Secnidazole
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Yashasvi Aryaputra[2];
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Secnidazole is a nitroimidazole antimicrobial that is FDA approved for the treatment of bacterial vaginosis in adult women. Common adverse reactions include vulvo-vaginal candidiasis, headache, nausea, dysgeusia, vomiting, diarrhea, abdominal pain, and vulvovaginal pruritus.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Indications:
- Secnidazole is indicated for the treatment of bacterial vaginosis in adult women.
Recommended Dosage:
- The recommended dosage of Secnidazole is a single 2-gram packet of granules taken once orally, without regard to the timing of meals.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Secnidazole Off-Label Guideline-Supported Use and Dosage (Adult) in the drug label.
Non–Guideline-Supported Use
There is limited information regarding Secnidazole Off-Label Non-Guideline-Supported Use and Dosage (Adult) in the drug label.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
There is limited information regarding Secnidazole FDA-Labeled Indications and Dosage (Pediatric) in the drug label.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Secnidazole Off-Label Guideline-Supported Use and Dosage (Pediatric) in the drug label.
Non–Guideline-Supported Use
There is limited information regarding Secnidazole Off-Label Non-Guideline-Supported Use and Dosage (Pediatric) in the drug label.
Contraindications
Hypersensitivity
- Secnidazole is contraindicated in patients who have shown hypersensitivity to Secnidazole, other ingredients of the formulation, or other nitroimidazole derivatives.
Warnings
Vulvo-Vaginal Candidiasis
- The use of Secnidazole may result in vulvo-vaginal candidiasis. In controlled clinical trials of non-pregnant women with bacterial vaginosis, vulvo-vaginal candidiasis developed in 19/197 (9.6%) of subjects who received 2 g Secnidazole and 4/136 (2.9%) subjects who received placebo. Symptomatic vulvo-vaginal candidiasis may require treatment with an antifungal agent.
Potential Risk for Carcinogenicity
- Carcinogenicity has been seen in mice and rats treated chronically with nitroimidazole derivatives which are structurally related to Secnidazole. It is unclear if the positive tumor findings in lifetime rodent studies of these nitroimidazoles indicate a risk to patients taking a single dose of Secnidazole to treat bacterial vaginosis. Avoid chronic use of Secnidazole.
Drug Resistance
- Prescribing Secnidazole in the absence of proven or strongly suspected bacterial infection or a prophylactic indication is unlikely to provide benefit to the patient and increases the risk of the development of drug-resistant bacteria.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- The safety data described below reflect exposure to 589 patients, of whom 518 received a 2 g dose of Secnidazole. Secnidazole was evaluated in three clinical trials of patients diagnosed with bacterial vaginosis: two placebo-controlled trials (Trial 1 n=215, Trial 2 n=189) and one uncontrolled safety trial (Trial 3 n=321).
- All patients received a single oral dose of study medication or placebo. Trial 1 evaluated a 1 g (this dose is not approved) dose (n=71) and a 2 g dose (n=72) of Secnidazole. Trial 2 evaluated a 2 g dose (n=125). The population was female, aged 15 to 54 years. Patients in the placebo- controlled trials were primarily Black or African American (54%) or Caucasian (41%).
- There were no deaths in the trials. Two patients in Trial 3 discontinued due to vulvovaginal candidiasis in the Secnidazole-treated arm.
Most Common Adverse Reactions
- Among 197 patients treated with a single 2 g dose of Secnidazole in the two placebo-controlled trials, Trial 1 and 2, adverse reactions were reported by approximately 29% of patients. Table 1 displays the most common adverse reactions (≥ 2 % in Secnidazole-treated patients) in these two trials.

- Among the 321 patients in an uncontrolled trial, Trial 3, adverse reactions were reported in 30% of patients. Vulvovaginal candidiasis (8.4%), nausea (5.3%), vomiting (2.5%) and dysgeusia (3.4%) were the most common adverse reactions reported in this trial.
Postmarketing Experience
- The following adverse reactions have been reported during use of other formulations of Secnidazole 2 g outside of the United States. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
- Reported adverse reactions were nausea, dysgeusia, abdominal pain, headache, and vomiting.
Drug Interactions
- Oral Contraceptives
Oral Contraceptives
- There was no clinically significant drug interaction between Secnidazole and the combination oral contraceptive, ethinyl estradiol plus norethindrone. Secnidazole can be co-administered with combination oral contraceptives (e.g., ethinyl estradiol plus norethindrone).
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA): Risk Summary
- Limited available data with Secnidazole use in pregnant women are insufficient to inform a drug associated risk of adverse developmental outcomes. In animal reproduction studies, there were no adverse developmental outcomes when Secnidazole was administered orally to pregnant rats and rabbits during organogenesis at doses up to 4 times the clinical dose.
- The estimated background risk of major birth defects and miscarriage for the indicated population is unknown. All pregnancies have a background risk of birth defect, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriages in clinically recognized pregnancies is 2 to 4% and 15 to 20%, respectively.
Data (Animal)
- In animal reproduction studies, pregnant rats were dosed orally with Secnidazole during organogenesis (gestational days 6-17) at 100, 300 and 1000 mg/kg/day, up to 4 times the clinical dose based on AUC comparisons. Animals showed no evidence of adverse developmental outcomes, but maternal toxicity (including reduced body weight gain) was observed at and above 300 mg/kg/day. In rabbits, no evidence of adverse developmental outcomes was observed when oral doses of Secnidazole were administered to dams during organogenesis (gestational days 7-20) at doses up to 100 mg/kg/day (about 0.1 times the clinical dose, based on AUC comparisons). Secnidazole was associated with maternal toxicity (reduced food consumption and markedly reduced body weight gain) in dams at 100 mg/kg/day.
- In a peri- and post-natal development study in rats, Secnidazole was administered at 30, 100 and 300 mg/kg/day from Day 6 of gestation through Day 20 of lactation. Secnidazole was not associated with any adverse effects on gestation, parturition, lactation or on subsequent development of first generation (F1) and second generation (F2) offspring at these doses, equivalent to up to 1.4 times the clinical dose based on AUC comparisons. Maternal toxicity (reduced gestational body weight gain) was evident at doses of 100 mg/kg and above (about 0.3 times the clinical dose based on AUC comparisons).
Pregnancy Category (AUS):
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Secnidazole in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Secnidazole during labor and delivery.
Nursing Mothers
Risk Summary
- There is no information on the presence of Secnidazole in human milk, the effects on the breast- fed child, or the effects on milk production. Other nitroimidazole derivatives are present in human milk. Because of the potential for serious adverse reactions, including tumorigenicity, advise patients that breastfeeding is not recommended during treatment with Secnidazole and for 96 hours (based on half-life) after administration of Secnidazole.
Clinical Considerations
- A nursing mother may choose to pump and discard her milk during treatment with Secnidazole and for 96 hours after administration of Secnidazole and feed her infant stored human milk or formula.
Pediatric Use
- The safety and effectiveness of Secnidazole in pediatric patients below the age of 18 years have not been established.
Geriatic Use
- Clinical studies with Secnidazole did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects.
Gender
There is no FDA guidance on the use of Secnidazole with respect to specific gender populations.
Race
There is no FDA guidance on the use of Secnidazole with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Secnidazole in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Secnidazole in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Secnidazole in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Secnidazole in patients who are immunocompromised.
Administration and Monitoring
Administration
Oral
- Administer without regard to meals.
- Sprinkle entire contents of packet onto applesauce, yogurt, or pudding and consume within 30 minutes without chewing or crunching the granules.
- To aid swallowing the mixture, take a glass of water after administration.
- Granules are not intended to dissolve in liquid.
Monitoring
- Improvement in the signs and symptoms of bacterial vaginosis may indicate efficacy.
IV Compatibility
There is limited information regarding the compatibility of Secnidazole and IV administrations.
Overdosage
There is limited information regarding Secnidazole overdosage. If you suspect drug poisoning or overdose, please contact the National Poison Help hotline (1-800-222-1222) immediately.
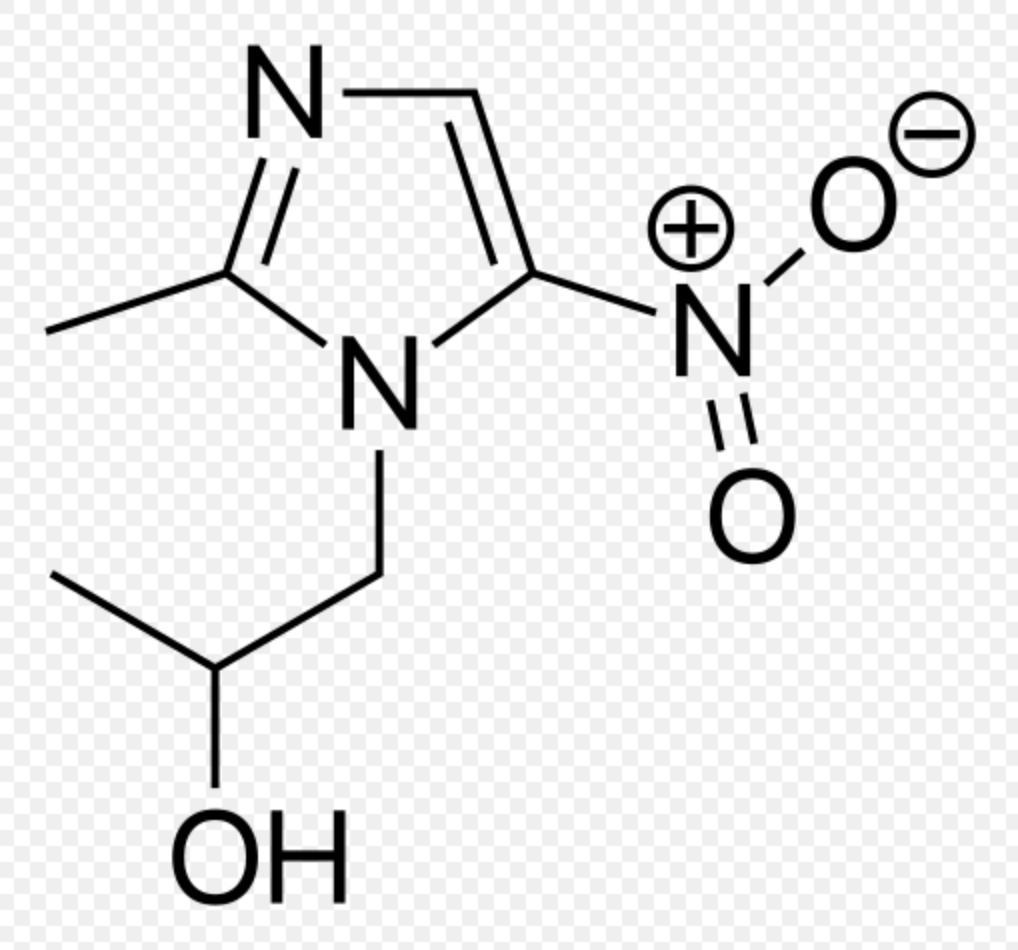
Pharmacology
| |
Secnidazole
| |
| Systematic (IUPAC) name | |
| 1-(2-Methyl-5-nitro-1H-imidazol-1-yl)propan-2-ol | |
| Identifiers | |
| CAS number | |
| ATC code | P01 |
| PubChem | |
| Chemical data | |
| Formula | Template:OrganicBox atomTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBox atomTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBoxTemplate:OrganicBox |
| Mol. mass | 185.180 g/mol |
| SMILES | & |
| Synonyms | PM 185184, RP 14539 |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status |
[[Prescription drug|Template:Unicode-only]](US) |
| Routes | Oral |
Mechanism of Action
- Secnidazole is a nitroimidazole antimicrobial drug.
Structure

Pharmacodynamics
- Secnidazole exposure-response relationships and the time course of pharmacodynamic response are unknown.
Cardiac Electrophysiology
- The effect of Secnidazole on the QTc interval was evaluated in a Phase 1 randomized, double blind, placebo- and positive-controlled four-period crossover thorough QTc study in 52 healthy adult subjects following single oral granule doses of 2 g and 6 g (3-times the recommended dose). Although there was a positive relationship of the QTc interval with Secnidazole concentrations, there was no clinically relevant increase in the QTc interval following either dose.
Pharmacokinetics
- A single oral dose of 2 g of Secnidazole in healthy adult female subjects, following an overnight fast and admixed with (4 oz) of applesauce, resulted in a mean (SD) Secnidazole peak plasma concentration (Cmax) of 45.4 (7.64) mcg/mL and mean (SD) systemic exposure (AUC0-inf) of 1331.6 (230.16) mcg•hr/mL. Median (range) time to peak concentration (Tmax) was 4.0 (3.0-4.0) hours. Following administration of the 2 g dose, mean Secnidazole plasma concentrations decreased to 22.1 mcg/mL at 24 hours, 9.2 mcg/mL at 48 hours, 3.8 mcg/mL at 72 hours, and 1.4 mcg/mL at 96 hours.
Absorption
Effect of Food
- Administration of 2 g of Secnidazole admixed with applesauce followed by ingestion of a high-fat meal (approximately 150 protein calories, 250 carbohydrate calories, and 500-600 fat calories) resulted in no significant change in the rate (Cmax) and extent (AUC) of Secnidazole exposure as compared to administration when admixed with applesauce and taken under fasted conditions. There was no effect of admixing Secnidazole with pudding and yogurt as compared to admixing with applesauce (TABLE 2).

Distribution
- The apparent volume of distribution of Secnidazole is approximately 42 L. The plasma protein binding of Secnidazole is <5%.
Elimination
- The total body clearance of Secnidazole is approximately 25 mL/min. The renal clearance of Secnidazole is approximately 3.9 mL/min.
- The plasma elimination half-life for Secnidazole is approximately 17 hours.
Metabolism
- Secnidazole is metabolized in vitro via oxidation by human hepatic CYP450 enzyme system with ≤ 1% conversion to metabolites.
Excretion
- Approximately 15% of a 2 g oral dose of Secnidazole is excreted as unchanged Secnidazole in the urine.
Drug Interactions
Oral Contraceptives
- Concomitant administration of 2 g of Secnidazole with the combination oral contraceptive (OC), ethinyl estradiol (EE) plus norethindrone (NE), to healthy adult female subjects resulted in a decrease in mean Cmax of EE of 29%, and no significant effect on the mean AUC of EE. Administration of 2 g of Secnidazole 1 day before combination OC administration resulted in no significant effect on mean Cmax or AUC of EE.
- Concomitant administration of 2 g of Secnidazole with the combination OC resulted in no significant effect on mean Cmax and AUC of NE (increases of 13% and 16%, respectively). Administration of 2 g of Secnidazole 1 day before combination OC administration also resulted in no significant effect on mean Cmax and AUC of NE.
Ethanol Metabolism
- In vitro studies showed that Secnidazole had no effect on aldehyde dehydrogenase activity.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Nitroimidazoles, which have similar chemical structures to Secnidazole, have been associated with tumors affecting the liver, lungs, mammary, and lymphatic tissues in animals after lifetime exposures. It is unclear if these positive tumor findings in lifetime rodent studies of these nitroimidazoles indicate a risk to patients taking a single dose of Secnidazole to treat bacterial vaginosis.
- Secnidazole was positive in the bacterial reverse mutation assay, but was negative for the rat micronucleus test and mouse lymphoma test.
- In a rat fertility study, females were dosed for two weeks prior to mating until Day 7 of gestation with males that were dosed for a minimum of 28 days before cohabitation. No parental toxicity or adverse effects on mating performance, estrous cycles, fertility or conception was observed at doses of up to the maximum tolerated dose (300 mg/kg/day, approximately 1.4 times the recommended dose based on AUC comparisons).
Clinical Studies
- Two randomized placebo-controlled clinical trials (Trial 1 and Trial 2) with similar designs were conducted to evaluate the efficacy of Secnidazole 2 gram for the treatment of bacterial vaginosis. A diagnosis of bacterial vaginosis was defined as all of (a) the presence of an off-white (milky or gray), thin, homogeneous vaginal discharge; (b) a vaginal pH ≥ 4.7; (c) the presence of Clue cells ≥ 20% of the total epithelial cells on a microscopic examination of the vaginal saline wet mount; (d) a positive "whiff" test (detection of amine odor on addition of 10% KOH solution to a sample of the vaginal discharge); and (e) a Nugent score ≥ 4.
- Trial 1 enrolled 144 non-pregnant female patients aged 19 to 54 years and Trial 2 enrolled 189 non-pregnant females aged 18 to 54 years. Black or African American subjects in both trials were 54%. Efficacy was assessed by clinical outcome evaluated 21 to 30 days following a single dose of Secnidazole. A clinical responder was defined as "normal" vaginal discharge, negative "whiff" test, and clue cells <20%. Additional endpoints included Nugent score cure (Nugent score of 0-3) and therapeutic outcome. A therapeutic responder was defined as a clinical responder with a Nugent score cure. In Trial 2, the endpoints were also assessed at Day 7-14.
- In both trials, a statistically significantly greater percentage of patients experienced clinical response, Nugent score cure, and therapeutic response at 21 to 30 days following a single dose of Secnidazole compared to placebo. Statistically significant results for the endpoints were also achieved at Day 7-14 in Trial 2.
- The percentage of patients with clinical response was also consistently higher in both trials in the Secnidazole arm compared to placebo among all subsets of patients: number of prior episodes of bacterial vaginosis (≤ 3 episodes and ≥ 4 episodes) in past 12 months, baseline Nugent score (score 4-6 and score 7-10), and race (Black/African American and White). TABLES 3 and 4 describe the efficacy of Secnidazole in the treatment of bacterial vaginosis.


How Supplied
- Secnidazole Oral Granules, 2 g, consists of off-white to slightly yellowish granules containing Secnidazole. Secnidazole is supplied in a unit-of-use package containing one packet of granules in an individual carton. Each packet contains 4.8 g of granules containing 2 g Secnidazole. Secnidazole is supplied as follows:
- NDC 27437-051-01 carton containing one unit-of-use 2 g packet.
Storage
- Store at 20°C to 25°C (68°F to 77°F); excursions permitted to 15°C to 30°C (59°F to 86°F).
Images
Drug Images
{{#ask: Page Name::Secnidazole |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel



{{#ask: Label Page::Secnidazole |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
Administration Instructions
- Instruct the patient:
- To sprinkle the entire contents of the packet of Secnidazole onto applesauce, yogurt or pudding and take all the mixture within 30 minutes without chewing or crunching the granules.
- That after consuming the mixture, they may take a glass of water to aid in swallowing.
- That Secnidazole is not intended to be dissolved in any liquid.
- Advise the patient that Secnidazole may be taken without regard to the timing of meals.
Lactation
- Advise women not to breastfeed during treatment with Secnidazole and to discontinue breastfeeding for 96 hours following the administration of Secnidazole. Also, advise a nursing mother that she may choose to pump and discard her milk for 96 hours after administration of Secnidazole and feed her infant stored human milk or formula.
Vulvo-Vaginal Candidiasis
- Advise the patient that use of Secnidazole may result in vulvo-vaginal candidiasis that may require treatment with an antifungal agent.
Drug Resistance
- Patients should be counseled that antibacterial drugs including Secnidazole should only be used to treat bacterial infections. They do not treat viral infections (e.g., the common cold). When Secnidazole is prescribed to treat a bacterial infection, patients should be told that although it is common to feel better early in the course of therapy, the medication should be taken exactly as directed. Skipping doses or not completing the full course of therapy may (1) decrease the effectiveness of the immediate treatment and (2) increase the likelihood that bacteria will develop resistance and will not be treatable by Secnidazole or other antibacterial drugs in the future.

Precautions with Alcohol
Alcohol-Secnidazole interaction has not been established. Talk to your doctor regarding the effects of taking alcohol with this medication.
Brand Names
- Solosec
Look-Alike Drug Names
There is limited information regarding Secnidazole Look-Alike Drug Names in the drug label.
Drug Shortage
Price
References
The contents of this FDA label are provided by the National Library of Medicine.