Supercritical fluid
A supercritical fluid is any substance at a temperature and pressure above its thermodynamic critical point. It can diffuse through solids like a gas, and dissolve materials like a liquid. Additionally, close to the critical point, small changes in pressure or temperature result in large changes in density, allowing many properties to be "tuned". Supercritical fluids are suitable as a substitute for organic solvents in a range of industrial and laboratory processes. Carbon dioxide and water are the most commonly used supercritical fluids, being used for decaffeination and power generation respectively.
Properties
In general terms, supercritical fluids have properties between those of a gas and a liquid. In Table 1, the critical properties are shown for some components, which are commonly used as supercritical fluids.
| Solvent | Molecular weight | Critical temperature | Critical pressure | Critical density |
|---|---|---|---|---|
| g/mol | K | MPa (atm) | g/cm3 | |
| Carbon dioxide (CO2) | 44.01 | 304.1 | 7.38 (72.8) | 0.469 |
| Water (H2O) | 18.02 | 647.3 | 22.12 (218.3) | 0.348 |
| Methane (CH4) | 16.04 | 190.4 | 4.60 (45.4) | 0.162 |
| Ethane (C2H6) | 30.07 | 305.3 | 4.87 (48.1) | 0.203 |
| Propane (C3H8) | 44.09 | 369.8 | 4.25 (41.9) | 0.217 |
| Ethylene (C2H4) | 28.05 | 282.4 | 5.04 (49.7) | 0.215 |
| Propylene (C3H6) | 42.08 | 364.9 | 4.60 (45.4) | 0.232 |
| Methanol (CH3OH) | 32.04 | 512.6 | 8.09 (79.8) | 0.272 |
| Ethanol (C2H5OH) | 46.07 | 513.9 | 6.14 (60.6) | 0.276 |
| Acetone (C3H6O) | 58.08 | 508.1 | 4.70 (46.4) | 0.278 |
Table 2 shows density, diffusivity and viscosity for typical liquids, gasses and supercritical fluids.
| Density (kg/m3) | Viscosity (cP) | Diffusivity (mm2 / s) | |
|---|---|---|---|
| Gases | 1 | 0.01 | 1-10 |
| Supercritical Fluids | 100-1000 | 0.05-0.1 | 0.01-0.1 |
| Liquids | 1000 | 0.5-1.0 | 0.001 |
In addition, there is no surface tension in a supercritical fluid, as there is no liquid / gas phase boundary. By changing the pressure and temperature of the fluid, the properties can be “tuned” to be more liquid or more gas like. One of the most important properties is the solubility of material in the fluid. Solubility in a supercritical fluid tends to increase with density of the fluid (at constant temperature). Since density increases with pressure, then solubility also tends to increase with pressure. The relationship with temperature is a little more complicated. At constant density, solubility will increase with temperature. However, close to the critical point, the density can drop sharply with a slight increase in temperature. Therefore, close to the critical temperature, solubility often drops with increasing temperature, then rises again[2].
All supercritical fluids are completely miscible with each other so for a mixture a single phase can be guaranteed if the critical point of the mixture is exceeded. The critical point of a binary mixture can be estimated as the arithmetic mean of the critical temperatures and pressures of the two components,
- Tc(mix) = (mole fraction A) x TcA + (mole fraction B) x TcB.
For greater accuracy, the critical point can be calculated using equations of state, such as the Peng Robinson, or group contribution methods. Other properties, such as density, can also be calculated using equations of state.[3].
Phase diagram
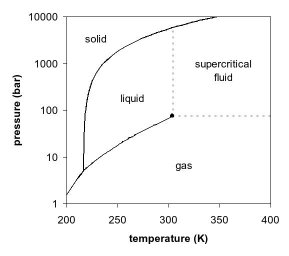
Figures 1 and 2 show projections of a phase diagram. In the pressure-temperature phase diagram (Fig. 1) the boiling separates the gas and liquid region and ends in the critical point, where the liquid and gas phases disappear to become a single supercritical phase. This can be observed in the density-pressure phase diagram for carbon dioxide, as shown in Figure 2. At well below the critical temperature, e.g. 280K, as the pressure increases, the gas compresses and eventually (at just over 40 bar) condenses into a much denser liquid, resulting in the discontinuity in the line (vertical dotted line). The system consists of 2 phases in equilibrium, a dense liquid and a low density gas. As the critical temperature is approached (300K), the density of the gas at equilibrium becomes denser, and that of the liquid lower. At the critical point, (304.1 K and 7.38 MPa (73.8 bar)). there is no difference in density, and the 2 phases become one fluid phase. Thus, above the critical temperature a gas cannot be liquefied by pressure. At slightly above the critical temperature (310K), in the vicinity of the critical pressure, the line is almost vertical. A small increase in pressure causes a large increase in the density of the supercritical phase. Many other physical properties also show large gradients with pressure near the critical point, e.g. viscosity, the relative permittivity and the solvent strength, which are all closely related to the density. At higher temperatures, the fluid starts to behave like a gas, as can be seen in Figure 2. For carbon dioxide at 400 K, the density increases almost linearly with pressure.
Many pressurised gases are actually supercritical fluids. For example, nitrogen has a critical point of 126.2K (- 147 °C) and 3.4 MPa (34 bar). Therefore, nitrogen in a gas cylinder above this pressure (or compressed air) is actually a supercritical fluid. These are more often known as permanent gases. At room temperature, they are well above their critical temperature, and therefore behave as a gas, similar to CO2 at 400K above. However, they cannot be liquified by pressure unless cooled below their critical temperature.
Natural occurrence
Submarine volcanoes
Submarine volcanoes are common features on the ocean floor. Some are active and, in shallow water, disclose their presence by blasting steam and rocky debris high above the surface of the sea. Many others lie at such great depths that the tremendous pressure from the weight of the water above them prevents the explosive release of steam and gases and this heats the water to over 375 degrees C turning the water in the hottest parts of the vents into a supercritical fluid since the pressure at this depth of over 3km is over 300 atmospheres, well above the 218 atmospheres required.
Venus
The atmosphere of Venus is 96.5% carbon dioxide and 3.5% nitrogen. The surface pressure is 9.3 MPa (93 bar) and the surface temperature is 735 K, above the critical points of both major constituents and making the surface atmosphere a supercritical fluid.
Applications
Supercritical fluid extraction
The advantages of supercritical fluid extraction (compared with liquid extraction) are that it is relatively rapid because of the low viscosities and high diffusivities associated with supercritical fluids. The extraction can be selective to some extent by controlling the density of the medium and the extracted material is easily recovered by simply depressurising, allowing the supercritical fluid to return to gas phase and evaporate leaving no or little solvent residues. Carbon dioxide is the most common supercritical solvent. It is used on a large scale for the decaffeination of green coffee beans, the extraction of hops for beer production,[4] and the production of essential oils and pharmaceutical products from plants. Few laboratory test methods include the use of supercritical fluid extraction as an extraction method instead of using traditional solvents[5][6][7].
Dry Cleaning
Uses supercritical carbon dioxide instead of PERC (perchloroethylene) or other undesirable solvents for dry cleaning. Detergents which are soluble in carbon dioxide improve the solvating power of the solvent.[8]
Supercritical fluid chromatography
Supercritical fluid chromatography (SFC) can be used on an analytical scale, where it combines many of the advantages of HPLC and GC. It can be used with non-volatile and thermally labile analytes (unlike GC) and can be used with the universal FID detector (unlike HPLC), as well as producing narrower peaks due to rapid diffusion. In practice, the advantages offered by SFC have not been sufficient to displace the widely used HPLC and GC, except in a few cases such as chiral separations and analysis of high molecular weight hydrocarbons.[9] For manufacturing, efficient preparative simulated moving bed units are available.[10] The purity of the final products is very high, but the cost makes it suitable only for very high value materials such as pharmaceuticals.
Chemical Reactions
Changing the conditions of the reaction solvent can allow separation of phases for product removal, or single phase for reaction. Rapid diffusion accelerates diffusion controlled reactions. Temperature and pressure can tune the reaction down preferred pathways, e.g . to improve yield of a particular chiral isomer.[11] There are also significant environmental benefits over conventional organic solvents.
Impregnation and dyeing
Impregnation is essentially the converse of extraction. A substance is dissolved in the supercritical fluid, the solution flowed past a solid substrate, and is deposited on or dissolves in the substrate. Dyeing, which is readily carried out on polymer fibres such as polyester using disperse (non-ionic) dyes, is a special case of this. Carbon dioxide also dissolves in many polymers, considerably swelling and plasticising them and further accelerating the diffusion process.
Nano and Micro Particle Formation
The formation of small particles of a substance with a narrow size distribution is an important process in the pharmaceutical and other industries. Supercritical fluids provide a number of ways of achieving this by rapidly exceding the saturation point of a solute by dilution, depressurisation or a combination of these. These processes occur faster in supercritical fluids than in liquids, promoting nucleation or spinodal decomposition over crystal growth and yielding very small and regularly sized particles.[12]
Supercritical Drying
Supercritical drying is a method of removing solvent without surface tension effects. As a liquid dries, the surface tension drags on small structures within a solid, causing distortion and shrinkage. Under supercritical conditions there is no surface tension, and the supercritical fluid can be removed without distortion. Supercritical drying is used for manufacture of aerogels and drying of delicate materials such as archeological samples and biological samples for electron microscopy.
Supercritical water oxidation
Supercritical water oxidation uses supercritical water to oxidise hazardous waste, eliminating production of toxic combustion products which burning can produce.
Supercritical water power generation
The efficiency of a heat engine is ultimately dependent on the temperature difference between heat source and sink (carnot cycle). To improve efficiency of power stations the operating temperature must be raised. Using water as the coolant, this takes it into supercritical conditions. Efficiencies can be raised from about 39% for sub critical operation to about 45% using current technology.[13] Supercritical water reactors (SCWRs) are promising advanced nuclear systems that offer similar thermal efficiency gains. Carbon dioxide can also be used in supercritical cycle nuclear plants, with similar efficiency gains.[14]
Biodiesel production
Conversion of vegetable oil to biodiesel is via a transesterification reaction, where the triglyceride is converted to the methyl ester plus glycerol. This is usually done using methanol and caustic or acid catalysts, but can be achieved using supercritical methanol without a catalyst. This has the advantage of allowing a greater range and water content of feedstocks (particularly used cooking oil), the product does not need to be washed to remove catalyst, and is easier to design as a continuous process.[15]
Supramics
Supramics, environmentally beneficial, low-cost substitutes for rigid thermoplastic and fired ceramic, are made using supercritical carbon dioxide as a chemical reagent. The supercritical carbon dioxide in these processes is reacted with the alkaline components of fully hardened hydraulic cement or gypsum plaster to form various carbonates. The sole by-product is ultra-pure water. Because supramics consume and sequester carbon as stable compounds in useful products, they may serve to reduce carbon that would otherwise be released into the environment.
Carbon capture and storage and Enhanced oil recovery
Supercritical carbon dioxide is used to enhance oil recovery in mature oil fields. At the same time, there is the possibility of using "clean coal technology" to combine enhanced recovery methods with carbon sequestation. The CO2 is separated from other flue gases either pre or post combustion, compressed to the supercritical state, and injected into geological storage, possibly into existing oil fields to improve yields. Currently, only schemes isolating fossil CO2 from natural gas actually use carbon storage, (e.g. Sleipner gas field) [16], but there are many plans for future CCS schemes involving pre- or post- combustion CO2. [17][18][19][20] There is also the possibility to reduce the amount of CO2 in the atmosphere by using biomass to generate power and sequestering the CO2 produced.
Refrigeration
Supercritical carbon dioxide is also an important emerging natural refrigerant, being used in new, low carbon solutions for domestic heat pumps.[21] These systems are undergoing continuous development with supercritical carbon dioxide heat pumps already being successfully marketed in Asia. The "EcoCute" systems from Japan, developed by consortium of companies including Mitsubishi, develop high temperature domestic water with small inputs of electric power by moving heat into the system from their surroundings. Their success makes a future use in other world regions possible.[22]
Supercritical Fluid Deposition
Suprercritical fluids can be used to deposit functional nanostructured films and nanometer-sized particles of metals onto surfaces. The gas-like surface tension, diffusivities, and viscosities allows access to nano pores much smaller than can be accessed by liquids, and the liquid-like solubilities allow much higher precursor concentrations than are typical in chemical vapour deposition.[23] This is crucial in developing more powerful electronic components, and metal particles deposited in this way are also powerful catalysts for chemical synthesis and electrochemical reactions.
History
In 1822, Baron Charles Cagniard de la Tour discovered the critical point of a substance in his famous cannon barrel experiments. Listening to discontinuities in the sound of a rolling flint ball in a sealed cannon filled with fluids at various temperatures, he observed the critical temperature. Above this temperature, the densities of the liquid and gas phases become equal and the distinction between them disappears, resulting in a single supercritical fluid phase.
References
- ↑ Edit Székely. "Supercritical Fluid Extraction". Budapest University of Technology and Economics. Retrieved 2007-11-20.
- ↑ "Supercritical Fluid Extraction, Density Considerations". Retrieved 2007-11-20.
- ↑ A.A. Clifford (2007-12-04). "Calculation of Thermodynamic Properties of CO2 using Peng Robinson equation of state". Critical Processes Ltd. Unknown parameter
|accsessdate=ignored (help) - ↑ "The Naked Scientist Interviews". Retrieved 2007-11-20.
- ↑ U.S.EPA Method 3560 Supercritical Fluid Extraction of Total Recoverable Hydrocarbons. http://www.epa.gov/SW-846/pdfs/3560.pdf
- ↑ U.S.EPA Method 3561 Supercritical Fluid Extraction of Polycyclic Aromatic Hydrocarbons. http://www.epa.gov/SW-846/pdfs/3561.pdf
- ↑ Use of Ozone Depleting Substances in Laboratories. TemaNord 2003:516. http://www.norden.org/pub/ebook/2003-516.pdf
- ↑ "Science News Online". Retrieved 2007-11-20.
- ↑ Bart, C. J. (2005). "Chapter 4: Separation Techniques". Additives in Polymers: industrial analysis and applications. John Wiley and Sons. p. 212. doi:10.1002/0470012064.ch4. ISBN 9780470012062.
- ↑ "Simulated Moving Bed Theory" (PDF). Retrieved 2007-11-20.
- ↑ R. Scott Oakes, Anthony A. Clifford, Keith D. Bartle, Mark Thornton Pett and Christopher M. Rayner (1999). "Sulfur oxidation in supercritical carbon dioxide: dramatic pressure dependent enhancement of diastereoselectivity for sulphoxidation of cysteine derivatives". Chemical Communications: 247–248. doi:10.1039/a809434i.
- ↑ Sang-Do Yeob and Erdogan Kirana (2005). "Formation of polymer particles with supercritical fluids: A review". The Journal of Supercritical Fluids. 34 (3): 287–308. doi:10.1016/j.supflu.2004.10.006.
- ↑ "Supercritical steam cycles for power generation applications" (PDF). Retrieved 2007-11-20.
- ↑ Dostal, M.J. Driscoll, P. Hejzlar. "A Supercritical Carbon Dioxide Cycle for Next Generation Nuclear Reactors" (PDF). MIT-ANP-TR-100. MIT-ANP-Series. Retrieved 2007-11-20.
- ↑ Kunchana Bunyakiat, Sukunya Makmee, Ruengwit Sawangkeaw, and Somkiat Ngamprasertsith (2006). "Continuous Production of Biodiesel via Transesterification from Vegetable Oils in Supercritical Methanol". Energy and Fuels. 20: 812–817. doi:10.1021/ef050329b.
- ↑ "Saline Aquifer CO2 Storage". Retrieved 2007-12-10.
- ↑ "The Hydrogen Economy: Opportunities, Costs, Barriers, and R&D Needs", p. 84 (2004)
- ↑ FutureGen Technology
- ↑ Øyvind Vessia: "Fischer- Tropsch reactor fed by syngas"
- ↑ Intergovernmental Panel on Climate Change IPCC Special Report on Carbon Dioxide Capture and Storage.
- ↑ FAQs - Supercritical CO2 in heat pumps and other applications
- ↑ Eco Cute hot water heat pumps in Japan
- ↑ Ye, Xiang-Rong (2003). "Supercritical fluid fabrication of metal nanowires and nanorods templated by multiwalled carbon nanotubes". Advanced Materials. 15 (4): 316–319. doi:10.1002/adma.200390077. Unknown parameter
|coauthors=ignored (help)
External links
- Handy calculator for density, enthalpy, entropy and other thermodynamic data of supercritical CO2
- Food Product Design
- CO2 as a natural refrigerant - FAQs
- animated presentation describing what a supercritical fluid is
Template:State of matter Template:WH Template:WS ko:초임계유체 id:Fluida superkritikal it:Stato supercritico nl:Superkritische fase sv:Superkritisk vätska