Sulfapyridine
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Micromedex Detailed Consumer Information |
| MedlinePlus | a682204 |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C11H11N3O2S |
| Molar mass | 249.29 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
|
WikiDoc Resources for Sulfapyridine |
|
Articles |
|---|
|
Most recent articles on Sulfapyridine Most cited articles on Sulfapyridine |
|
Media |
|
Powerpoint slides on Sulfapyridine |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Sulfapyridine at Clinical Trials.gov Trial results on Sulfapyridine Clinical Trials on Sulfapyridine at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Sulfapyridine NICE Guidance on Sulfapyridine
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Sulfapyridine Discussion groups on Sulfapyridine Patient Handouts on Sulfapyridine Directions to Hospitals Treating Sulfapyridine Risk calculators and risk factors for Sulfapyridine
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Sulfapyridine |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Sulfapyridine, original UK spelling sulphapyridine, is a sulfonamide antibacterial. At one time it was commonly referred to as M&B.
Sulfapyridine is no longer prescribed for treatment of infections in humans. However, it may be used to treat linear IgA disease. It is a good antibacterial drug, but its water solubility is very pH dependent. Thus there is a risk of crystallization within the bladder or urethra, which could lead to pain or blockage. As with other sulfonamides, there is a significant risk of agranulocytosis, and this, rather than the development of resistance by bacteria, is the main reason for its decline in use.
It was discovered by Lionel Whitby at the British firm May & Baker Ltd and logged in their Test Book on 2 November, 1937 under Code No M&B 693.[1] It was successfully used to treat Winston Churchill's bacterial pneumonia in 1942. In a subsequent radio broadcast he said: "This admirable M&B from which I did not suffer any inconvenience, was used at the earliest moment and, after a week's fever, the intruders were repulsed." In 1944 M&B 693 also saved Nero, the Royal Circus lion, from pneumonia. [Glasgow Evening News January 1944.]
The drug sulfasalazine is structurally one molecule of mesalamine linked to one molecule of sulfapyridine with an azo bond.
M&B 693 was one of the first generation of sulphonamide antibiotics. It has been reported as the first chemical cure for pneumonia. It could either be taken in tablet form or the powder could be placed in wounds. It was used so widely during the Second World War that May & Baker had difficulty keeping up with demand. It was later largely superseded by penicillin and other sulfonamides.
Synthesis
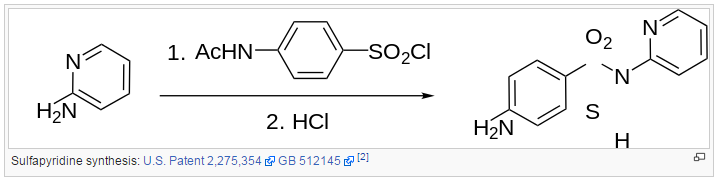
References
- ↑ Lesch, John (2007). "Chapter 7". The First Miracle Drugs (illustrated ed.). Oxford University Press. ISBN 0-19-518775-X.
|access-date=requires|url=(help)
- Pages with script errors
- Pages using citations with accessdate and no URL
- Template:drugs.com link with non-standard subpage
- Articles with changed CASNo identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Drugs with no legal status
- Drugboxes which contain changes to verified fields
- Drug
- Sulfonamide antibiotics
- Pyridines