Neoplastic meningitis pathophysiology
|
Neoplastic meningitis Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case Studies |
|
Neoplastic meningitis pathophysiology On the Web |
|
American Roentgen Ray Society Images of Neoplastic meningitis pathophysiology |
|
Risk calculators and risk factors for Neoplastic meningitis pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]Associate Editor(s)-in-Chief: Sujit Routray, M.D. [2]
Overview
Neoplastic meningitis refers to the spread of malignant cells through the cerebrospinal fluid space. These cells can be originated both in primary CNS tumors (e.g. drop-metastases), as well as from distant tumors that have metastasized (hematogenous spread). The microscopic pathology of neoplastic meningitis may vary according to the primary cancer involved. Generally, on microscopic histopathological analysis, neoplastic meningitis is characterized by large, hyperchromatic cells.
Pathogenesis
Neoplastic meningitis most commonly occurs secondary to other malignancies, when the malignant cells spread to meningeal layer through reaching to cerebrospinal fluid . The primary region of this malignant cells can be both inside the CNS or outside the CNS, which is going to be considered as distant metastasis.[1]
From primary cancer to the meninges
- It is hypothesized that the primary source malignant cells that contribute to neoplastic meningitis has the capability to secrete a specific enzyme. This is enzyme has the capability to distort blood vessels at a microscopic level, thus helping the metastatic cells to enter blood vessels, traveling through the body and seed in any part of the body they want. These cells can secrete these enzymes to penetrate through blood-brain-barrier, and reach to CSF and thud to brain. As the CSF is the primary source of fluid in the CNS, it continues to carry these metastatic cells all over the CNS, helping them spread in CNS.
- The cancerous cells can also obstruct small CNS vessels, probably due to a more adhesive property that they have. By obstructing small CNS vessels, they can cause cerebral ischemia. They also can make damage to these small CNS vessels while occluding them, and reach to Virchow-Robin spaces by this damage. Through this space, they can reach subarachnoid space.[2]
Invasion routes
- Hematogenous spread occurs either through the venous plexus of Batson or by arterial dissemination. This occurs with arterioles as a result of tumor cells being lodged in vessels that feed the meninges and later causing leakage into the meninges and CSF. This same situation also appears with spinal arteries where leakage of tumor cells is into the nerve roots. Tumor cells may also seed the choroid plexus, where CSF is produced, and ultimately gaining direct access to the CSF. Seeding of the choroid plexus is most common in patients with third and lateral ventricular hydrocephalus.[2]
- Venous spread may occur when intra-abdominal or thoracic pressure increases and venous flow is retrograde which then allows the tumor cells in the systemic venous system to enter the vertebral venous system.
- Centripetal migration from systemic tumors along perineural, invasion of nerve space, or perivascular spaces. Malignant cells can migrate along spinal or cranial nerve (epineurium-perineurium), invade the subpial space, travel along blood vessels into the endoneurial space, or invade the nerve parenchyma.
- Often, the infiltration happens at the base of the brain, dorsal surface, and especially at the cauda equina which is largely due to the effect of gravity. Once in the CSF, malignant cells can extend along the membrane surfaces or spread freely in the CSF and attach to other locations. These cells have the ability to penetrate the pial membrane and invade the spinal cord and cranial nerves.
Infiltration to spinal cord
- Infiltration from the subarachnoid space into the spinal cord occurs primarily along the perivascular tissues that surround blood vessels at the brain entrance. Infiltration from the anterior median fissure, a 3mm deep furrow on the anterior side of the spinal cord, to the anterior horn of the spinal cord, the ventral grey matter of the spinal cord, is found along the central artery. Direct infiltration of the nerve roots is also observed, mostly from the dorsal roots (the afferent sensory root of the spinal nerve) than the ventral roots (the efferent motor root of a spinal nerve).
- With mild infiltration, tumor cells are found diffusely in the subarachnoid space from the cervical to sacral levels. In some cases, there are no differences between spine levels. Infiltration from the subarachnoid space into the spinal cord occurs mainly along the perivascular space of the white matter. However, in some cases, direct infiltration into the spinal cord parenchyma is found together with destruction of the piamater.
Macroscopic pathology
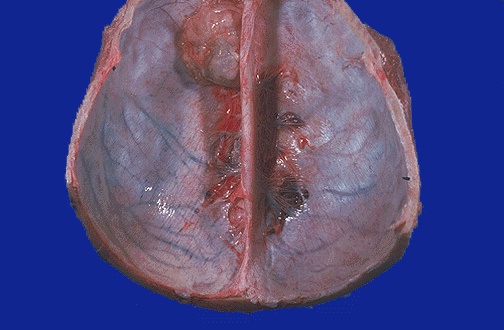
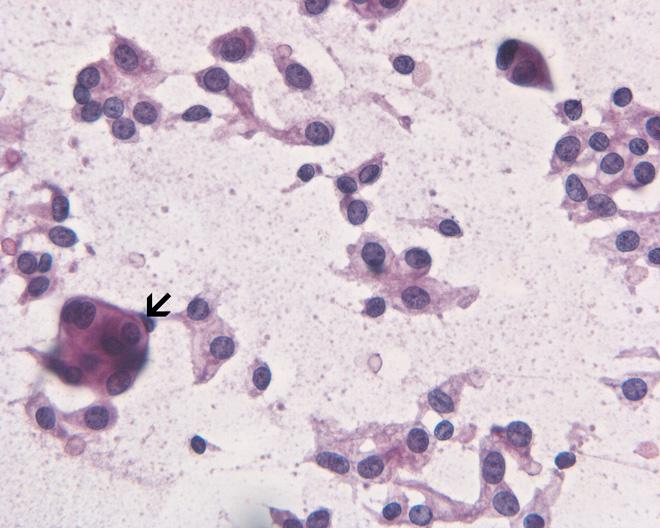
Microscopic pathology
- The microscopic pathology of neoplastic meningitis may vary according to the primary cancer involved. Generally, on microscopic histopathological analysis, neoplastic meningitis may be characterized by: [3]
- Identical histology to any grade I variant meningioma yet contain increased mitoses (4 - 19/10 HPFs)
- Increased cellularity or areas of small cell collections
- Increased cellularity or areas of small cell collections
- Sheet-like growth pattern
- Areas of spontaneous necrosis
- Macronucleoli
References
- ↑ Leptomeningeal metastases. Dr Bruno Di Muzio and A.Prof Frank Gaillard et al. Radiopaedia 2016. http://radiopaedia.org/articles/leptomeningeal-metastases. Accessed on January 20, 2016
- ↑ 2.0 2.1 Causes of neoplastic meningitis. Wikipedia 2016. https://en.wikipedia.org/wiki/Neoplastic_meningitis. Accessed on January 20, 2016
- ↑ Berzero, Giulia; Diamanti, Luca; Di Stefano, Anna Luisa; Bini, Paola; Franciotta, Diego; Imarisio, Ilaria; Pedrazzoli, Paolo; Magrassi, Lorenzo; Morbini, Patrizia; Farina, Lisa Maria; Bastianello, Stefano; Ceroni, Mauro; Marchioni, Enrico (2015). "Meningeal Melanomatosis: A Challenge for Timely Diagnosis". BioMed Research International. 2015: 1–6. doi:10.1155/2015/948497. ISSN 2314-6133.