Travoprost
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Vignesh Ponnusamy, M.B.B.S. [2]
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Travoprost is a prostaglandin analog that is FDA approved for the {{{indicationType}}} of open-angle glaucoma or ocular hypertension. Common adverse reactions include conjunctival hyperemia.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Ocular Hypertension
- TRAVATAN Z® (travoprost ophthalmic solution) 0.004% is indicated for the reduction of elevated intraocular pressure in patients with open-angle glaucoma or ocular hypertension.
- The recommended dosage is one drop in the affected eye(s) once daily in the evening. TRAVATAN Z® (travoprost ophthalmic solution) should not be administered more than once daily since it has been shown that more frequent administration of prostaglandin analogs may decrease the intraocular pressure lowering effect.
- Reduction of the intraocular pressure starts approximately 2 hours after the first administration with maximum effect reached after 12 hours.
- TRAVATAN Z® may be used concomitantly with other topical ophthalmic drug products to lower intraocular pressure. If more than one topical ophthalmic drug is being used, the drugs should be administered at least five (5) minutes apart.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Travoprost in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Travoprost in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding FDA-Labeled Use of Travoprost in pediatric patients.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Travoprost in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Travoprost in pediatric patients.
Contraindications
- None
Warnings
Precautions
- Pigmentation
- Travoprost ophthalmic solution has been reported to cause changes to pigmented tissues. The most frequently reported changes have been increased pigmentation of the iris, periorbital tissue (eyelid) and eyelashes. Pigmentation is expected to increase as long as travoprost is administered. The pigmentation change is due to increased melanin content in the melanocytes rather than to an increase in the number of melanocytes. After discontinuation of travoprost, pigmentation of the iris is likely to be permanent, while pigmentation of the periorbital tissue and eyelash changes have been reported to be reversible in some patients. Patients who receive treatment should be informed of the possibility of increased pigmentation. The long term effects of increased pigmentation are not known.
- Iris color change may not be noticeable for several months to years. Typically, the brown pigmentation around the pupil spreads concentrically towards the periphery of the iris and the entire iris or parts of the iris become more brownish. Neither nevi nor freckles of the iris appear to be affected by treatment. While treatment with TRAVATAN Z® (travoprost ophthalmic solution) 0.004% can be continued in patients who develop noticeably increased iris pigmentation, these patients should be examined regularly.
- Eyelash Changes
- TRAVATAN Z® may gradually change eyelashes and vellus hair in the treated eye. These changes include increased length, thickness, and number of lashes. Eyelash changes are usually reversible upon discontinuation of treatment.
- Intraocular Inflammation
- TRAVATAN Z® should be used with caution in patients with active intraocular inflammation (e.g., uveitis) because the inflammation may be exacerbated.
- Macular Edema
- Macular edema, including cystoid macular edema, has been reported during treatment with travoprost ophthalmic solution. TRAVATAN Z® should be used with caution in aphakic patients, in pseudophakic patients with a torn posterior lens capsule, or in patients with known risk factors for macular edema.
- Angle-closure, Inflammatory or Neovascular Glaucoma
- TRAVATAN Z® has not been evaluated for the treatment of angle-closure, inflammatory or neovascular glaucoma.
- Bacterial Keratitis
- There have been reports of bacterial keratitis associated with the use of multiple-dose containers of topical ophthalmic products. These containers had been inadvertently contaminated by patients who, in most cases, had a concurrent corneal disease or a disruption of the ocular epithelial surface.
- Use with Contact Lenses
- Contact lenses should be removed prior to instillation of TRAVATAN Z® and may be reinserted 15 minutes following its administration.
Adverse Reactions
Clinical Trials Experience
- Because clinical studies are conducted under widely varying conditions, adverse reaction rates observed in the clinical studies of a drug cannot be directly compared to rates in the clinical studies of another drug and may not reflect the rates observed in practice.
- The most common adverse reaction observed in controlled clinical studies with TRAVATAN® (travoprost ophthalmic solution) 0.004% and TRAVATAN Z® (travoprost ophthalmic solution) 0.004% was ocular hyperemia which was reported in 30 to 50% of patients. Up to 3% of patients discontinued therapy due to conjunctival hyperemia. Ocular adverse reactions reported at an incidence of 5 to 10% in these clinical studies included decreased visual acuity, eye discomfort, foreign body sensation, pain and pruritus.
- Ocular adverse reactions reported at an incidence of 1 to 4% in clinical studies with TRAVATAN® or TRAVATAN Z® included abnormal vision, blepharitis, blurred vision, cataract, conjunctivitis, corneal staining, dry eye, iris discoloration, keratitis, lid margin crusting, ocular inflammation, photophobia, subconjunctival hemorrhage and tearing.
- Nonocular adverse reactions reported at an incidence of 1 to 5% in these clinical studies were allergy, angina pectoris, anxiety, arthritis, back pain, bradycardia, bronchitis, chest pain, cold/flu syndrome, depression, dyspepsia, gastrointestinal disorder, headache, hypercholesterolemia, hypertension, hypotension, infection, pain, prostate disorder, sinusitis, urinary incontinence and urinary tract infections.
Postmarketing Experience
- In postmarketing use with prostaglandin analogs, periorbital and lid changes including deepening of the eyelid sulcus have been observed.
Drug Interactions
There is limited information regarding Travoprost Drug Interactions in the drug label.
Use in Specific Populations
Pregnancy
- Pregnancy Category C
- Teratogenic effects: Travoprost was teratogenic in rats, at an intravenous (IV) dose up to 10 mcg/kg/day (250 times the maximal recommended human ocular dose (MRHOD), evidenced by an increase in the incidence of skeletal malformations as well as external and visceral malformations, such as fused sternebrae, domed head and hydrocephaly. Travoprost was not teratogenic in rats at IV doses up to 3 mcg/kg/day (75 times the MRHOD), or in mice at subcutaneous doses up to 1 mcg/kg/day (25 times the MRHOD). Travoprost produced an increase in post-implantation losses and a decrease in fetal viability in rats at IV doses > 3 mcg/kg/day (75 times the MRHOD) and in mice at subcutaneous doses > 0.3 mcg/kg/day (7.5 times the MRHOD).
- In the offspring of female rats that received travoprost subcutaneously from Day 7 of pregnancy to lactation Day 21 at doses of = 0.12 mcg/kg/day (3 times the MRHOD), the incidence of postnatal mortality was increased, and neonatal body weight gain was decreased. Neonatal development was also affected, evidenced by delayed eye opening, pinna detachment and preputial separation, and by decreased motor activity.
- There are no adequate and well-controlled studies of TRAVATAN Z® (travoprost ophthalmic solution) 0.004% administration in pregnant women. Because animal reproductive studies are not always predictive of human response, TRAVATAN Z® should be administered during pregnancy only if the potential benefit justifies the potential risk to the fetus.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Travoprost in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Travoprost during labor and delivery.
Nursing Mothers
- A study in lactating rats demonstrated that radiolabeled travoprost and/or its metabolites were excreted in milk. It is not known whether this drug or its metabolites are excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when TRAVATAN Z® is administered to a nursing woman.
Pediatric Use
- Use in pediatric patients below the age of 16 years is not recommended because of potential safety concerns related to increased pigmentation following long-term chronic use.
Geriatic Use
- No overall clinical differences in safety or effectiveness have been observed between elderly and other adult patients.
Gender
There is no FDA guidance on the use of Travoprost with respect to specific gender populations.
Race
There is no FDA guidance on the use of Travoprost with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Travoprost in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Travoprost in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Travoprost in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Travoprost in patients who are immunocompromised.
Administration and Monitoring
Administration
- Topical
Monitoring
There is limited information regarding Monitoring of Travoprost in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Travoprost in the drug label.
Overdosage
Chronic Overdose
There is limited information regarding Chronic Overdose of Travoprost in the drug label.
Pharmacology
| |
Travoprost
| |
| Systematic (IUPAC) name | |
| propan-2-yl 7-[3,5-dihydroxy-2-[3-hydroxy-4-[3-(trifluoromethyl) phenoxy]-but-1-enyl]-cyclopentyl]hept-5-enoate | |
| Identifiers | |
| CAS number | |
| ATC code | S01 |
| PubChem | |
| DrugBank | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 500.548 g/mol |
| SMILES | & |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
C US |
| Legal status |
Rx only (US) |
| Routes | Topical (eye drops) |
Mechanism of Action
- Travoprost free acid, a prostaglandin analog is a selective FP prostanoid receptor agonist which is believed to reduce intraocular pressure by increasing uveoscleral outflow. The exact mechanism of action is unknown at this time.
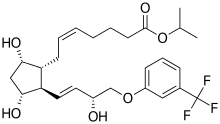
Structure
- Travoprost is a synthetic prostaglandin F analogue. Its chemical name is [1R-[1α(Z),2β(1E,3R*),3α,5α]]-7-[3,5-Dihydroxy-2-[3-hydroxy-4-[3-(trifluoromethyl) phenoxy]-1-butenyl]cyclopentyl]-5-heptenoic acid, 1-methylethylester. It has a molecular formula of C26H35F3O6 and a molecular weight of 500.55. The chemical structure of travoprost is:
- Travoprost is a clear, colorless to slightly yellow oil that is very soluble in acetonitrile, methanol, octanol, and chloroform. It is practically insoluble in water.
- TRAVATAN Z® (travoprost ophthalmic solution) 0.004% is supplied as sterile, buffered aqueous solution of travoprost with a pH of approximately 5.7 and an osmolality of approximately 290 mOsmol/kg.
- TRAVATAN Z® contains Active: travoprost 0.04 mg/mL; Inactives: polyoxyl 40 hydrogenated castor oil, sofZia® (boric acid, propylene glycol, sorbitol, zinc chloride), sodium hydroxide and/or hydrochloric acid (to adjust pH) and purified water, USP. Preserved in the bottle with an ionic buffered system, sofZia®.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Travoprost in the drug label.
Pharmacokinetics
- Travoprost is absorbed through the cornea and is hydrolyzed to the active free acid. Data from four multiple dose pharmacokinetic studies (totaling 107 subjects) have shown that plasma concentrations of the free acid are below 0.01 ng/ml (the quantitation limit of the assay) in two-thirds of the subjects. In those individuals with quantifiable plasma concentrations (N=38), the mean plasma Cmax was 0.018 ± 0.007 ng/ml (ranged 0.01 to 0.052 ng/mL) and was reached within 30 minutes. From these studies, travoprost is estimated to have a plasma half-life of 45 minutes. There was no difference in plasma concentrations between Days 1 and 7, indicating steady-state was reached early and that there was no significant accumulation.
- Travoprost, an isopropyl ester prodrug, is hydrolyzed by esterases in the cornea to its biologically active free acid. Systemically, travoprost free acid is metabolized to inactive metabolites via beta-oxidation of the α(carboxylic acid) chain to give the 1,2-dinor and 1,2,3,4-tetranor analogs, via oxidation of the 15-hydroxyl moiety, as well as via reduction of the 13,14 double bond.
- The elimination of travoprost free acid from plasma was rapid and levels were generally below the limit of quantification within one hour after dosing. The terminal elimination half-life of travoprost free acid was estimated from fourteen subjects and ranged from 17 minutes to 86 minutes with the mean half-life of 45 minutes. Less than 2% of the topical ocular dose of travoprost was excreted in the urine within 4 hours as the travoprost free acid.
Nonclinical Toxicology
- Two-year carcinogenicity studies in mice and rats at subcutaneous doses of 10, 30, or 100 mcg/kg/day did not show any evidence of carcinogenic potential. However, at 100 mcg/kg/day, male rats were only treated for 82 weeks, and the maximum tolerated dose (MTD) was not reached in the mouse study. The high dose (100 mcg/kg) corresponds to exposure levels over 400 times the human exposure at the maximum recommended human ocular dose (MRHOD) of 0.04 mcg/kg, based on plasma active drug levels.
- Travoprost was not mutagenic in the Ames test, mouse micronucleus test or rat chromosome aberration assay. A slight increase in the mutant frequency was observed in one of two mouse lymphoma assays in the presence of rat S-9 activation enzymes.
- Travoprost did not affect mating or fertility indices in male or female rats at subcutaneous doses up to 10 mcg/kg/day [250 times the maximum recommended human ocular dose of 0.04 mcg/kg/day on a mcg/kg basis (MRHOD)]. At 10 mcg/kg/day, the mean number of corpora lutea was reduced, and the post-implantation losses were increased. These effects were not observed at 3 mcg/kg/day (75 times the MRHOD).
Clinical Studies
- In clinical studies, patients with open-angle glaucoma or ocular hypertension and baseline pressure of 25-27 mmHg who were treated with TRAVATAN® (travoprost ophthalmic solution) 0.004% or TRAVATAN Z® (travoprost ophthalmic solution) 0.004% dosed once-daily in the evening demonstrated 7-8 mmHg reductions in intraocular pressure. In subgroup analyses of these studies, mean IOP reduction in black patients was up to 1.8 mmHg greater than in non-black patients. It is not known at this time whether this difference is attributed to race or to heavily pigmented irides.
- In a multi-center, randomized, controlled trial, patients with mean baseline intraocular pressure of 24-26 mmHg on TIMOPTIC* 0.5% BID who were treated with TRAVATAN® (travoprost ophthalmic solution) 0.004% dosed QD adjunctively to TIMOPTIC* 0.5% BID demonstrated 6-7 mmHg reductions in intraocular pressure.
How Supplied
- TRAVATAN Z® (travoprost ophthalmic solution) 0.004% is a sterile, isotonic, buffered, preserved, aqueous solution of travoprost (0.04 mg/mL) supplied in Alcon's oval DROP-TAINER® package system.
- TRAVATAN Z® is supplied as a 2.5 mL solution in a 4 mL and a 5 mL solution in a 7.5 mL natural polypropylene dispenser bottle with a natural polypropylene dropper tip and a turquoise polypropylene or high density polyethylene overcap. Tamper evidence is provided with a shrink band around the closure and neck area of the package.
- 2.5 mL fill NDC 0065-0260-25
- 5 mL fill NDC 0065-0260-05
- Storage: Store at 2° - 25°C (36° - 77°F).
Storage
There is limited information regarding Travoprost Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Travoprost |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Travoprost |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Potential for Pigmentation
- Patients should be advised about the potential for increased brown pigmentation of the iris, which may be permanent. Patients should also be informed about the possibility of eyelid skin darkening, which may be reversible after discontinuation of TRAVATAN Z® (travoprost ophthalmic solution) 0.004%.
- Potential for Eyelash Changes
- Patients should also be informed of the possibility of eyelash and vellus hair changes in the treated eye during treatment with TRAVATAN Z®. These changes may result in a disparity between eyes in length, thickness, pigmentation, number of eyelashes or vellus hairs, and/or direction of eyelash growth. Eyelash changes are usually reversible upon discontinuation of treatment.
- Handling the Container
- Patients should be instructed to avoid allowing the tip of the dispensing container to contact the eye, surrounding structures, fingers, or any other surface in order to avoid contamination of the solution by common bacteria known to cause ocular infections. Serious damage to the eye and subsequent loss of vision may result from using contaminated solutions.
- When to Seek Physician Advice
- Patients should also be advised that if they develop an intercurrent ocular condition (e.g., trauma or infection), have ocular surgery, or develop any ocular reactions, particularly conjunctivitis and eyelid reactions, they should immediately seek their physician's advice concerning the continued use of TRAVATAN Z®.
- Use with Contact Lenses
- Contact lenses should be removed prior to instillation of TRAVATAN Z® and may be reinserted 15 minutes following its administration.
- Use with Other Ophthalmic Drugs
- If more than one topical ophthalmic drug is being used, the drugs should be administered at least five (5) minutes between applications.
Precautions with Alcohol
- Alcohol-Travoprost interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- TRAVATAN Z®[1]
Look-Alike Drug Names
There is limited information regarding Travoprost Look-Alike Drug Names in the drug label.
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
{{#subobject:
|Page Name=Travoprost |Pill Name=No image.jpg |Drug Name= |Pill Ingred=|+sep=; |Pill Imprint= |Pill Dosage= |Pill Color=|+sep=; |Pill Shape= |Pill Size (mm)= |Pill Scoring= |Pill Image= |Drug Author= |NDC=
}}
{{#subobject:
|Label Page=Travoprost |Label Name=Travoprost02.png
}}
{{#subobject:
|Label Page=Travoprost |Label Name=Travoprost03.png
}}
{{#subobject:
|Label Page=Travoprost |Label Name=Travoprost04.png
}}
