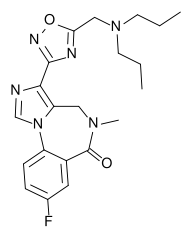
Ro48-6791
 | |
| Identifiers | |
|---|---|
| |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C21H25FN6O2 |
| Molar mass | 412.47 |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Ro48-6791 is a drug which is a benzodiazepine derivative developed by Hoffman-LaRoche in the 1990s.
Ro48-6791 was developed as an alternative to the short acting imidazobenzodiazepine midazolam, for use in induction of anaesthesia and conscious sedation for minor invasive procedures. Ro48-6791 has similar properties to midazolam, being water soluble, with a fast onset and short duration of action. It is 4-6x more potent than midazolam,[1] and slightly shorter acting,[2] and produces similar side effects such as sedation and amnesia.
It was tested up to Phase II human trials, but while it produced less respiratory depression than propofol, it had a longer recovery time and was deemed not to have any significant advantages over the older drug.[3] Similarly when Ro48-6791 was compared to midazolam, it had similar efficacy, higher potency and a shorter recovery time, but produced less of a synergistic effect on opioid-induced analgesia and produced more severe side effects such as dizziness after the procedure.[4] Consequently it was dropped from clinical development,[5] although it is still used in scientific research.[6]
References
- ↑ Dingemanse J, van Gerven JM, Schoemaker RC, Roncari G, Oberyé JJ, van Oostenbruggen MF, Massarella J, Segala P, Zell M, Cohen AF. Integrated pharmacokinetics and pharmacodynamics of Ro 48-6791, a new benzodiazepine, in comparison with midazolam during first administration to healthy male subjects. British Journal of Clinical Pharmacology. 1997 Nov;44(5):477-86. PMID 9384465
- ↑ Hering W, Ihmsen H, Albrecht S, Schwilden H, Schüttler J. Ro 48-6791 - a short acting benzodiazepine. Pharmacokinetics and pharmacodynamics in young and old subjects in comparison to midazolam. Anaesthesist. 1996 Dec;45(12):1211-4. PMID 9065257
- ↑ Wrigley PJ, Elliott DW, Blake D. A phase 2 clinical trial comparing Ro 48-6791, a new short-acting benzodiazepine, with propofol for induction of anaesthesia. Anaesthesia and Intensive Care. 1998 Oct;26(5):509-14. PMID 9807605
- ↑ Tang J, Wang B, White PF, Gold M, Gold J. Comparison of the sedation and recovery profiles of Ro 48-6791, a new benzodiazepine, and midazolam in combination with meperidine for outpatient endoscopic procedures. Anesthesia and Analgesia. 1999 Oct;89(4):893-8. PMID 10512261
- ↑ Gold ME, Todd SA, Spiegler C, Gold JA. When the drug trial fails: an approach to clinical drug studies. AANA Journal. 1999 Dec;67(6):505-12. PMID 10876442
- ↑ Ihmsen H, Albrecht S, Hering W, Schüttler J, Schwilden H. Modelling acute tolerance to the EEG effect of two benzodiazepines. British Journal of Clinical Pharmacology. 2004 Feb;57(2):153-61. PMID 14748814
- Pages with script errors
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Chemical articles without CAS registry number
- Articles without EBI source
- Chemical pages without ChemSpiderID
- Chemical pages without DrugBank identifier
- Articles without KEGG source
- Articles without InChI source
- Articles without UNII source
- Drugs missing an ATC code
- Drugs with no legal status
- Articles containing unverified chemical infoboxes
- Benzodiazepines