Urethritis laboratory findings
Jump to navigation
Jump to search
|
Urethritis Microchapters | |
|
Diagnosis | |
|
Treatment | |
|
Case Studies | |
|
Urethritis laboratory findings On the Web | |
|
American Roentgen Ray Society Images of Urethritis laboratory findings | |
|
Risk calculators and risk factors for Urethritis laboratory findings | |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Seyedmahdi Pahlavani, M.D. [2]
Overview
Gram stain of urethral discharge and nucleic acid amplification tests (NAATs) are the main laboratory investigations in an appropriate clinical setting.
Laboratory Findings
- Urethritis is considered on the basis of any of the following:[1][2][3]
- Mucoid, mucopurulent, or purulent discharge on examination
- Gram stain of urethral secretions demonstrating ≥2 WBC per field
- Positive leukocyte esterase test on first-void urine or microscopic examination of sediment from a spun first-void urine demonstrating ≥10 WBC per high power field.[4][5]
- Once the diagnosis based on any of the above mentioned methods is confirmed, further testing to establish the etiology by nucleic acid amplification tests (NAATs) is recommended.[3]
- Nucleic acid amplification tests (NAATs) are recommended if the diagnostic criteria have not been met and the clinical suspicion for urethritis is high.[3]
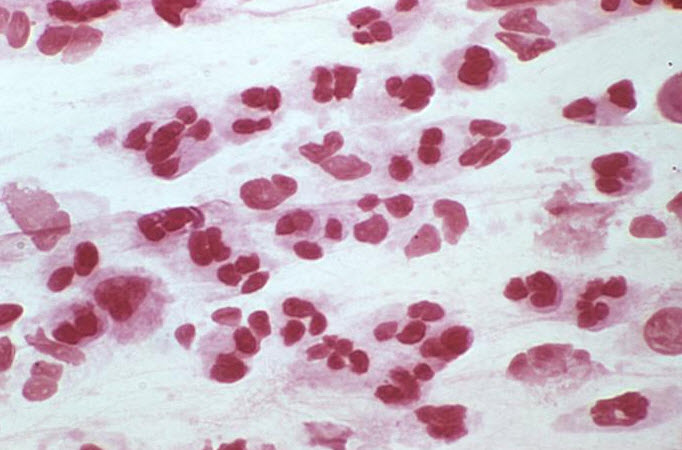
- The presence of PMNs without any visible organisms is consistent with nongonococcal urethritis.[1]
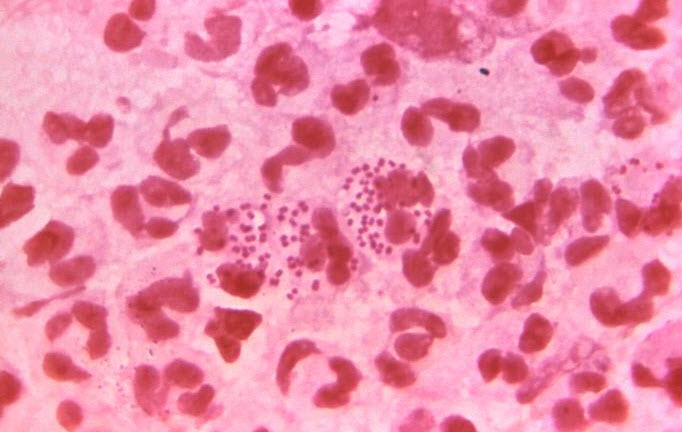
- Presumed gonococcal infection is established by documenting the presence of WBC containing intracellular purple diplococci in gram stain.[6]
-
Gram stain of urethral discharge. Note the numerous PMNs without intracellular bacteria in nongonococcal urethritis.
-
Gram stain of urethral discharge. Note the numerous PMNs with intracellular diplococcal bacteria in gonococcal urethritis.
Nucleic Acid Amplification Tests (NAAT)
These tests are more sensitive than cultures and retain high specificity. NAATs have replaced culture as the test of choice for identification of Chlamydia trachomatis, Neisseria gonorrhoeae, and Mycoplasma genitalium.[7][8]
References
- ↑ 1.0 1.1 Jacobs NF, Kraus SJ (1975). "Gonococcal and nongonococcal urethritis in men. Clinical and laboratory differentiation". Ann. Intern. Med. 82 (1): 7–12. PMID 67816.
- ↑ Bennett, John (2015). Mandell, Douglas, and Bennett's principles and practice of infectious diseases. Philadelphia, PA: Elsevier/Saunders. ISBN 9781455748013.
- ↑ 3.0 3.1 3.2 Workowski KA, Bolan GA (2015). "Sexually transmitted diseases treatment guidelines, 2015". MMWR Recomm Rep. 64 (RR-03): 1–137. PMID 26042815.
- ↑ Horner PJ, Taylor-Robinson D (2007). "Is there a role for leucocyte esterase testing in non-invasive screening using nucleic acid amplification tests of asymptomatic men?". Int J STD AIDS. 18 (2): 73–4. doi:10.1258/095646207779949718. PMID 17331273.
- ↑ Chernesky M, Jang D, Chong S, Sellors J, Mahony J (2003). "Impact of urine collection order on the ability of assays to identify Chlamydia trachomatis infections in men". Sex Transm Dis. 30 (4): 345–7. PMID 12671557.
- ↑ Bennett, John (2015). Mandell, Douglas, and Bennett's principles and practice of infectious diseases. Philadelphia, PA: Elsevier/Saunders. ISBN 9781455748013.
- ↑ Puolakkainen M, Hiltunen-Back E, Reunala T, Suhonen S, Lähteenmäki P, Lehtinen M, Paavonen J (1998). "Comparison of performances of two commercially available tests, a PCR assay and a ligase chain reaction test, in detection of urogenital Chlamydia trachomatis infection". J. Clin. Microbiol. 36 (6): 1489–93. PMC 104866. PMID 9620366.
- ↑ Jaschek G, Gaydos CA, Welsh LE, Quinn TC (1993). "Direct detection of Chlamydia trachomatis in urine specimens from symptomatic and asymptomatic men by using a rapid polymerase chain reaction assay". J. Clin. Microbiol. 31 (5): 1209–12. PMC 262905. PMID 8501220.