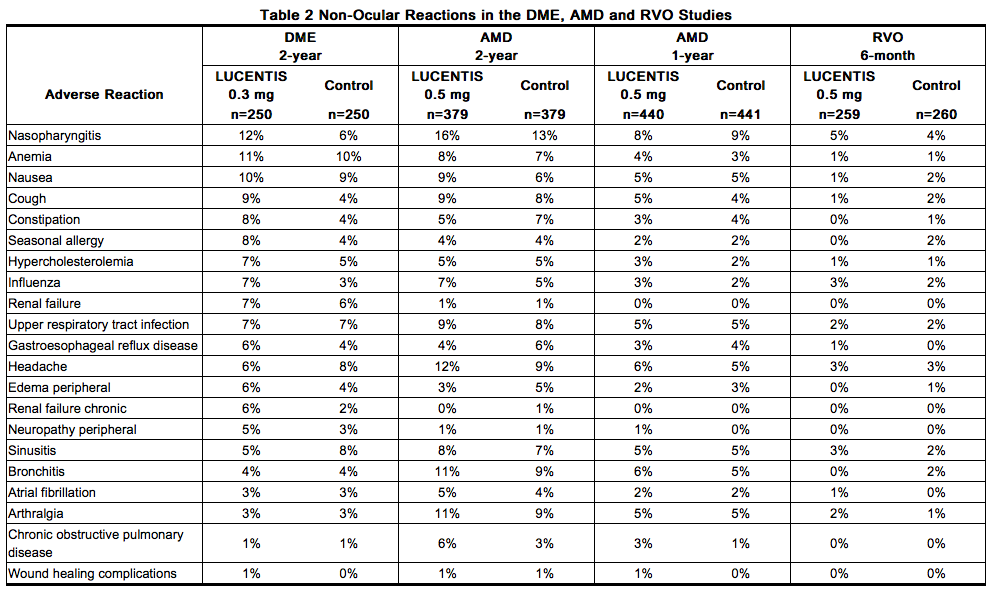
Non-Ocular Reactions
Non-ocular adverse reactions with an incidence of ≥ 5% in patients receiving Ranibizumab for DME, AMD, and/or RVO and which occurred at a ≥ 1% higher frequency in patients treated with Ranibizumab compared to control are shown in Table 2. Though less common, wound healing complications were also observed in some studies.
Immunogenicity
As with all therapeutic proteins, there is the potential for an immune response in patients treated with Ranibizumab. The immunogenicity data reflect the percentage of patients whose test results were considered positive for antibodies to Ranibizumab in immunoassays and are highly dependent on the sensitivity and specificity of the assays. The pre-treatment incidence of immunoreactivity to Ranibizumab was 0%-5% across treatment groups. After monthly dosing with Ranibizumab for 6 to 24 months, antibodies to Ranibizumab were detected in approximately 1%-9% of patients. The clinical significance of immunoreactivity to Ranibizumab is unclear at this time. Among neovascular AMD patients with the highest levels of immunoreactivity, some were noted to have iritis or vitritis. Intraocular inflammation was not observed in DME or RVO patients with the highest levels of immunoreactivity.
Postmarketing Experience
The following adverse reactions have been identified during post-approval use of Ranibizumab. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure. ‘’‘Ocular’‘’: Tear of retinal pigment epithelium among patients with neovascular AMD
Drug Interactions
Drug interaction studies have not been conducted with Ranibizumab. Ranibizumab intravitreal injection has been used adjunctively with verteporfin photodynamic therapy (PDT). Twelve (12) of 105 (11%) patients with neovascular AMD developed serious intraocular inflammation; in 10 of the 12 patients, this occurred when Ranibizumab was administered 7 days (± 2 days) after verteporfin PDT.
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): C
There are no studies of Ranibizumab in pregnant women. An embryo-fetal developmental toxicity study was performed on pregnant cynomolgus monkeys. Pregnant animals received intravitreal injections of ranibizumab every 14 days starting on Day 20 of gestation, until Day 62 at doses of 0, 0.125, and 1 mg/eye. Skeletal abnormalities including incomplete and/or irregular ossification of bones in the skull, vertebral column, and hindlimbs and shortened supernumerary ribs were seen at a low incidence in fetuses from animals treated with 1 mg/eye of ranibizumab. The 1 mg/eye dose resulted in trough serum ranibizumab levels up to 13 times higher than predicted Cmax levels with single eye treatment in humans. No skeletal abnormalities were seen at the lower dose of 0.125 mg/eye, a dose which resulted in trough exposures equivalent to single eye treatment in humans. No effect on the weight or structure of the placenta, maternal toxicity, or embryotoxicity was observed.
Animal reproduction studies are not always predictive of human response. It is also not known whether ranibizumab can cause fetal harm when administered to a pregnant woman or can affect reproduction capacity. Based on the anti-VEGF mechanism of action for ranibizumab, treatment with Ranibizumab may pose a risk to embryo-fetal development (including teratogenicity) and reproductive capacity. Ranibizumab should be given to a pregnant woman only if clearly needed.
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Ranibizumab in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Ranibizumab during labor and delivery.
Nursing Mothers
It is not known whether ranibizumab is excreted in human milk. Because many drugs are excreted in human milk, and because the potential for absorption and harm to infant growth and development exists, caution should be exercised when Ranibizumab is administered to a nursing woman.
Pediatric Use
The safety and effectiveness of Ranibizumab in pediatric patients have not been established.
Geriatic Use
In the clinical studies, approximately 79% (2387 of 3005) of patients randomized to treatment with Ranibizumab were ≥ 65 years of age and approximately 54% (1636 of 3005) were ≥ 75 years of age. No notable differences in efficacy or safety were seen with increasing age in these studies. Age did not have a significant effect on systemic exposure.
Gender
There is no FDA guidance on the use of Ranibizumab with respect to specific gender populations.
Race
There is no FDA guidance on the use of Ranibizumab with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Ranibizumab in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Ranibizumab in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Ranibizumab in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Ranibizumab in patients who are immunocompromised.
Administration and Monitoring
Administration
FOR OPHTHALMIC INTRAVITREAL INJECTION ONLY
Monitoring
FDA Package Insert for Ranibizumab contains no information regarding drug monitoring.
IV Compatibility
There is limited information about the IV Compatibility.
Overdosage
More concentrated doses as high as 2 mg ranibizumab in 0.05 mL have been administered to patients. No additional unexpected adverse reactions were seen.
Pharmacology
Ranibizumab?
| |
| Therapeutic monoclonal antibody | |
| Source | zu/o |
| Target | VEGF-A |
| Identifiers | |
| CAS number | |
| ATC code | S01 |
| PubChem | ? |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 48,350 g/mol |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | Approx. 9 days[2] |
| Excretion | ? |
| Therapeutic considerations | |
| Licence data |
, |
| Pregnancy cat. |
C(US) |
| Legal status |
POM(UK) [[Prescription drug|Template:Unicode-only]](US) |
| Routes | Intravitreal injection |
Mechanism of Action
Ranibizumab binds to the receptor binding site of active forms of VEGF-A, including the biologically active, cleaved form of this molecule, VEGF110. VEGF-A has been shown to cause neovascularization and leakage in models of ocular angiogenesis and vascular occlusion and is thought to contribute to pathophysiology of neovascular AMD, macular edema following RVO, and DME. The binding of ranibizumab to VEGF-A prevents the interaction of VEGF-A with its receptors (VEGF1 and VEGFR2) on the surface of endothelial cells, reducing endothelial cell proliferation, vascular leakage, and new blood vessel formation.
Structure
Ranibizumab® (ranibizumab injection) is a recombinant humanized IgG1 kappa isotype monoclonal antibody fragment designed for intraocular use. Ranibizumab binds to and inhibits the biologic activity of human vascular endothelial growth factor A (VEGF-A). Ranibizumab, which lacks an Fc region, has a molecular weight of approximately 48 kilodaltons and is produced by an E. coli expression system in a nutrient medium containing the antibiotic tetracycline. Tetracycline is not detectable in the final product. Ranibizumab is a sterile, colorless to pale yellow solution in a single-use glass vial. Ranibizumab is supplied as a preservative-free, sterile solution in a single-use glass vial designed to deliver 0.05 mL of 10 mg/mL Ranibizumab (0.5 mg dose vial) or 6 mg/mL Ranibizumab (0.3 mg dose vial) aqueous solution with 10 mM histidine HCl, 10% α,α-trehalose dihydrate, 0.01% polysorbate 20, pH 5.5.
Pharmacodynamics
Increased retinal thickness (i.e., center point thickness (CPT) or central foveal thickness (CFT)), as assessed by optical coherence tomography (OCT) is associated with neovascular AMD, macular edema following RVO, and DME. Leakage from choroidal neovascularization (CNV) as assessed by fluorescein angiography (FA) is associated with neovascular AMD.
Neovascular (Wet) Age-Related Macular Degeneration
In Study AMD-3, CPT was assessed by time domain (TD)-OCT in 118 of 184 patients. TD-OCT measurements were collected at baseline, Months 1, 2, 3, 5, 8, and 12. In patients treated with Ranibizumab, CPT decreased, on average, more than in the sham group from baseline through Month 12. CPT decreased by Month 1 and decreased further at Month 3, on average. In this study, CPT data did not provide information useful in influencing treatment decisions. In Study AMD-4, CFT was assessed by spectral domain (SD)-OCT in all patients; on average, CFT reductions were observed beginning at Day 7 following the first Ranibizumab injection through Month 24. CFT data did not provide information capable of predicting final visual acuity results . In patients treated with Ranibizumab, the area of CNV leakage, on average, decreased by Month 3 as assessed by FA. The area of CNV leakage for an individual patient was not correlated with visual acuity.
Macular Edema Following Retinal Vein Occlusion
On average, CPT reductions were observed in Studies RVO-1 and RVO-2 beginning at Day 7 following the first Ranibizumab injection through Month 6. CPT was not evaluated as a means to guide treatment decisions.
Diabetic Macular Edema On average, CPT reductions were observed in Studies DME-1 and DME-2 beginning at Day 7 following the first Ranibizumab injection through Month 36. CPT data did not provide information useful in influencing treatment decisions.
Pharmacokinetics
In animal studies, following intravitreal injection, ranibizumab was cleared from the vitreous with a half-life of approximately 3 days. After reaching a maximum at approximately 1 day, the serum concentration of ranibizumab declined in parallel with the vitreous concentration. In these animal studies, systemic exposure of ranibizumab was more than 2000-fold lower than in the vitreous. In patients with neovascular AMD, following monthly intravitreal administration, maximum ranibizumab serum concentrations were low (0.3 ng/mL to 2.36 ng/mL). These levels were below the concentration of ranibizumab (11 ng/mL to 27 ng/mL) thought to be necessary to inhibit the biological activity of VEGF-A by 50%, as measured in an in vitro cellular proliferation assay. The maximum observed serum concentration was dose proportional over the dose range of 0.05 to 1 mg/eye. Serum ranibizumab concentrations in RVO and DME patients were similar to those observed in neovascular AMD patients. Based on a population pharmacokinetic analysis of patients with neovascular AMD, maximum serum concentrations of 1.5 ng/mL are predicted to be reached at approximately 1 day after monthly intravitreal administration of Ranibizumab 0.5 mg/eye. Based on the disappearance of ranibizumab from serum, the estimated average vitreous elimination half-life was approximately 9 days. Steady-state minimum concentration is predicted to be 0.22 ng/mL with a monthly dosing regimen. In humans, serum ranibizumab concentrations are predicted to be approximately 90,000-fold lower than vitreal concentrations. In pharmacokinetic covariate analyses, 48% (520/1091) of patients had renal impairment (35% mild, 11% moderate, and 2% severe). Because the increases in plasma ranibizumab exposures in these patients are not considered clinically significant, no dosage adjustment is needed based on renal impairment status.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
No carcinogenicity or mutagenicity data are available for ranibizumab injection in animals or humans. No studies on the effects of ranibizumab on fertility have been conducted. Although systemic exposure following ocular administration is expected to be low, effects on female fertility are possible due to the anti-VEGF mechanism of action for ranibizumab
Clinical Studies
Unless otherwise noted, visual acuity was measured at a distance of 4 meters.
Neovascular (Wet) Age-Related Macular Degeneration (AMD)
The safety and efficacy of Ranibizumab were assessed in three randomized, double-masked, sham- or active-controlled studies in patients with neovascular AMD. A total of 1323 patients (Ranibizumab 879, control 444) were enrolled in the three studies. Studies AMD-1 and AMD-2 In Study AMD-1, patients with minimally classic or occult (without classic) CNV lesions received monthly Ranibizumab 0.3 mg or 0.5 mg intravitreal injections or monthly sham injections. Data are available through Month 24. Patients treated with Ranibizumab in Study AMD-1 received a mean of 22 total treatments out of a possible 24 from Day 0 to Month 24. In Study AMD-2, patients with predominantly classic CNV lesions received one of the following: 1) monthly Ranibizumab 0.3 mg intravitreal injections and sham PDT; 2) monthly Ranibizumab 0.5 mg intravitreal injections and sham PDT; or 3) sham intravitreal injections and active verteporfin PDT. Sham PDT (or active verteporfin PDT) was given with the initial Ranibizumab (or sham) intravitreal injection and every 3 months thereafter if fluorescein angiography showed persistence or recurrence of leakage. Data are available through Month 24. Patients treated with Ranibizumab in Study AMD-2 received a mean of 21 total treatments out of a possible 24 from Day 0 through Month 24. In both studies, the primary efficacy endpoint was the proportion of patients who maintained vision, defined as losing fewer than 15 letters of visual acuity at 12 months compared with baseline. Almost all Ranibizumab-treated patients (approximately 95%) maintained their visual acuity. Among Ranibizumab-treated patients, 31% to 37% experienced a clinically significant improvement in vision, defined as gaining 15 or more letters at 12 months. The size of the lesion did not significantly affect the results. Detailed results are shown in Table 3, Table 4, and Figure 1 below.
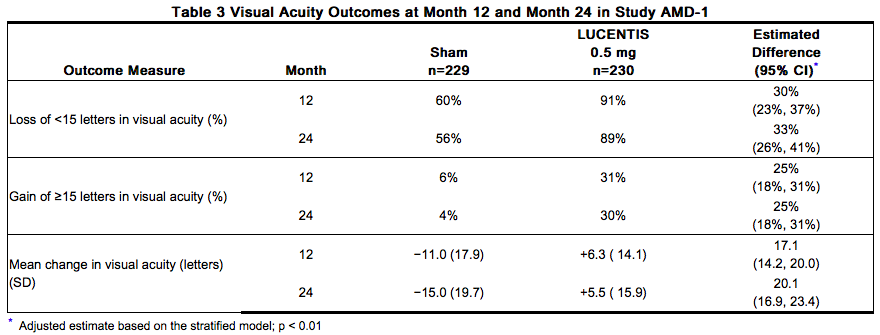
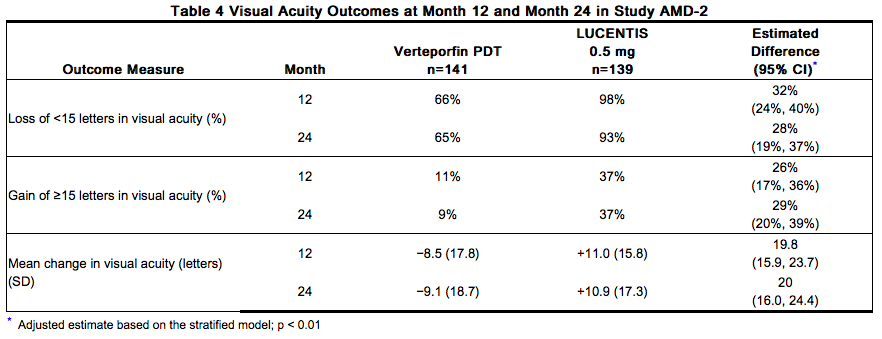
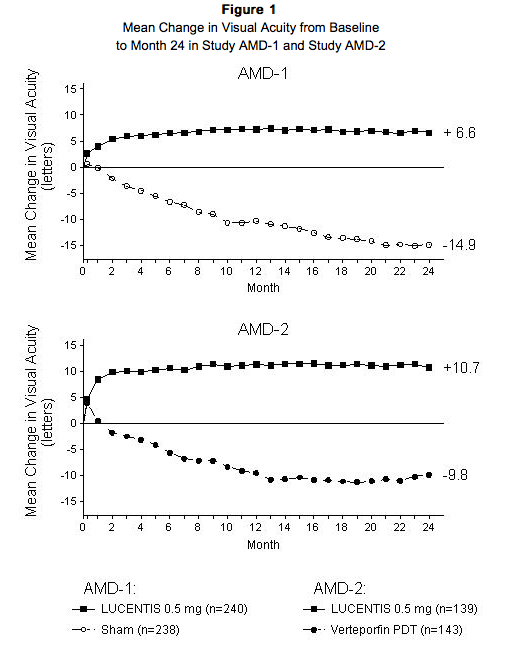
Visual acuity was measured at a distance of 2 meters Patients in the group treated with Ranibizumab had minimal observable CNV lesion growth, on average. At Month 12, the mean change in the total area of the CNV lesion was 0.1-0.3 disc areas (DA) for Ranibizumab versus 2.3-2.6 DA for the control arms. At Month 24, the mean change in the total area of the CNV lesion was 0.3-0.4 DA for Ranibizumab versus 2.9-3.1 DA for the control arms.
Study AMD-3
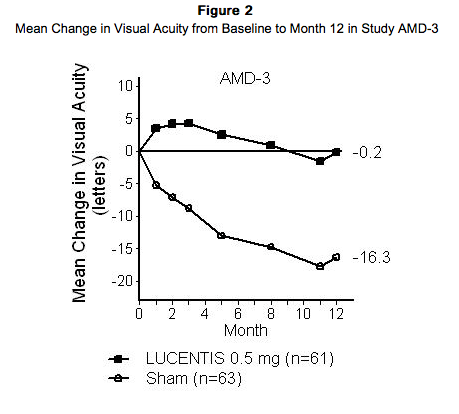
Study AMD-3 was a randomized, double-masked, sham-controlled, two-year study designed to assess the safety and efficacy of Ranibizumab in patients with neovascular AMD (with or without a classic CNV component). Data are available through Month 12. Patients received Ranibizumab 0.3 mg or 0.5 mg intravitreal injections or sham injections once a month for 3 consecutive doses, followed by a dose administered once every 3 months for 9 months. A total of 184 patients were enrolled in this study (Ranibizumab 0.3 mg, 60; Ranibizumab 0.5 mg, 61; sham, 63); 171 (93%) completed 12 months of this study. Patients treated with Ranibizumab in Study AMD-3 received a mean of 6 total treatments out of a possible 6 from Day 0 through Month 12. In Study AMD-3, the primary efficacy endpoint was mean change in visual acuity at 12 months compared with baseline (see Figure 2). After an initial increase in visual acuity (following monthly dosing), on average, patients dosed once every 3 months with Ranibizumab lost visual acuity, returning to baseline at Month 12. In Study AMD-3, almost all Ranibizumab-treated patients (90%) lost fewer than 15 letters of visual acuity at Month 12.
Study AMD-4
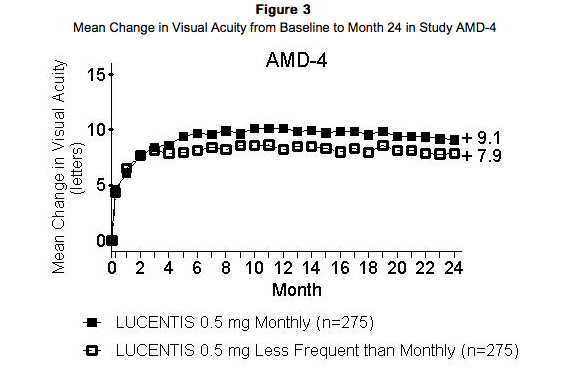
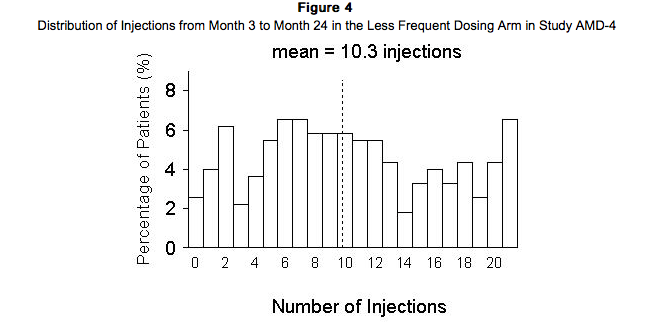
Study AMD-4 was a randomized, double-masked, active treatment-controlled, two-year study designed to assess the safety and efficacy of Ranibizumab 0.5 mg administered monthly or less frequently than monthly in patients with neovascular AMD. Patients randomized to the Ranibizumab 0.5 mg less frequent dosing arm received 3 monthly doses followed by monthly assessments where patients were eligible to receive Ranibizumab injections guided by pre-specified re-treatment criteria. A total of 550 patients were enrolled in the two 0.5 mg treatment groups with 467 (85%) completing through Month 24. Data are available through Month 24. Clinical results at Month 24 remain similar to that observed at Month 12. From Month 3 through Month 24, visual acuity decreased by 0.3 letters in the 0.5 mg less frequent dosing arm and increased by 0.7 letters in the 0.5 mg monthly arm. Over this 21 month period, patients in the 0.5 mg less frequent dosing and the 0.5 mg monthly arms averaged 10.3 and 18.5 injections, respectively. The distribution of injections received in the less frequent dosing arm is shown in Figure 4.
Macular Edema Following Retinal Vein Occlusion (RVO)
The safety and efficacy of Ranibizumab were assessed in two randomized, double-masked, 1-year studies in patients with macular edema following RVO. Sham controlled data are available through Month 6. Patient age ranged from 20 to 91 years, with a mean age of 67 years. A total of 789 patients (Ranibizumab 0.3 mg, 266 patients; Ranibizumab 0.5 mg, 261 patients; sham, 262 patients) were enrolled, with 739 (94%) patients completing through Month 6. All patients completing Month 6 were eligible to receive Ranibizumab injections guided by pre-specified re-treatment criteria until the end of the studies at Month 12. In Study RVO-1, patients with macular edema following branch or hemi-RVO, received monthly Ranibizumab 0.3 mg or 0.5 mg intravitreal injections or monthly sham injections for 6 months. All patients were eligible for macular focal/grid laser treatment beginning at Month 3 of the 6-month treatment period. Macular focal/grid laser treatment was given to 26 of 131 (20%) patients treated with 0.5 mg Ranibizumab and 71 of 132 (54%) patients treated with sham. In Study RVO-2, patients with macular edema following central RVO received monthly Ranibizumab 0.3 mg or 0.5 mg intravitreal injections or monthly sham injections for 6 months. At Month 6, after monthly treatment with 0.5 mg Ranibizumab, the following clinical results were observed:
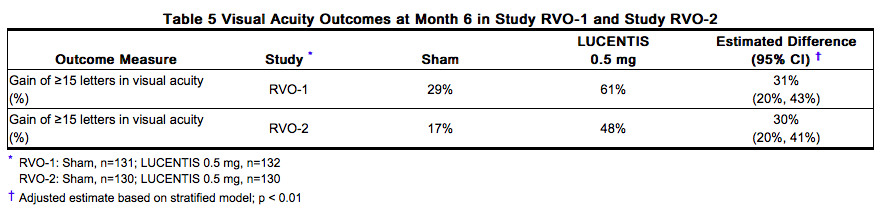
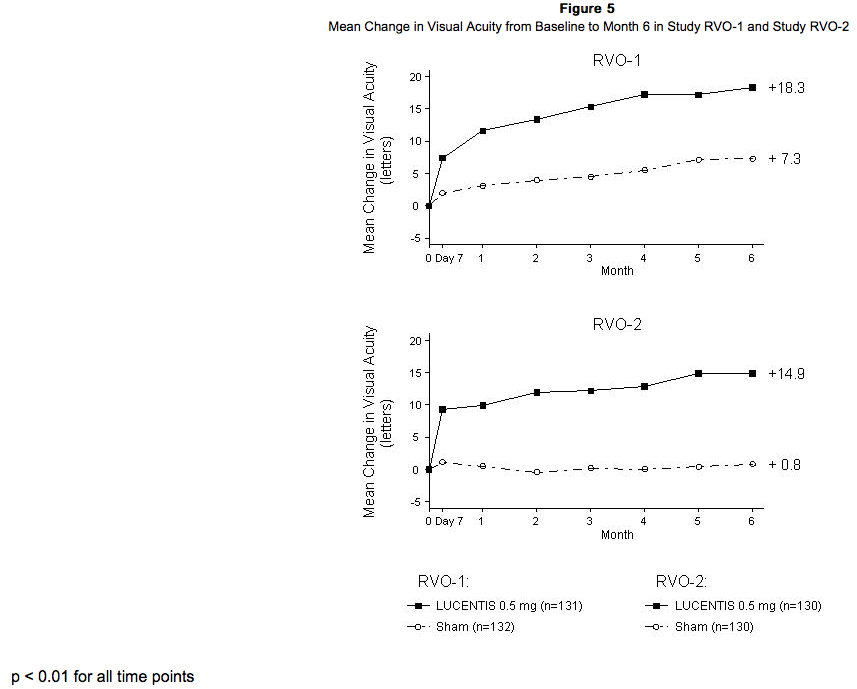
Diabetic Macular Edema (DME)
The safety and efficacy of Ranibizumab were assessed in two randomized, double-masked, 3-year studies in patients with DME. The studies were sham-controlled through Month 24. Patient age ranged from 21 to 91 years, with a mean age of 62 years. A total of 759 patients (Ranibizumab 0.3 mg, 250 patients; Ranibizumab 0.5 mg, 252 patients; sham, 257 patients) were enrolled, with 582 (77%) completing through Month 36. In Studies DME-1 and DME-2, patients received monthly Ranibizumab 0.3 mg or 0.5 mg intravitreal injections or monthly sham injections during the 24-month controlled treatment period. From Months 25 through 36, patients who previously received sham were eligible to receive monthly Ranibizumab 0.5 mg and patients originally randomized to monthly Ranibizumab 0.3 mg or 0.5 mg continued to receive their assigned dose. All patients were eligible for macular focal/grid laser treatment beginning at Month 3 of the 24-month treatment period or panretinal photocoagulation (PRP) as needed. Through Month 24, macular focal/grid laser treatment was administered in 94 of 250 (38%) patients treated with Ranibizumab 0.3 mg and 185 of 257 (72%) patients treated with sham; PRP was administered in 2 of 250 (1%) patients treated with Ranibizumab 0.3 mg and 30 of 257 (12%) patients treated with sham. Compared to monthly Ranibizumab 0.3 mg, no additional benefit was observed with monthly treatment with Ranibizumab 0.5 mg. At Month 24, after monthly treatment with Ranibizumab 0.3 mg, the following clinical results were observed:
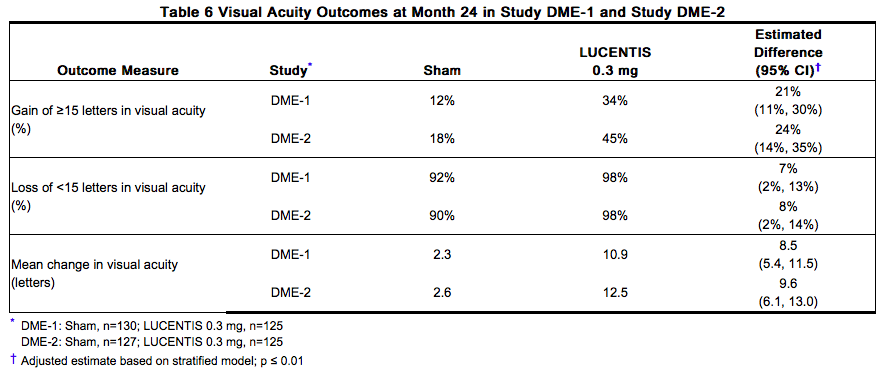
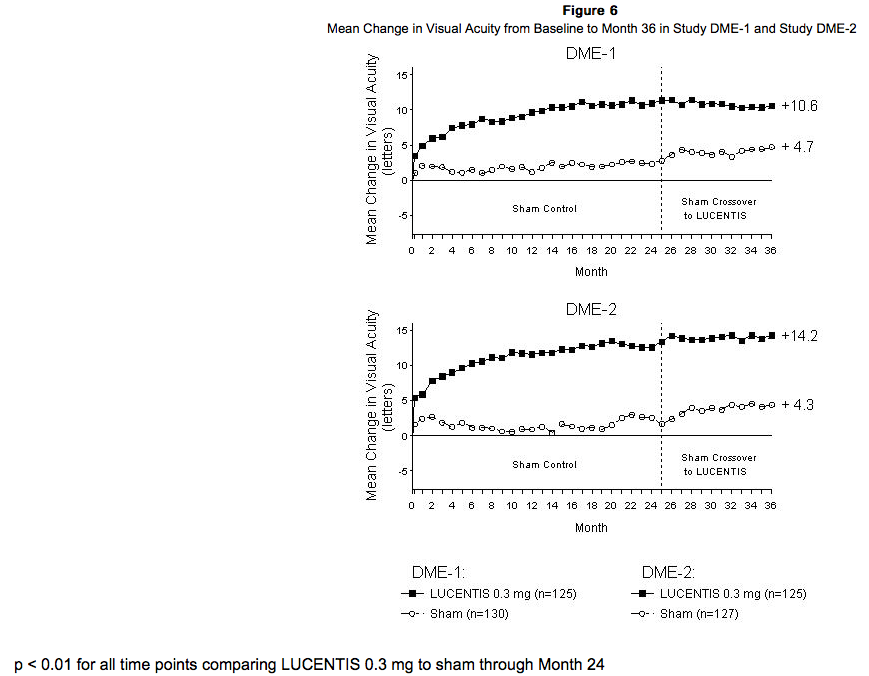
VA outcomes observed at Month 24 in patients treated with Ranibizumab 0.3 mg were maintained with continued treatment through Month 36 in both DME studies. Patients in the sham arms who received Ranibizumab 0.5 mg beginning at Month 25 achieved lesser VA gains compared to patients who began treatment with Ranibizumab at the beginning of the studies.
How Supplied
- Each Ranibizumab 0.5 mg carton (NDC 50242-080-01) contains a single-use, 2-cc glass vial with a BLUE CAP designed to deliver 0.05 mL of 10 mg/mL ranibizumab.
- Each Ranibizumab 0.3 mg carton (NDC 50242-082-01) contains a single-use, 2-cc glass vial with a WHITE CAP designed to deliver 0.05 mL of 6 mg/mL ranibizumab.
In addition, each carton contains one 5-micron, 19-gauge × 1-1/2-inch filter needle for withdrawal of the vial contents; one 30-gauge × 1/2-inch injection needle for the intravitreal injection; and one package insert . VIALS ARE FOR SINGLE-EYE USE ONLY.
Storage
Ranibizumab should be refrigerated at 2°-8°C (36°-46°F). DO NOT FREEZE. Do not use beyond the date stamped on the label. Ranibizumab vials should be protected from light. Store in the original carton until time of use.
Images
Drug Images
{{#ask: Page Name::Ranibizumab |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Ranibizumab |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
In the days following Ranibizumab administration, patients are at risk of developing endophthalmitis. If the eye becomes red, sensitive to light, painful, or develops a change in vision, the patient should seek immediate care from an ophthalmologist. Ranibizumab® [ranibizumab injection] Manufactured by: Genentech, Inc. A Member of the Roche Group 1 DNA Way South San Francisco, CA 94080-4990 Ranibizumab® is a registered trademark of Genentech, Inc. ©2014 Genentech, Inc. Representative sample of labeling (see the HOW SUPPLIED section for complete listing): PRINCIPAL DISPLAY PANEL - 0.5 mg Vial Carton NDC 50242-080-01 10140896 Ranibizumab® RANIBIZUMAB INJECTION 0.5 mg DOSE SINGLE-USE VIAL INDICATED FOR Neovascular (wet) age-related macular degeneration (wAMD) Macular edema following retinal vein occlusion (RVO) 0.5 mg wAMD
Precautions with Alcohol
Alcohol-Ranibizumab interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
LUCENTIS
Look-Alike Drug Names
There is limited information regarding Ranibizumab Look-Alike Drug Names in the drug label.
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ Iacono P, Parodi MB, Papayannis A, Kontadakis S, Sheth S, Cascavilla ML; et al. (2012). "Intravitreal ranibizumab versus bevacizumab for treatment of myopic choroidal neovascularization". Retina. 32 (8): 1539–46. doi:10.1097/IAE.0b013e31826956b7. PMID 22922846.
- ↑ Ranibizumab Prescribing Information. Genentech. June 2010.
{{#subobject:
|Label Page=Ranibizumab |Label Name=Ranibizumab_label_01.jpg
}}
{{#subobject:
|Label Page=Ranibizumab |Label Name=Ranibizumab_label_02.jpg
}}
{{#subobject:
|Label Page=Ranibizumab |Label Name=Ranibizumab_panel_01.png
}}
{{#subobject:
|Label Page=Ranibizumab |Label Name=Ranibizumab_panel_02.png
}}