Sulfadimidine
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
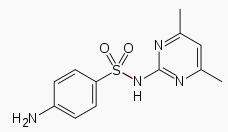
| Formula | C12H14N4O2S |
| Molar mass | 278.33 g/mol |
| 3D model (JSmol) | |
| Melting point | 176 °C (348.8 °F) |
| |
| |
| (verify) | |
|
WikiDoc Resources for Sulfadimidine |
|
Articles |
|---|
|
Most recent articles on Sulfadimidine Most cited articles on Sulfadimidine |
|
Media |
|
Powerpoint slides on Sulfadimidine |
|
Evidence Based Medicine |
|
Clinical Trials |
|
Ongoing Trials on Sulfadimidine at Clinical Trials.gov Trial results on Sulfadimidine Clinical Trials on Sulfadimidine at Google
|
|
Guidelines / Policies / Govt |
|
US National Guidelines Clearinghouse on Sulfadimidine NICE Guidance on Sulfadimidine
|
|
Books |
|
News |
|
Commentary |
|
Definitions |
|
Patient Resources / Community |
|
Patient resources on Sulfadimidine Discussion groups on Sulfadimidine Patient Handouts on Sulfadimidine Directions to Hospitals Treating Sulfadimidine Risk calculators and risk factors for Sulfadimidine
|
|
Healthcare Provider Resources |
|
Causes & Risk Factors for Sulfadimidine |
|
Continuing Medical Education (CME) |
|
International |
|
|
|
Business |
|
Experimental / Informatics |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Sulfadimidine or sulfamethazine is a sulfonamide antibacterial.
There are non-standardizeda abbreviations for it as "sulfadimidine" (abbreviated SDI[1][2] and more commonly but less reliablyb SDD[3][4]) and as "sulfamethazine" (abbreviated SMT[5][6] and more commonly but less reliablyc SMZ[7][8]). Other names include sulfadimerazine, sulfadimezine, and sulphadimethylpyrimidine.
Notes
- ^a Abbreviations are not found in the databases (such as ChemDB, ChemIDplus, PubChem), but often seen in the published literature.[citation needed]
- ^b "SDD" is not found in databases, but often seen in the published literature; it could however be confused with Tiferron/Sodium catechol sulfate (1,2-Dihydroxybenzene-3,5-disulfonic acid disodium Salt), uncommon but found officially abbreviated SDD in the ChemIDplus database.[9]
- ^c "SMZ" is not found in databases, but often seen in the published literature; it could however be confused with sulfamethoxazole, also seen abbreviated SMZ.[citation needed]
References
- ↑ Romváry, A; Simon, F (1992). "Sulfonamide residues in eggs". Acta veterinaria Hungarica. 40 (1–2): 99–106. ISSN 0236-6290. PMID 1476095.
- ↑ Reddy; Jain, S. K.; Uppal, R. P. (1988). "Pharmacokinetic studies of sulphonamides in poultry". Indian Journal of Animal Sciences.
- ↑ Kamakura, K; Hasegawa, M; Koiguchi, S; Miyata, M; Okamoto, K; Narita, M; Hirahara, Y; Yamana, T; et al. (1993). "Studies on the identification of sulfadimidine in pork by high performance liquid chromatography with photodiode array detector and gas chromatograph-mass spectrometry". Eisei Shikenjo hokoku. Bulletin of National Institute of Hygienic Sciences (111): 61–5. ISSN 0077-4715. PMID 7920569.
- ↑ Garg, SK; Ghosh, SS; Mathur, VS (Jan 1986). "Comparative pharmacokinetic study of four different sulfonamides in combination with trimethoprim in human volunteers". International journal of clinical pharmacology, therapy, and toxicology. 24 (1): 23–5. ISSN 0174-4879. PMID 3485584.
- ↑ Peña, MS; Salinas, F; Mahedero, MC; Aaron, JJ (Feb 1994). "Solvent effect on the determination of sulfamethazine by room-temperature photochemically induced fluorescence". Talanta. 41 (2): 233–6. doi:10.1016/0039-9140(94)80113-4. ISSN 0039-9140. PMID 18965913.
- ↑ Kaniou, S; Pitarakis, K; Barlagianni, I; Poulios, I (Jul 2005). "Photocatalytic oxidation of sulfamethazine". Chemosphere. 60 (3): 372–80. doi:10.1016/j.chemosphere.2004.11.069. ISSN 0045-6535. PMID 15924956.
- ↑ Calvo, R; Sarabia, S; Carlos, R; Du Souich, P (Mar 1987). "Sulfamethazine absorption and disposition: effect of surgical procedures for gastroduodenal ulcers". Biopharmaceutics & drug disposition. 8 (2): 115–24. doi:10.1002/bdd.2510080203. ISSN 0142-2782. PMID 3593892.
- ↑ De Liguoro, M; Fioretto, B; Poltronieri, C; Gallina, G (Jun 2009). "The toxicity of sulfamethazine to Daphnia magna and its additivity to other veterinary sulfonamides and trimethoprim". Chemosphere. 75 (11): 1519–24. doi:10.1016/j.chemosphere.2009.02.002. ISSN 0045-6535. PMID 19269673.
- ↑ http://chem.sis.nlm.nih.gov/chemidplus/ProxyServlet?objectHandle=Search&actionHandle=getAll3DMViewFiles&nextPage=jsp%2Fcommon%2FChemFull.jsp%3FcalledFrom%3Dlite&chemid=000149451&formatType=_3D[dead link]
Further reading
- ChemDB. "Sulfamethazine", ChemDB, National Institute of Allergy and Infectious Diseases (NIAID), National Institutes of Health (NIH)
- Template:ChemID
- PubChem. "Sulfamethazine - Substance Summary", PubChem, National Center for Biotechnology Information (NCBI), National Library of Medicine (NLM), National Institutes of Health (NIH)
- Pages with script errors
- All articles with dead external links
- Articles with dead external links from September 2009
- Articles with invalid date parameter in template
- Template:drugs.com link with non-standard subpage
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Infobox drug articles with non-default infobox title
- Drugs with no legal status
- All articles with unsourced statements
- Articles with unsourced statements from September 2009
- Drug
- Sulfonamide antibiotics
- Pyrimidines