Congenital adrenal hyperplasia Overview: Difference between revisions
No edit summary |
|||
| (2 intermediate revisions by the same user not shown) | |||
| Line 10: | Line 10: | ||
Prenatal diagnosis can be made in both of these disorders by chorionic villous sampling, but this can only be done at 8-10 weeks. In order to prevent the deleterious effect of excess androgens on genital (and brain!) development, therapy must be started earlier. This is most often considered if there is an affected sibling. Treatment is dexamethasone, which is not degraded by the placenta, but is associated with significant maternal weight gain, hypertension, and edema. | Prenatal diagnosis can be made in both of these disorders by chorionic villous sampling, but this can only be done at 8-10 weeks. In order to prevent the deleterious effect of excess androgens on genital (and brain!) development, therapy must be started earlier. This is most often considered if there is an affected sibling. Treatment is dexamethasone, which is not degraded by the placenta, but is associated with significant maternal weight gain, hypertension, and edema. | ||
== | ==Classification== | ||
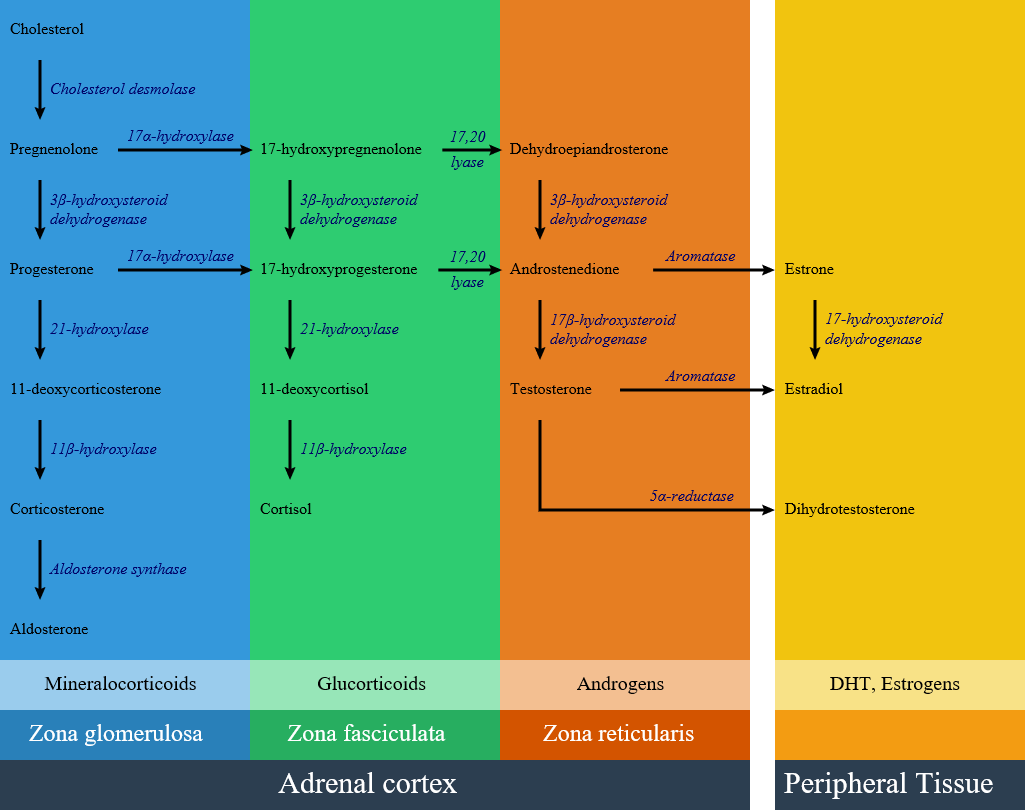
Congenital adrenal hyperplasia is classified into seven types based on the genetic causes that lead to hyperplasia and hormonal imbalance. There are three zones of hormonal synthesis in adrenal cortex that are shown below, and impairment of each pathway may lead to a specific subtype of congenital adrenal hyperplasia. | |||
[[image:Adrenal Steroids.png|600px]] | |||
[[ | |||
{| align="center" class="wikitable" style="border: 0px; font-size: 90%; margin: 3px;" | {| align="center" class="wikitable" style="border: 0px; font-size: 90%; margin: 3px;" | ||
| Line 188: | Line 145: | ||
|} | |} | ||
== | ==Differential Diagnosis== | ||
[[Congenital adrenal hyperplasia]] must be differentiated from diseases that cause [[ambiguous genitalia]]:<ref name="pmid17875484">{{cite journal |vauthors=Hughes IA, Nihoul-Fékété C, Thomas B, Cohen-Kettenis PT |title=Consequences of the ESPE/LWPES guidelines for diagnosis and treatment of disorders of sex development |journal=Best Pract. Res. Clin. Endocrinol. Metab. |volume=21 |issue=3 |pages=351–65 |year=2007 |pmid=17875484 |doi=10.1016/j.beem.2007.06.003 |url=}}</ref><ref name="pmid10857554">{{cite journal |vauthors=White PC, Speiser PW |title=Congenital adrenal hyperplasia due to 21-hydroxylase deficiency |journal=Endocr. Rev. |volume=21 |issue=3 |pages=245–91 |year=2000 |pmid=10857554 |doi=10.1210/edrv.21.3.0398 |url=}}</ref> | [[Congenital adrenal hyperplasia]] must be differentiated from diseases that cause [[ambiguous genitalia]]:<ref name="pmid17875484">{{cite journal |vauthors=Hughes IA, Nihoul-Fékété C, Thomas B, Cohen-Kettenis PT |title=Consequences of the ESPE/LWPES guidelines for diagnosis and treatment of disorders of sex development |journal=Best Pract. Res. Clin. Endocrinol. Metab. |volume=21 |issue=3 |pages=351–65 |year=2007 |pmid=17875484 |doi=10.1016/j.beem.2007.06.003 |url=}}</ref><ref name="pmid10857554">{{cite journal |vauthors=White PC, Speiser PW |title=Congenital adrenal hyperplasia due to 21-hydroxylase deficiency |journal=Endocr. Rev. |volume=21 |issue=3 |pages=245–91 |year=2000 |pmid=10857554 |doi=10.1210/edrv.21.3.0398 |url=}}</ref> | ||
{| class="wikitable" | {| class="wikitable" | ||
| Line 376: | Line 324: | ||
* [[Infertility]], [[galactorrhea]] | * [[Infertility]], [[galactorrhea]] | ||
|} | |} | ||
==Screening== | |||
According to Endocrine Society Clinical Practice Guideline, screening for 21-hydroxylase deficiency by measuring 17a-hydroxyprogesterone is recommended for all newborns. | |||
*Blood sample on filter paper should be obtained from heel puncture preferably between two and four days after birth. | |||
*Screening programs should be done using a two-tier protocol (initial immunoassay with further evaluation of positive tests by liquid chromatography/tandem mass spectrometry. | |||
*Most affected neonates have concentrations greater than 3500 ng/dL (105 nmol/L).<ref name="pmid2208708">{{cite journal |vauthors=Gonzalez RR, Mäentausta O, Solyom J, Vihko R |title=Direct solid-phase time-resolved fluoroimmunoassay of 17 alpha-hydroxyprogesterone in serum and dried blood spots on filter paper |journal=Clin. Chem. |volume=36 |issue=9 |pages=1667–72 |year=1990 |pmid=2208708 |doi= |url=}}</ref><ref name="pmid20823466">{{cite journal |vauthors=Speiser PW, Azziz R, Baskin LS, Ghizzoni L, Hensle TW, Merke DP, Meyer-Bahlburg HF, Miller WL, Montori VM, Oberfield SE, Ritzen M, White PC |title=Congenital adrenal hyperplasia due to steroid 21-hydroxylase deficiency: an Endocrine Society clinical practice guideline |journal=J. Clin. Endocrinol. Metab. |volume=95 |issue=9 |pages=4133–60 |year=2010 |pmid=20823466 |pmc=2936060 |doi=10.1210/jc.2009-2631 |url=}}</ref> | |||
===Genetic counseling=== | |||
The Endocrine Society's Clinical Practice Guideline recommends that genetic counseling be provided for individuals who are planning to conceive, and there is a family history of 21-hydroxylase deficiency.<ref name="pmid20823466">{{cite journal |vauthors=Speiser PW, Azziz R, Baskin LS, Ghizzoni L, Hensle TW, Merke DP, Meyer-Bahlburg HF, Miller WL, Montori VM, Oberfield SE, Ritzen M, White PC |title=Congenital adrenal hyperplasia due to steroid 21-hydroxylase deficiency: an Endocrine Society clinical practice guideline |journal=J. Clin. Endocrinol. Metab. |volume=95 |issue=9 |pages=4133–60 |year=2010 |pmid=20823466 |pmc=2936060 |doi=10.1210/jc.2009-2631 |url=}}</ref> | |||
==References== | ==References== | ||
{{reflist|2}} | {{reflist|2}} | ||
Latest revision as of 19:26, 1 August 2017
|
Congenital adrenal hyperplasia main page |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Mehrian Jafarizade, M.D [2]
Synonyms and keywords: Congenital adrenal hyperplasia, CAH, Adrenal hyperplasia
Overview
Congenital adrenal hyperplasia (CAH) refers to any of several autosomal recessive conditions resulting from biochemical paths of the steroidogenesis of cortisol from cholesterol by the adrenal glands. Most of these conditions involve greater or lesser production of sex steroids and can alter development of primary or secondary sex characteristics in affected infants, children, and adults. Only a small minority of people with CAH can be said to have an intersex condition, but this attracted American public attention in the late 1990s and many accounts of varying accuracy have appeared in the popular media. Approximately 95% of cases of CAH are due to 21-hydroxylase deficiency. Prenatal diagnosis can be made in both of these disorders by chorionic villous sampling, but this can only be done at 8-10 weeks. In order to prevent the deleterious effect of excess androgens on genital (and brain!) development, therapy must be started earlier. This is most often considered if there is an affected sibling. Treatment is dexamethasone, which is not degraded by the placenta, but is associated with significant maternal weight gain, hypertension, and edema.
Classification
Congenital adrenal hyperplasia is classified into seven types based on the genetic causes that lead to hyperplasia and hormonal imbalance. There are three zones of hormonal synthesis in adrenal cortex that are shown below, and impairment of each pathway may lead to a specific subtype of congenital adrenal hyperplasia.
| Disease | History and symptoms | Laboratory findings | Defective gene | ||||
|---|---|---|---|---|---|---|---|
| Blood pressure | Genitalia | Increased | Decreased | K levels | |||
| 21-hydroxylase deficiency | Classic type |
|
|
|
|
| |
| Non-classic type |
|
|
response to ACTH |
|
| ||
| 17-α hydroxylase deficiency |
|
|
|
| |||
| 11-β hydroxylase deficiency |
|
|
|
|
| ||
| 3β-Hydroxysteroid Dehydrogenase |
|
||||||
| Cytochrome P450-oxidoreductase (POR) deficiency (ORD) | |||||||
| Congenital lipoid adrenal hyperplasia | |||||||
| Cholesterol side-chain cleavage enzyme deficiency | |||||||
Differential Diagnosis
Congenital adrenal hyperplasia must be differentiated from diseases that cause ambiguous genitalia:[1][2]
| Disease name | Laboratory tests | Important clinical findings | |
|---|---|---|---|
| Increased | Decreased | ||
| Classic type of 21-hydroxylase deficiency |
|
| |
| 11-β hydroxylase deficiency |
|
| |
| 17-α hydroxylase deficiency |
| ||
| 3β-Hydroxysteroid Dehydrogenase |
| ||
| Gestational hyperandrogenism |
|
| |
congenital adrenal hyperplasia must be differentiated from diseases that cause virilization and hirsutism in female:[3][2][4]
| Disease name | Steroid status | Other laboratory | Important clinical findings |
|---|---|---|---|
| Non-classic type of 21-hydroxylase deficiency | Increased:
response to ACTH |
|
|
| 11-β hydroxylase deficiency | Increased:
Decreased: |
|
|
| 3β-Hydroxysteroid Dehydrogenase | Increased:
Decreased: |
|
|
| Polycystic ovary syndrome |
|
|
|
| Adrenal tumors |
|
|
|
| Ovarian virilizing tumor |
|
|
|
| Cushing's syndrome |
|
||
| Hyperprolactinemia |
|
|
Screening
According to Endocrine Society Clinical Practice Guideline, screening for 21-hydroxylase deficiency by measuring 17a-hydroxyprogesterone is recommended for all newborns.
- Blood sample on filter paper should be obtained from heel puncture preferably between two and four days after birth.
- Screening programs should be done using a two-tier protocol (initial immunoassay with further evaluation of positive tests by liquid chromatography/tandem mass spectrometry.
- Most affected neonates have concentrations greater than 3500 ng/dL (105 nmol/L).[5][6]
Genetic counseling
The Endocrine Society's Clinical Practice Guideline recommends that genetic counseling be provided for individuals who are planning to conceive, and there is a family history of 21-hydroxylase deficiency.[6]
References
- ↑ Hughes IA, Nihoul-Fékété C, Thomas B, Cohen-Kettenis PT (2007). "Consequences of the ESPE/LWPES guidelines for diagnosis and treatment of disorders of sex development". Best Pract. Res. Clin. Endocrinol. Metab. 21 (3): 351–65. doi:10.1016/j.beem.2007.06.003. PMID 17875484.
- ↑ 2.0 2.1 White PC, Speiser PW (2000). "Congenital adrenal hyperplasia due to 21-hydroxylase deficiency". Endocr. Rev. 21 (3): 245–91. doi:10.1210/edrv.21.3.0398. PMID 10857554.
- ↑ Hohl A, Ronsoni MF, Oliveira M (2014). "Hirsutism: diagnosis and treatment". Arq Bras Endocrinol Metabol. 58 (2): 97–107. PMID 24830586. Vancouver style error: initials (help)
- ↑ Melmed, Shlomo (2016). Williams textbook of endocrinology. Philadelphia, PA: Elsevier. ISBN 978-0323297387.=
- ↑ Gonzalez RR, Mäentausta O, Solyom J, Vihko R (1990). "Direct solid-phase time-resolved fluoroimmunoassay of 17 alpha-hydroxyprogesterone in serum and dried blood spots on filter paper". Clin. Chem. 36 (9): 1667–72. PMID 2208708.
- ↑ 6.0 6.1 Speiser PW, Azziz R, Baskin LS, Ghizzoni L, Hensle TW, Merke DP, Meyer-Bahlburg HF, Miller WL, Montori VM, Oberfield SE, Ritzen M, White PC (2010). "Congenital adrenal hyperplasia due to steroid 21-hydroxylase deficiency: an Endocrine Society clinical practice guideline". J. Clin. Endocrinol. Metab. 95 (9): 4133–60. doi:10.1210/jc.2009-2631. PMC 2936060. PMID 20823466.